Meta-context
I wrote a within-cause prioritization report discussing why we should continue studying shrimp sentience and how we can actually do it. Whether you are just curious about how we actually study sentience in invertebrates or are interested in doing or funding such research, this report is for you.
I highly encourage you to read this report through its original format by clicking here. I copied its content here to make it easier to discuss and share, but the formatting is slightly off and some color coding is missing. Enjoy!
Executive summary
Context
- Shrimp are the most killed animals for food production globally: around 440 billion shrimps are farmed (Penaeid species) and 25 trillion shrimps are wild-caught (Sergestidae species) every year.
- Despite their massive numbers, shrimp welfare is poorly considered while their sentience is still a highly debated matter both in the scientific literature and among the general public.
Methodology
- The sentience framework developed by (J. Birch et al., 2021) is used as a reference.
- Multiple considerations are used to argue which criteria from the framework we should prioritize researching, such as their relative weights in the framework, time necessary to study, invasiveness of protocols, etc.
- For each criteria, I present how they are studied and/or how they could be studied.
Key points
- I argue that further studying shrimp sentience is useful, critical and urgent.
- I provide a rule-of-thumb that sentience research costs roughly $100k per criteria.
- I argue that criteria 5 to 7 are of highest priority to study in shrimps, followed by criteria 8 and 4. Criteria 1 to 3 should not be prioritized.
- I recommend grantmakers to submit calls for proposals of research projects related to these criteria (with the help of the paradigms presented in the report) and donors or advocates to express their interest in funding such research, especially to Arthropoda Foundation.
Criteria | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
Priority | Low | Low | Low | Medium | High | High | High | Medium |
Introduction
Context
Shrimps are the most commonly used and killed animals for food production with around 440 billion shrimps farmed and 25 trillion wild shrimps caught annually, yet very little is done to protect them from potentially harmful farming and catching practices (Waldhorn & Autric, 2023). Organizations that promote a better consideration of shrimp welfare, such as Shrimp Welfare Project or Crustacean Compassion argue that shrimps display a sufficient amount of evidence supporting that they may be sentient, and thus be worthy of moral consideration. However, arguments such as from this forum post, this debate, or this conference paper recurrently state that the evidence supporting shrimp sentience only concerns species that are farmed in negligible numbers. The scientific literature that specifically looks at the most farmed and caught species (from the Dendrobranchiata suborder), on the other hand, would not provide enough evidence supporting the presence of sentience markers. Arguments often go even further, claiming that the evidence points to the absence of brain parts, connections, or behavioral proxies necessary for the presence of sentience in these species.
A recent report from (J. Birch et al., 2021) refined a now widely discussed sentience framework and reviewed all of the available evidence of sentience in both cephalopods and decapod crustaceans. Their report recommended that “all cephalopod molluscs and decapod crustaceans be regarded as sentient animals for the purposes of UK animal welfare law”, thus including all shrimps, a recommendation that has effectively been adopted in the UK’s Animal Welfare Act in 2022.
However, it is true that the report’s review of shrimps from the Dendrobranchiata suborder are way less confident than for cephalopods or for other decapods. They note that “there is very little evidence of sentience at present in penaeid shrimps”, but also that they “reject the suggestion that protection should only be extended to specific infraorders of decapod” and insist that “low confidence implies only that the scientific evidence one way or the other is weak, not that the animal fails or is likely to fail the criterion”. Considering that the species from this specific infraorder are the most farmed and the most caught, they also get the most attention for better welfare interventions. However, as we clearly lack evidence of sentience markers in these species, these interventions should be backed up by more solid evidence, either to argue their effectiveness, or to redirect their efforts towards other species.
Defining terms
The use of the term shrimp can sometimes be misleading. For example, some species are sometimes both called shrimps or prawns: the whiteleg shrimp (P. vannamei) is also sometimes called the king prawn, but also the pacific white shrimp, or simply the white shrimp (see this page for more specifics). Taxonomically, the classification is very different. The vast majority of species that we farm, catch and eat are from families within the Dendrobranchiata suborder. These include all species that are commonly named either shrimps or prawns. Those we farm are mostly Penaeidae shrimps (especially P. vannamei), and those we wild-catch are mostly Sergestidae (especially A. japonicus). What we call “true shrimps” are species from another infraorder: Caridea. These species are more used as ornaments in aquariums and are more present in the scientific literature. Thus, in this report, I will simplify the use of these terms.
- Caridea shrimps will be called true shrimps.
- Penaeidae shrimps will simply be called shrimps.
- Sergestidae shrimps will not be specifically discussed in this report. However, as we can consider that most of this report will also be applicable to these species, I’ll consider them in the term shrimp as well.
Taxon | Name in this report | Relevance | |
Dendrobranchiata | Penaeidae | Shrimps | Most farmed species |
Sergestidae | Most caught species | ||
Pleocyemata | Caridea | True shrimps | Most studied species |
You may also observe that the most farmed species of shrimps are either called L. vannamei (Litopenaeus) or P. vannamei (Penaeus). They are the same species. Both genus terms (P. or L.) are used in the literature, with P. vannamei being a more historical one, and L. vannamei being used more recently following a 1997 classification update. However, this update has not been universally accepted, and the use of P. vannamei still seems more appropriate (Yang et al., 2023). For example, the FAO and Rethink Priorities also prefer the use of P. vannamei, so I’ll stick to it too.
Poll results
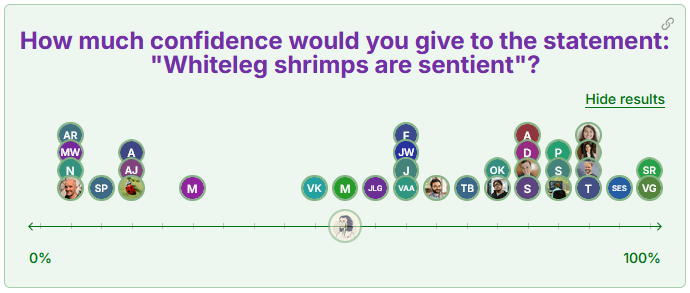
In a previous EA Forum post, I asked you this question:
We can clearly see that the confidence follows a bimodal distribution: participants were either very skeptical or pretty optimistic, in a broader range, that shrimps may be sentient.
Here are a few recurring thoughts that I got from comments and discussions with respondents:
- The confidence of respondents is usually pretty volatile. They know that evidence is scarce: they easily allow their confidence to change based on new evidence.
- Most rely on experts' opinions: the direct evidence and literature reviews are not so accessible. The consideration of all decapods as sentient (J. Birch et al., 2021) is well known, but the specific case of Dendrobranchiata not so much.
- Analogous brain structures to those of vertebrates are relevant, but behavioral markers like decision-making or analgesia preference seem more convincing.
- Even though evidence is lacking, a lot of the optimistic confidence is based on the fact that shrimps are evolutionary close to the other decapods (or even insects) that are considered sentient more consensually, or that at least show more evidence for sentience criteria.
The translatability of sentience across species is a highly debated subject. Critiques on shrimp sentience often argue that Dendrobranchiata are too evolutionary far from other decapods to value the translatability of sentience markers across all decapods. The scientific literature is definitely not fixed on this subject, but it seems way more reasonable to expect shrimp sentience criteria to actually be very closely related to other decapods than to consider them as outsiders (Browning & Veit, 2022; Walters & Williams, 2019).
The fact that many respondents are so optimistic about the sentience of shrimps although the evidence is very limited can also alert us. Interventions to improve shrimp welfare already allocate millions of dollars annually and claim to help billions of individuals from long minutes of potential suffering ((Shr)Impact, n.d.). If many are so uncertain about their sentience and/or the resulting effectiveness of such interventions, more evidence will be highly valuable. Still, the accumulating evidence of strong conservation of fundamental mechanisms that induce and maintain pain-like alterations should encourage us to err on the side of caution, which can also be held true towards shrimps (J. Birch, 2024; Walters & Williams, 2019).
Preamble assumptions
While sentience is still a vast field of study in which we are uncertain about what it exactly is by itself, here are some assumptions that this report will make without further questioning:
- For a discussion about the definition of sentience, please refer to (J. Birch et al., 2021) and (J. Birch, 2024).
A sentient being (in the sense relevant to the present framework) is a system with the capacity to have valenced experiences, such as experiences of pain and pleasure. |
- Sentience, in its narrow sense (including valenced experiences as presented here), is regarded as a moral and legal foundation for animal welfare (J. Birch, 2024; Browning & Veit, 2022), no matter to which degree and intensity (Browning, 2026; Fischer, 2023).
- Sentience research allows for increased awareness and advocacy (de Oliveira et al., 2024; Tookey, 2024).
- Shrimps are considered investigating priorities (J. Birch, 2024).
A system S is a sentience candidate if there is an evidence base that (a) implies a realistic possibility of sentience in S that it would be irresponsible to ignore when making policy decisions that will affect S, and (b) is rich enough to allow the identification of welfare risks and the design and assessment of precautions.
A system S is an investigation priority if it falls short of the requirements for sentience candidature, yet (a) further investigation could plausibly lead to the recognition of S as a sentience candidate and (b) S is affected by human activity in ways that may call for precautions if S were a sentience candidate. |
Goals
Consequently, I assume that further studying shrimp sentience is: useful, critical, and urgent. However, not all studies interested in shrimp sentience may have the same impact on the global consideration of shrimps as sentient animals, that’s why we may want to prioritize research on some criteria more than others so that we can update our confidence on the global consideration of shrimp as sentient (or not sentient) animals as effectively as possible.
This report does not discuss cost-effectiveness of shrimp interventions. The aim of this report is to help both researchers and grantmakers look for studies that have the potential to advance the science of shrimp sentience the most, as well as those interested in how we actually study sentience criteria. It is then these necessary sentience studies (and the broader field of welfare studies) that will help us build confidence about the cost-effectiveness of shrimp (and other invertebrates) interventions.
If the evidence points to the absence of sentience, then money and energy put into shrimp interventions might be better spent elsewhere. If the evidence confirms the presence of sentience, then it could better promote advocacy for shrimp interventions, saving time, money, and billions of lives from long minutes of suffering all over the world.
Methodology
Sentience Framework
This report will be based on the sentience framework developed by (J. Birch et al., 2021). Here’s how it works:
- Eight criteria are looked for in the literature for a given taxon or species, each being indicators of traits thought to be relevant for the presence of sentience in any animal
- Each criterion is given a confidence level considering "both the amount of evidence for a claim and the reliability and quality of the scientific work, which were agreed through discussion among the authors"
|
- Finally, the number of criteria that reached at least a high confidence builds the final evidence for sentience:
|
Here is the confidence (J. Birch et al., 2021) had for each criteria in various crustacean decapods groups:
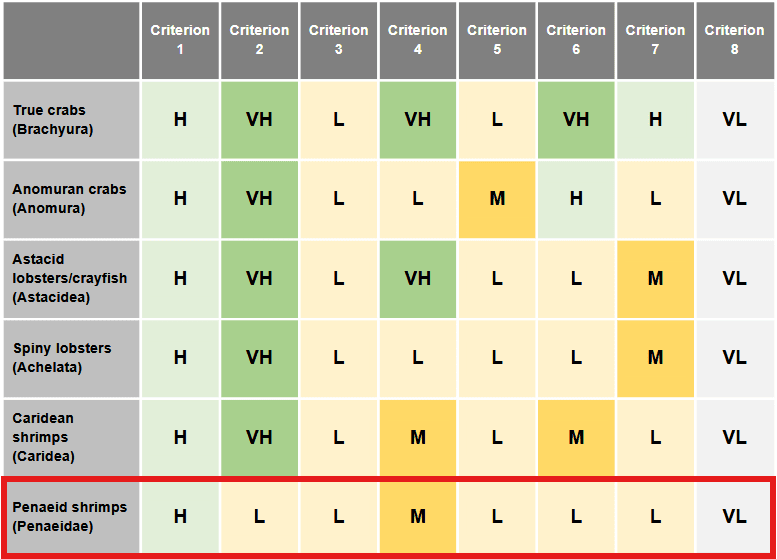 |
Table 3 from (J. Birch et al., 2021). A summary of the evidence of sentience in decapods. The colours and letters represent our confidence level that the criterion in question (column) is satisfied by the order (or orders) of animals in question (row). VH (dark green) indicates very high confidence, H (light green) indicates high confidence, M (dark yellow) indicates medium confidence, and L (light yellow) represents low confidence. Since we have not found evidence to support criterion 8 in any decapod, we have used the category of very low confidence (VL, light grey) in this case. Importantly, low/very low confidence implies only that the scientific evidence one way or the other is weak, not that the animal fails or is likely to fail the criterion. |
To put this framework into perspective, we have to note that its aim is not to assess sentience directly, nor does it claim that any species is or is not sentient. What this framework claims is that, given a broad range of relevant scientific evidence, we can more or less expect sentience to be present. The more evidence we have for each criterion, the more confident we can be on the total expectation of sentience, each criterion being considered equally as important. As (Fischer et al., 2025) phrases, “the greater the number of satisfied criteria, the more likely it is that the organism being evaluated is sentient and the greater the case for precautionary measures”. The expected presence of sentience is then argued for legislative (and moral) protection of the concerned species.
However, as argued by (Fischer et al., 2025), this choice of equal consideration for each criteria may be permeable to many false positives, meaning that it may be too inclusive (non-sentient animals might get falsely considered sentient) because all criteria “don’t have the same evidential value” and they “could adopt a more sophisticated evolutionary approach to trait analysis”. In other words, we can expect some traits relevant to some criteria to be more or less conserved across the animal kingdom and/or to be more or less relevant for the presence of sentience. In a commentary, (Walters, 2022) also argues that “criteria 1 to 4 and 6 represent features that are either expressed too ubiquitously in the Animal Kingdom to be of much use in inferring pain or can be mediated straightforwardly by automatic mechanisms without a need for emotional contributions”. Birch further explains his precautionary framework in his book (J. Birch, 2024), and insists that we need to “err on the side of caution in the face of radical uncertainty about sentience”. The main idea is that false negatives (considering an animal not sentient while they are) would allow high amounts of gratuitous suffering, while false positives (considering an animal sentient while they are not) would not. Thus, the framework benefits from being more inclusive than strict. (J. Birch, 2024) also develops the idea that there is a “very wide ‘zone of reasonable disagreement’ and a good framework for making decisions will respect all the views that lie within that zone” in which their framework lies.
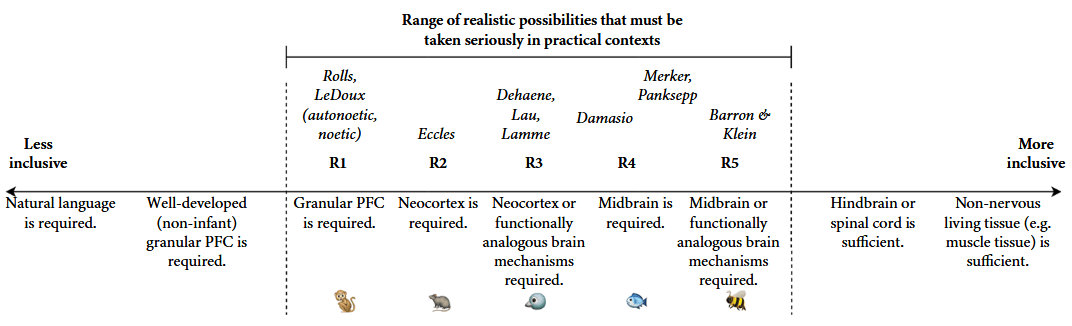
Fig. 6.2 from (J. Birch, 2024). A proposed scientific meta-consensus on the neural system requirements for sentience (the capacity for valenced experience). There is no consensus about which position within the reasonable range is correct. However, a meta-consensus can form around the idea that positions R1–R5 describe realistic possibilities that must be taken seriously, given current evidence. There can also be a meta-consensus on the ordering of these views from less inclusive to more inclusive and on the challenges facing any view that falls outside this range. Figure © Jonathan Birch. |
(Fischer et al., 2025) recognizes that the framework’s “original version was meant to be a simple instrument for making policy recommendations” and that the authors themselves “have quite nuanced views about the relative evidential value of each criterion”. However, they also argue that “precautionary measures have costs: it can be expensive (in many senses of ‘expensive’) to implement and enforce them. If we can prevent an excessive rate of false negatives with fewer false positives, then we should, as otherwise the costs of precautionary measures would not be justified. So, we have reason to try to refine the Birch et al. framework so that it still prevents an excessive rate of false negatives but without as many false positives.”
While (Fischer et al., 2025) did not provide a revised version of the framework yet (their publication was only meant to promote discussion), this current report on research prioritization for shrimp sentience is a perfect use-case for adopting these suggestions. Thus, I will not propose an entirely revised framework either - that would be completely beyond scope - but I will argue prioritization for each criteria using both (J. Birch, 2024; J. Birch et al., 2021) and (Fischer et al., 2025) considerations.
Prioritization
There are numerous ways to think about prioritization in this report. I present here the various considerations that I will use to argue prioritization for every criteria.
Relative weights
First, as we mentioned in the introduction, we may weigh each criteria differently based on how much they would help us build confidence on the overall consideration of sentience.
To give concrete examples, here are my a priori confidence levels for each criterion, meaning how confidently we could already expect shrimps to actually validate these criteria, even if we don’t have any evidence:
Criterion | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| A priori confidence | High | High | High | Medium | Low | Low | Medium | Low |
This table does not mean that I expect shrimps to invalidate criteria 5, 6 or 8: only that, given the current limited evidence, we may still be more confident than what the evidence currently shows for a few criteria, notably the first ones. In other words, we may have good reasons to believe that shrimps will probably validate the first four criteria (they already do for criterion 1) and criterion 7. The arguments will be discussed more thoroughly in each criteria’s sections, but they basically rely strongly on evolutionary pressure, so they are present in almost all animals, and they are all present at high confidence in other decapods or other relatively closely related animals (cephalopods, insects, etc).
However, we can also argue that it is essentially because these criteria are overall more ubiquitous in the animal kingdom that they also carry less weight for the global sentience framework, as mentioned by (Walters, 2022). Similarly, we could consider the lower criteria to be prerequisites for the higher behavioral criteria: if shrimps validate with high confidence criterion 4 for example, it would be highly improbable that they do not validate criterions 1 to 3. As another example, criterion 8 would also imply the validation of criteria 1 to 4 and 7, at least. Thus, I would also add a relative weight on each criteria (which will be further discussed in each criterion section):
Criterion | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| A priori confidence | High | High | High | Medium | Low | Low | Medium | Low |
Weight | Low | Low | Low | Medium | High | Medium | High | High |
Time
Now, we can think about prioritization as very short-term, i.e. what are the very first studies that we should perform to validate as many criteria as quickly as possible, whichever ones;
Or we can think about medium- to long-term priority, i.e. what are the criteria that will allow a more consensual view on the global classification of shrimps in the sentience framework.
- Short-term prioritization would argue that, because we have good reasons to think shrimps will validate criteria 1-4, confirming these criteria are high-priority. It would be an “easy” way to add some high/very high confidence to the list quickly. On the other hand, validating them would not weigh much on the global consideration of sentience. On top of that, if multiple studies consistently show that shrimps fail these criteria, it would take much longer to check whether the protocols were done properly or if they simply failed to demonstrate the absence of these properties. In other words, we can argue that it could take much longer to properly show that a species fails a criterion than to show that they validate it. At the end, studying higher-weighted criteria (5-8) would still be necessary later on, no matter the early outcomes of criteria 1-4. Practically however, short-term prioritization could also promote more immediate funding for further research. This could be taken into account, but it is a bit out of scope of this report.
- Medium- to long-term prioritization, however, would argue that because we can already expect shrimps to validate criteria 1-4, we should prioritize studying criteria of higher incertitude first (5-8). If criteria 5-8 are shown to validate with high confidence, we have much more reasons to consider shrimps as sentient even if criteria 1-4 still lack data for two main reasons: because we already have reasons to expect 1-4 to validate as well, and because 1-4 are direct or indirect prerequisites to criteria 5-8. Hence, if criteria 5-8 are confidently validated, then other criteria are almost certain to validate as well (they depend on them). Thus, in the long run, if criteria 5-8 are validated with more confidence, then we can probably perform fewer studies on criteria 1-4. However, if 5-8 fail to validate first, then we can prioritize criteria 1-4.
Considering these scenarios, I will tend to focus on long-term prioritization as our goal is to update our confidence on the global sentience consideration, not on specific criteria.
Inter-dependability of criteria
As mentioned already, not all criteria can be considered independent. For example, if an animal fails criterion 1 or 2, it is very much likely that all other criteria will fail as well, as later criteria depend on them.
Criteria 1 and 2 are basically prerequisites for every other criteria. They are explicit prerequisites for criterion 3, and indirect prerequisite for all others. Similarly, criterion 7 can be considered a prerequisite for criterion 8, also maybe for criterion 5. The contraposition of this claim is that if we confidently validate higher criteria, then lower criteria are extremely likely to validate as well.
For example, the fact that criterion 8 would validate but with a mechanism other than nociceptors and integrative brain regions is highly unlikely (and would make zero evolutionary sense for shrimps), so a high confidence in criterion 8 would also imply higher expected confidence in at least criteria 1, 2, 3, and most likely 4, 5 and 7 as well, as argued before.
Costs
To estimate the costs of the studies suggested in this report, there are two ways of proceeding: we can either estimate the cost of building a whole “sentience lab” from scratch, or estimate the funds necessary for academic labs to appropriate the subject.
In this report, although some groundwork would be necessary to adapt some protocols for crustaceans and more specifically for shrimps, most techniques suggested are routinely used in most life science (and more generally neuroscience) institutes and labs, which means that the infrastructures and the equipments are already available - for example, we can simply expect to allocate money to use an electrophysiology or microscopy platform, so we don’t have to pay for a brand new electrophysiology setup or an electronic microscope ourselves. Furthermore, as most criteria involve the use of narcotics, illicit and/or dangerous molecules, such as opioids (e.g. morphine, fentanyl) or other anaesthetics, it would be much easier for academic labs to acquire and use these kinds of molecules safely.
In this case, most of the funds would go towards researchers' wages: PhDs, postdocs, research engineers, etc. Personnel usually represent around 80% of a research project’s budget.
Thus, budget estimations are not so much about what or how we study these sentience criteria, but rather how long it would take. For example, we could assume that $100k approximately covers one year of research for one PhD or postdoc + material, supplies, travel, etc. We can expect that most criteria would need around a year to be studied properly (preliminary experiments, tests, analysis, writing, peer-reviewing, etc).
However, as mentioned in the corresponding criteria, we can see that criteria 1 to 3 are especially complex and demanding: we don’t have much groundwork to study shrimp brains, and electrophysiology and imaging techniques are especially long and expensive to set up, while also being more demanding in expertise from trained scientists. Criteria 4 to 8, on the other hand, involve much (relatively) simpler behavioral setups. These “higher” criteria involve other challenges: they are especially prone to confounding factors, and thus demanding in controls (negatives, positives, yoked controls, etc), and their results should be thoroughly discussed with peers, but it still seems that these higher criteria should be manageable more quickly compared to ambitious neuroanatomical research projects. We should mention that, as we lack important groundwork for many of these criteria still (e.g. associative learning protocols, analgesia preference protocols, etc) this rule of thumb might be pretty optimistic: we could expect behavioral protocols to be much more demanding than we expect, which could significantly extend the budget and the time needed to publish such studies.
This rule of thumb ($100k/criteria/species), also used by Bob Fischer (personal communication), still gives a good idea of how much it would cost to advance the science of sentience in some species. However, this rule of thumb does not tell us how much more confident we would be towards the criteria if one researcher worked on them for a year: only the results of these studies could give us the answer. Ideally, they would be sufficient. In reality, we may need more.
As a concrete example, a rather ambitious £118k research project from the University of Stirling led to the publication of an article (a preprint yet) exploring indicators of consciousness in shrimps which suggests that electric shocks prior to dumping shrimps in ice is an effective way to reduce signs of consciousness and potential suffering (Somerville et al., 2026). They did not directly study sentience criteria (although the study is somewhat related to criteria 1, 2, 3 and 6), but it definitely helps reduce uncertainties about the relevance of shrimp welfare interventions.
Number of animal studied and invasiveness of protocols
Utilitarianism may argue that humanely euthanising a few hundred shrimps for the sake of studying them is not a big deal since it will potentially help billions others, but it is still important to think about if and how we can still reduce this number (as per the 3R considerations).
Similarly to the previous considerations, it’s mostly the first 3 criteria that necessarily involve invasive procedures and euthanising animals to retrieve and study their nervous system in detail. Criterion 4 to 8 mostly involve behavioural paradigms, and while animals may be subjected to surgical procedures or brief painful stimuli, they should be able to retire healthy.
All in all, considering that criteria 1 to 3 are the most complex (as it will be discussed in the report), that they almost necessarily involve euthanising animals, that they can be considered as the lowest weighting criteria, that they are the most expected to validate, that they are the most common in the entire animal kingdom and that they are prerequisites for the next criteria, their prioritisation is highly impacted downwards. Behavioral cues for criteria 4 to 8, on the contrary, will globally be highly prioritized for the exact same (but opposite) arguments.
Labs
Finally, although not many labs produce research relevant for the specific study of sentience in shrimps, there are many labs studying crustaceans more generally that have most of the equipment and expertise already available.
Here are a few examples:
- University of Kentucky, US, hosts Dr. Robin L. Cooper’s lab which studies sensory systems of crayfish
- Mahidol University, Thailand, has a Center of Excellence for Shrimp Molecular Biology and Biotechnology (CENTEX Shrimp) which studies farmed shrimp health and the environmental impacts of shrimp farming
- Queen’s University, UK, hosts Pr. Robert Elwood’s lab, which has been highly valuable for this field of research over the last decades, and is cited many times in this report for their studies on crabs
- Kentucky State University, US, hosts an Aquaculture research division which studies shrimp farming practices
- University of Arizona, US, hosts the Aquaculture Pathology Lab, which studies Penaeid shrimps health
- University of Gothenburg, Sweden, hosts the Aquatic Animal Welfare research group led by Pr. Lynne Sneddon, which also works on crustaceans welfare, e.g. (Kasiouras et al., 2026).
- University of Stirling, UK, hosts an Institute of Aquaculture, which currently has two projects (1 & 2) closely related to sentience markers in P. vannamei, funded by Coefficient Giving
- Federal University of Rio Grande do Norte (UFRN), Brazil, hosts the Sensory Ecology Lab which studies, among many other things, crustaceans behavior, for example shelter preference in prawns
- All other labs whose studies are mentioned throughout this report
- And many more
The message I want to bring here is that research labs exist, the infrastructure exists, the expertise exists, appropriate working conditions exist, etc. Labs should be able to appropriate research projects for any sentience criteria they are the most relevant to study.
Criterion 1: Nociceptors
Definition
The animal possesses receptors sensitive to noxious stimuli (nociceptors).
Summary
- Current confidence: High
- Reason: High indirect evidence for every decapod but low direct evidence
- Priority: Low priority
Description
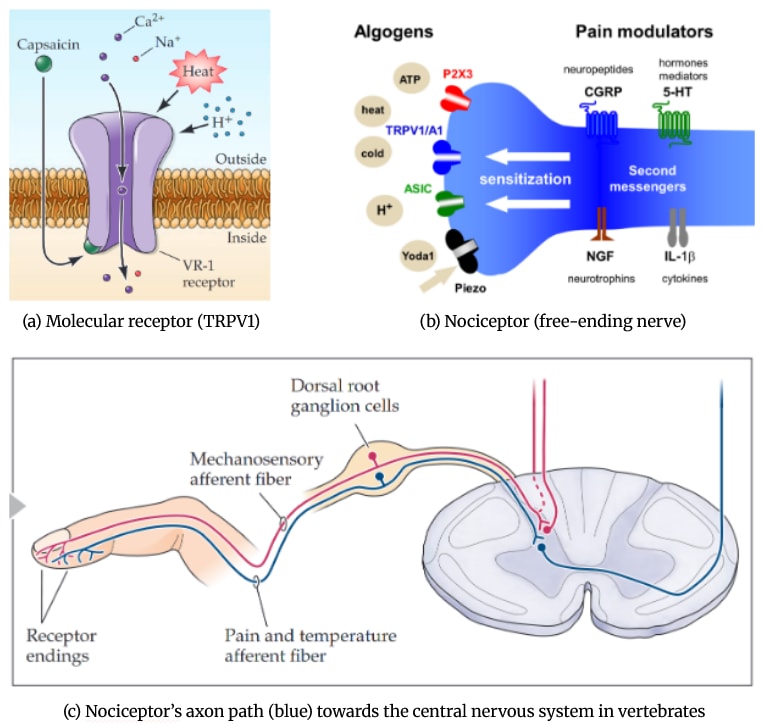
Nociceptors, as defined by the International Association for the Study of Pain (IASP, 2020), are “high-threshold sensory receptors of the peripheral somatosensory nervous system that are capable of transducing and encoding noxious stimuli”. In other words, they are the receptors specialized in detecting stimuli that have the potential to be damaging to the tissues they are located in (cuts, burns, noxious chemicals, etc).
We have to be careful however when using the term “receptors”. The “first-line” receptors are the transmembrane ion- or G-coupled molecular receptors (figure 1a), also sometimes wrongly called nociceptors. Those molecular receptors translate various types of information (e.g. thermal, chemical, acidic, mechanical) into electrical information into the nervous system through a process called transduction.
These molecular receptors are expressed in all kinds of cells, but are mainly present in nerve cells that are specialized in detecting noxious stimuli (such as free-ending nerves). These specialized nerve cells thus express a high quantity of these molecular receptors and have a particularly high threshold to be activated: they will only be activated in case of particularly high amounts of heat, cold, pressure, etc. In other words, they are specialized in detecting stimuli that could potentially be harmful for the integrity of the tissue where they are located (or anything that is threatening the survival of the individual, basically). It is those specialized neuron endings that are called nociceptors (figure 1b).
These nociceptors will then extend their axons (which will usually be called a fiber) towards the central nervous system to transmit the “nociceptive signals” (figure 1c). We could also call the whole neuron a nociceptor (instead of nociceptive neuron) for the sake of simplicity. The fate of the nociceptive signal that goes through nociceptors varies greatly between species, but as their goal is to signal threats, nociceptive signals usually end up in integrative regions where they induce either reflexes (immediate motor avoidance responses, mostly in the spinal cord or an equivalent) and/or more complex behavioral responses after being processed in the brain (or an equivalent ganglion).
Figure 1. The three orders of magnitude of nociception. (a) A molecular receptor specialized in the detection of heat in many animals. Figure from (Purves et al., 2017) (b) A nociceptor (free-ending nerve) expressing various molecular receptors. Figure from (Giniatullin, 2020) (c) The path nociceptors mostly take in vertebrates (blue line): first-line nociceptors are activated by noxious stimuli in the periphery and reach the central nervous system to transmit “nociceptive signals” to second-line neurons, which will then go towards the brain. Figure from (Purves et al., 2017). |
Schematically, the noxious stimulus information goes like this:
Molecular receptors are activated by noxious stimuli → Nociceptor is activated through transduction (produces an action potential, sending an electrical signal) → Nociceptor’s axon (fiber) sends the signal to the central nervous system.
This whole process is what is called nociception. The (IASP, 2020) definition states: ”The neural process of encoding noxious stimuli”.
Now, what we know is that molecular receptors are extremely well conserved through the animal kingdom (Zhai & Wang, 2023). For example, receptors from the TRP or ASIC families can be found in basically all animals, even sponges (which don’t have a proper nervous system), jellyfish, etc. They are not exactly identical proteins, but they are sufficiently conserved to have the same structure and very similar properties.
Nociceptors as a whole, on the other hand, have more properties that need to be satisfied: they also need to have a high threshold of activation, and should be able to conduct their electrical information towards the central nervous system (or equivalent). However, nociceptors are also extremely common in the animal kingdom: they may take other shapes and forms, but as far as we know, all vertebrates and almost all invertebrates have some sort of specialized neurons for the detection of noxious signals (Smith & Lewin, 2009; Sneddon, 2018).
How to study
To properly identify functional nociceptors, we mainly need electrophysiological recordings of nerve cells under noxious stimuli. Oftentimes, thin brain slices of euthanized animals are kept in a specific ionic solution so the slice can still function for a few hours and nerve cells can still fire normally when we apply various stimuli (e.g. an irritant chemical, heat, etc), which we measure using electrodes. This can also be done in vivo using anesthetized animals instead and putting electrodes in their head directly. If noxious stimuli induce action potentials specifically at high thresholds, then we know that specialized nerve cells are present (nociceptors). The type of nerve cells that are going to be activated depends on the stimuli we give: if the stimulus is heat, then heat-sensitive nociceptors will fire. It’s because we input various noxious stimuli that we output the activation of noxious-specialized nerve cells, and that’s how we know they are nociceptors. Also, as the molecular receptors are very well conserved, we know that what is considered a “noxious” stimuli is usually very similar for all species (e.g. temperatures above 42°C and below 15°C, chemicals such as formalin, capsaicin, etc).
We could then go further and try to identify exactly which neurons are specifically activated at high thresholds by using higher resolution techniques, which are also more complex and expensive. Instead of using large open electrodes near a group of nerves, we can measure the electrical activity of a single neuron using patch-clamp electrophysiology. The issue with single-neuron electrophysiology is that, for exploratory research, we don’t know which neuron to look for: we don’t want to randomly choose one fiber and hope it’s from a nociceptive neuron. To get better precision, we can add a calcium imaging technique for example: a calcium marker is expressed in the nervous system, and when noxious stimuli are applied, nociceptive neurons fire, which makes them glow. We can then patch-clamp those fluorescent neurons to get a better chance at measuring specific nociceptive neurons individually (if that sounds easy: it’s definitely not!). However, it’s not mandatory to identify the presence of individual nociceptors: we just need to know that nociceptors are present, so low resolution electrophysiology is good enough.
Protocols summary
- Electrophysiology
- Extracellular (low resolution)
- Patch-clamp (high resolution)
What we currently know
As mentioned by (Walters, 2018), we surprisingly have hardly any direct evidence of primary nociceptors in crustaceans, but lots of indirect evidence supports their existence.
Basically, any defensive behavior elicited by noxious stimuli is indirect evidence for functional nociceptors by definition. While nociceptors might take another shape, if the animal can detect and react to noxious stimuli (whether reflexively or consciously, as we will discuss more in other criteria), it necessarily means that noxious receptors are present. Nociceptors are the norm rather than the exception in the animal kingdom.
To approach direct evidence, we have a few electrophysiological studies in crayfish. For example, (Puri & Faulkes, 2015) showed an increased extracellular activity of antenna nerves when 60°C water was applied to the antenna. However, it is not clear whether those neurons could fire at non-nociceptive temperatures as well or not, so it was not very specific. Other studies also fail to clearly demonstrate the presence of nociceptors (as it was not their goal), but they still give some insights into the physiology of noxious stimuli detection (J. Birch et al., 2021). Accordingly, (Elwood, 2025) argues that these results show “clear evidence of neuronal responses to heat in the isolated antennule that was consistent with the idea of nociception and, thus, the presence of nociceptors”.
Some indirect evidence of nociceptors in decapods also lies in transcriptomics (the analysis of what genes produce in the cells). In species of lobster, crayfish and crabs have been identified various homologues receptors from the TRP family, especially in the antennae, their main sensory organ.
Other indirect evidence can be made from behavioral evidence (which will be further discussed in later criteria). As noted by (J. Birch et al., 2021), nociceptors also have high survival value, so it is very unlikely that specific crustaceans taxa lost these features.
Concerning shrimps, it seems that no direct nociceptors study has been performed yet. However, considering all the indirect evidence presented here, the presence of nociceptors is very likely as well, as per the “high confidence” from (J. Birch et al., 2021).
What we would need
Considering this criterion already has high confidence, we can consider this criterion as low priority. However, to get definite proof, we would need direct evidence of nociceptors by electrophysiological recordings in shrimps. Such research would probably go in pairs with criteria 2 and 3.
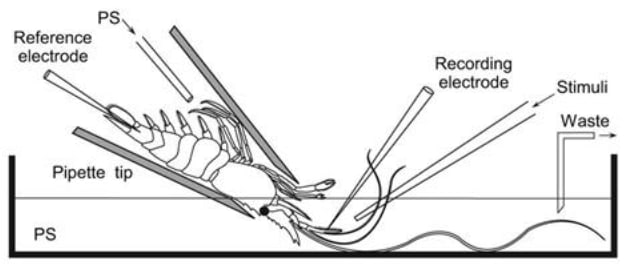
For example, (Machon et al., 2016) adapted a method called electroantennography (EAG) to measure the activity of individual neurons in the antenna of a “true” shrimp species (Palaemon elegans) while keeping the animal underwater, his natural condition (it is otherwise usually done aerially).
While this study was interested in chemoreceptor neurons to study olfaction, this technique could well be adapted to study nociceptive neurons. The authors also state that this method “can easily be adapted to other marine or freshwater decapods”, and because Palaemon elegans shrimps are usually pretty small (a few cm), we could probably easily adapt this technique to whiteleg shrimps as well.
As another example, (Somerville et al., 2026) recently measured electrical activity of the supraesophageal ganglion in P. vannamei using electrodes that were glued to keep them in place while the animal was free to move after waking up from anaesthesia. This study focused on the potential desensitizing effects of electrical shocks before euthanasia in an ice slurry, but we could easily adapt this protocol to observe the presence of nociceptors. See (Puri & Faulkes, 2015) for an example of a similar electrophysiology protocol in crayfish looking specifically for nociceptive electrical responses: in their study the antenna was cut at the base, an electrode was placed on one of the two nerve branches of the antenna, and the tip of the antenna was briefly exposed to hot water (60°C) to elicit nociceptive responses.
Criterion 2: Integrative brain regions
Definition
The animal possesses integrative brain regions capable of integrating information from different sensory sources.
Summary
- Current confidence: Low
- Reason: Lack of data for shrimps but high evidence for every other decapod
- Priority: Low priority
Description
As its title suggests, this criterion ensures that sensory inputs (mechanical, visual, thermal…) are integrated together in some part of the nervous system, usually in a centralized brain part or ganglion, for the individual to correctly adapt and react to their environment. This rather large description will work in combination with criterion 1 to construct criterion 3.
How we study this criterion will hugely depend on the species of interest. While we may find very structured and specialized integrative brain regions in vertebrates, integrative regions in invertebrates may be less structured, less specialized, and thus less understood. However, sensory integration is a very basic feature of multicellular organisms, and can be considered ubiquitous in the animal kingdom. For example, cnidarians (jellyfish and polyps), which don’t even have any form of centralized nerve system (they have a “nerve net”), still display sensory neurons that are able to integrate photo-, chemo- and mechanosensory cues in a “complex and hierarchical manner that that was dependent on specific combinations of cues, an indication of multisensory integration” (S. Birch & Plachetzki, 2023). Hence, this criterion does not specifically predict the presence of sentience, but rather constitutes a prerequisite for all other criteria.
How to study
This criterion is highly interdisciplinary and usually implies taking account of many, often indirect, sources of evidence.
Indirect behavioral evidence can be insightful enough and may avoid us from proceeding with highly invasive, cost- and time-heavy protocols. Indeed, this criteria is arguably an indirect prerequisite for all “higher” criteria 5 to 8 and a direct prerequisite for criterion 3. If the animal of interest somehow does not possess integrative brain regions, it is very likely that all other criteria will fail. Similarly, if the animal of interest validates other criteria, it is very likely that they might validate this one as well. Thus, it benefits from evidence for most other criteria as well. However, it might still be useful to study specifics of shrimps’ brain regions to better understand how their environment may impact their internal states.
Direct evidence of integrative brain regions involves huge amounts of neuroanatomical techniques (immunohistochemistry, neuron tracing…) as well as imaging techniques (microscopy, optogenetics…), electrophysiology, connectivity mapping, etc. To get a glimpse of how much is involved for such projects in decapods, you can take a look at (Utting et al., 2000) and (Meth et al., 2017).
Protocols summary
- Neuroanatomical mapping: IHC, Neuron tracing, Electrophysiology, etc.
- Imaging: Microscopy, MRI, Tomography, etc.
What we currently know
The shrimps’ case for this criterion is of particular interest as their hemiellipsoid body, an important region involved in learning and memory, seems significantly smaller than for other decapods. However, as for other species that show relatively small hemiellipsoid bodies, such as crayfish and lobsters, other accessory lobes (deutocerebrum) regions seem to compensate for these sensory integration. “It is possible that integration of information can be done either primarily in the accessory lobe, as in crayfish and lobsters, or primarily in the hemiellipsoid bodies, as in crabs and some shrimp species” (J. Birch et al., 2021).
In his book, (J. Birch, 2024) further explains:
At the same time, it also revealed miniaturization and weak differentiation of those regions in the world’s most commercially important farmed shrimp, Penaeus vannamei (...). Are these miniaturized hemiellipsoid bodies still performing important functions for the animal? Since nervous tissue is energetically expensive to maintain, this sort of diminution is what we expect to see when a particular system is no longer functionally significant. Rebecca Meth and colleagues suggest one possible explanation: the size of the hemiellipsoid bodies is largely driven, they suggest, by the richness of the olfactory input they receive, but the sensory ecology of whiteleg shrimps in the wild gives reduced importance to olfaction, particularly in comparison to terrestrial decapods, and increased importance to vision in low-light conditions (i.e. turbid water). So, it seems the lineage has evolved to invest more energy in large visual neuropils and less in the hemiellipsoid bodies. Could it be that some crustacean lineages have evolved capacities for learning and memory and then lost them? Indeed, could some lineages have evolved sentience and subsequently lost it? That is a possibility we need to take seriously, but one that is very hard to establish with confidence, given the lack of agreement on markers of the absence of sentience. Ultimately, we handled this complex picture by reporting ‘low confidence’ that penaeid shrimps have functional, integrative brain regions. It was a difficult case, because the immunostaining work shows that whiteleg shrimp do have homologues of such regions, while also casting doubt on their functionality. A similar integrative centre linked to learning and memory—the mushroom body—is well documented in insects. Indeed, Strausfeld et al. used their results to argue for the homology of the insect mushroom body and the decapod hemiellipsoid body. The evidence for sentience in decapods cannot, therefore, be disentangled from the emerging picture of insects as sentience candidates (...). |
While strong evidence for shrimps is lacking, it still seems pretty likely that they do possess integrative brain regions, especially since other decapod species that also have smaller hemiellipsoid bodies display high evidence for this criterion.
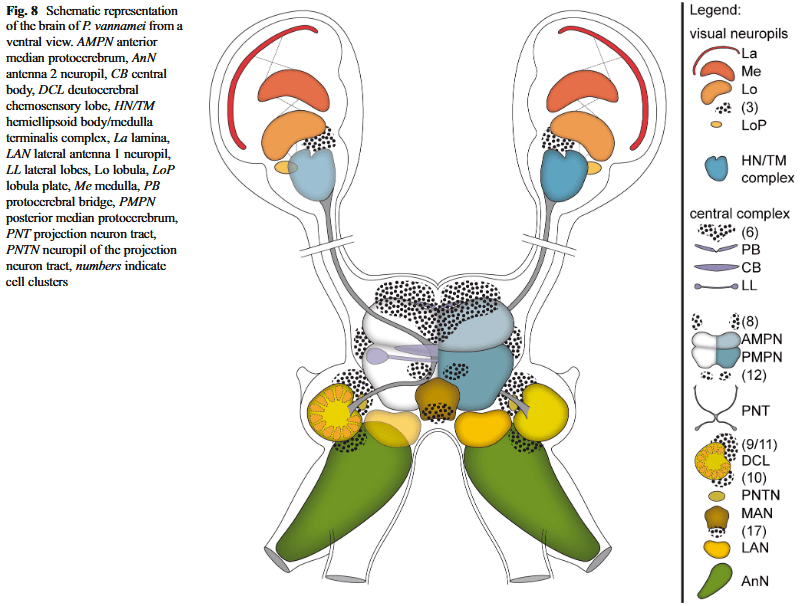
Here’s a schematic representation of the brain of P. vannamei, from (Meth et al., 2017):
We already have a pretty good idea of how their brain and global nervous system is structured, and it is clear that they are quite similar to all other decapods (Machon et al., 2020). The reason this criterion shows low confidence is because we still lack specific studies on how each brain part interacts with each other, and how sensory signals may be integrated centrally. However, as we have sufficient evidence for (J. Birch et al., 2021) to consider very high confidence for this criterion in all other decapods, it is very likely that shrimps also possess such integrative brain regions.
What we would need
Considering that the presence of integrative brain regions can be considered ubiquitous in the animal kingdom, and that every other decapod taxa showed very high evidence for this criterion, it may be reasonable to expect that shrimps validate this criterion as well until proven otherwise. Furthermore, as the study of this criterion is highly costly, it is probably best to first focus on indirect behavioral cues, and then eventually study the specific pathways that sensory information is integrated into, and into which brain regions, only if we have high incentives to do so. Thus, this criterion can be considered low priority, as convincing behavioral cues (criteria 5-8) should first be observed to give us enough incentives to look for specific brain connections.
Criterion 3: Integrated nociceptors
Definition
The animal possesses neural pathways connecting the nociceptors to the integrative brain regions.
Summary
- Current confidence: Low
- Reason: Lack of data for every decapod
- Priority: Low priority
Description
As its title describes it, this criterion looks for connections from nociceptors (criterion 1) to the integrative brain regions (criterion 2). The goal of this criterion is to ensure that nociceptive inputs could at least be potentially integrated with other sensory inputs, which would reveal its importance.
How to study
To get direct evidence of connections between neurons and specific brain regions, we only have one way to go: tracing the exact paths of nociceptors into the CNS. The goal here would be to precisely trace the path that nociceptive signals take into the nervous system, with similar techniques than for criterion 2: nerve cells tracing, imaging, electrophysiology, etc.
However, direct causal evidence by tracing nerve paths is expensive, time-consuming, implies euthanising animals, and may not be necessary. Similarly than for criterion 2, most evidence showing connections between nociceptors and integrative brain regions is usually correlative: we can observe correlated activation of integrative brain regions with noxious stimuli. While the exact nerve path may not be known, correlative evidence is sufficient to indicate that the nociceptive signal somehow reaches integrative brain regions, similarly to any other sensory pathway. A combination of various indirect evidence would also easily produce high confidence in this criterion.
Protocols summary
- Neuroanatomical and imaging techniques: Nerve tracing, Calcium imaging, Connectome, MRI, Microscopy, etc.
- Functional analysis: activate or inhibit nociceptors and look for activity in integrative brain regions or behavior directly using Pharmaco-/Opto-genetics, Electrophysiology, etc.
What we currently know
Unfortunately, we don’t have any specific evidence for this criterion in any decapods. The confidence for this criterion is low rather than very low as it benefits from indirect evidence from criterion 2, but we don’t have much more evidence to look into.
(J. Birch et al., 2021) insists: “To be clear, the problem is the absence of high quality evidence one way or the other—not that there is evidence against the nociceptors connecting to the integrative brain regions. It remains plausible that they do so”.
Similarly to the fact that the presence of nociceptors is the norm rather than the exception, the fact that nociceptors are integrated in brain regions with other sensory inputs is also the norm rather than the exception. Animals either do not have a nervous system at all (sponges), a loose nerve net that shows polymodal sensory integration (cnidarians, see criterion 2), or at least one form of nerve centralization (ganglion/brain) that clearly exhibit sensory integration, including nociceptive integration. Until proven otherwise, it would be reasonable to expect nociceptive inputs to be connected to integrative brain regions, at least partially or indirectly. Add this to the fact that all other decapods exhibit integrative brain regions and complex behavioral responses to noxious stimuli, it would be unreasonable to expect nociceptors to be completely isolated from other sensory inputs.
What we would need
Indirect evidence from other criteria also suggest that noxious stimuli induce aversive behaviors beyond simple reflexes, so while research is needed to confirm how nociceptive inputs are integrated, it may be considered low priority.
Similarly to criterion 2, this criterion can be studied in many different ways, and would benefit from evidence from various techniques. However, the most interesting and direct way to show that nociceptors connect to integrative brain regions would be to trace them.
For example, (Faulkes, 2015) studied the mechanisms of the escape response (tail flips) and was able to trace motor neurons in L. setiferus using a “backfilling” method, which consists of applying a labeling molecule (e.g. neurobiotin, or nickel and cobalt in this case) to a nerve of interest and then observe visually how it diffuses towards cell bodies in various body parts. Similar techniques can be used to fill nociceptors and observe where their cell bodies are located and what brain regions their axons connect to.
Criterion 4: Behavior modulation
Definition
The animal’s behavioural response to a noxious stimulus is modulated by chemical compounds affecting the nervous system in either or both of the following ways:
a. The animal possesses an endogenous neurotransmitter system that modulates (in a way consistent with the experience of pain, distress or harm) its responses to threatened or actual noxious stimuli.
b. Putative local anaesthetics, analgesics (such as opioids), anxiolytics or anti-depressants modify an animal's responses to threatened or actual noxious stimuli in a way consistent with the hypothesis that these compounds attenuate the experience of pain, distress or harm.
Summary
- Current confidence: Medium
- Reason: Mixed evidential picture for shrimps but high evidence for true crabs and Astacidae lobsters/crayfish
- Priority: Medium priority
Description
What this criterion mainly looks for is the ability of the nociceptive system and integrative brain regions to be modulated by either endogenous molecules and circuits (part a) or by exogenous molecules (part b), which in turn modulates the behavioral response of the animal. The presence of these modulatory processes are especially relevant as they show that the sensory and integrative systems (criteria 1-3) are directly associated with a potential subjective experience of pain that can be transcribed from behavioural outputs, and that modulating this potential subjective experience of pain is actually valuable for the animal.
- Part a
While sensory organs get information from the periphery and transfer information through ascending pathways towards the central nervous system (CNS) (or equivalent), most animals also possess descending pathways that allow the CNS to modulate how ascending sensory information is processed. The descending pain control pathway is an example of a particularly well studied descending pathway in vertebrates. For example, as described by (Gibbons, Sarlak, et al., 2022), “if an animal is injured during a fight, the dampening of their nociceptive processing may increase the animal’s fighting performance by ensuring they do not waste time or energy on responding to the injury. Likewise, when the animal has returned to safety, the descending controls can facilitate nociceptive processing, encouraging the animal to protect the injured location so that its healing is promoted”. Endogenous opioids are often involved in this process, allowing the inhibition of ascending nociceptive pathways directly in the periphery, but other neurotransmitters can also be involved, such as somatostatin, or more broadly GABA. While evidence for descending pathways is much more scarce in invertebrates, we still have insights into similar mechanisms, for example in insects (Gibbons, Sarlak, et al., 2022).
- Part b
While details on endogenous descending pathways in invertebrates are scarce, we have much more evidence regarding the effects of local applications of analgesics on noxious behavioral responses. The goal here is to observe the modulation of nociception using exogenous compounds rather than endogenous pathways. While the use of molecules that the animal does not synthesize itself may not reflect real modulatory processes, it still gives us insights into the mechanisms involved, the receptors that might be concerned, and the possibility that unknown (or known) similar endogenous molecules engage in those modulatory processes. This is especially relevant when paired with the first part of the criterion: if nociceptive inputs can be modulated endogenously, and if we can modulate the same noxious behaviors in the same way with exogenous molecules, it thus means that endogenous compounds concerned are probably similar or at least act similarly.
How to study
This criterion (part a. specifically) would imply surgical interventions and/or euthanizing of animals to study the content in neurotransmitters of specific nervous regions (e.g. immunohistochemistry, HPLC, mass spectrometry, etc). However, one way to study the presence of modulatory descending pathways non-invasively is through Stress-Induced Analgesia paradigms. As explained before, stressful experiences can activate descending pathways to inhibit nociceptive inputs; we can thus induce stressful stimuli (confinement, predator cues…) and perform nociceptive assays a few seconds/minutes later. Usually, nociceptive reactions are attenuated after stressful expositions. As per usual, such a behavioral cue is only correlational, but it gives sufficient insights into the physiological modulation of sensory inputs, especially when paired with the second part of the criterion.
The second part or the criterion involves injecting or applying analgesics locally (e.g. to a paw) before exposing the animal to noxious stimuli (preferably specific nociceptive tests such as Von Frey, Hot Plate, or Tail Flick tests, which are very common nociceptive tests for rodents); or other behavioral paradigms studying the affective–motivational aspects of pain (which will also be discussed more for other criteria), such as CPP paradigms.
Protocols summary
- Behavioral paradigms: Stress-induced analgesia, nociceptive tests
- Bioanalysis: IHC, HPLC, MS, etc
What we currently know
(J. Birch et al., 2021) states that “it is a clear point of scientific consensus that decapod crustaceans have endogenous neurotransmitter systems, including endogenous opioid, serotonergic, dopaminergic and octopaminergic systems”. Beyond possessing such endogenous systems, we are also interested in how they modulate responses to noxious stimuli, and various studies on true crabs clearly show that opioids and anti-depressants are able to modulate how crabs react to electric shocks, predators presence, or aversive environments, coherently with evidence from other taxa.
Studies on shrimp, however, show a more nuanced picture. In a study on whiteleg shrimp (P. vannamei), (Taylor, 2004) showed that if shrimps were applied the anaesthetic lidocaine before eyestalk ablation, they later showed a much less erratic swimming behavior. Similarly, (Diarte-Plata et al., 2012) showed the same effect on a species of the “true” shrimp Macrobrachium americanum. In (Barr et al., 2008), Palaemon elegans prawns’ antennas were exposed to irritant chemicals (HCl and NaOH), which induced grooming and rubbing behaviors towards the antenna (as it will be discussed in criterion 6), and were exposed to benzocaine, a local anaesthetic, which surprisingly also triggered grooming and tail flippings. However, if an application of HCl or NaOH was performed after the benzocaine, then the aversive behaviors were reduced compared to a single application of HCl or NaOH, which the authors interpreted as a suggestion that “benzocaine was effective in preventing the perception of the noxious treatment”. On the other hand, (J. Birch et al., 2021) argued that “this has to be interpreted in light of the aversive nature of the benzocaine. It may be due to an anaesthetic effect but may also be due to depleted energy levels following an aversive reaction to the benzocaine”. In (Puri & Faulkes, 2010), which aimed to replicate the results of (Barr et al., 2008), they showed that other shrimp species did not show any sign of grooming or rubbing behavior in response to acidic solutions applied to their antennas, nor did they show any nociceptive neuronal activity; which suggests that nociceptive receptors for extreme pH may be absent in these species.
In his review however, (Elwood, 2025) argues that (Puri & Faulkes, 2010) did not properly replicate the methodology of (Barr et al., 2008), and that their results should be interpreted with caution, although their negative results “are not easily explained”:
The negative result of Puri and Faulkes [17] appears to be an outlier, and are not easily explained. Puri and Faulkes [17] used three species, and found similar results for all of them. However, the sample sizes, experimental design, and analyses appear to differ to those of Barr et al. (2008) [16]. Barr et al. [16] used a two-stage design with application of water or benzocaine to one antenna in the first stage and then water, NaOH, acetic acid, or pinching to the same antenna in the second. The sample size was 144, or 288 when activities directed at each of the paired antennae were examined. Analyses used statistically powerful three-way ANOVAs with repeated measures (treated and untreated antennae), which allows for the interactions between factors to be explored. By contrast, Puri and Faulkes [17] did not use a two-stage design, and write: “Half the individuals swabbed with a control (water or sea water for NaCl and HCl; ethanol for benzocaine) on the distal half of one second antenna, and half were swabbed the stimulus. Thus, each individual had an antenna that was not swabbed, so that any effects of the mechanical action of swabbing alone could be detected”. The sample size was 17 to 24 for each analysis. That is, even the largest sample was less than 10% of that used by Barr et al. [16]. Independent t tests were used, although treated and untreated antennae were compared, which would have required paired t tests or two-way ANOVA with one repeated factor. Note that the terms “paired” and “repeated measures” have been taken by some to be different, but are the same in statistical tests [24]. The lack of a suitable protocol means the Puri and Faulkes paper could not test for the anaesthetic effects of benzocaine. The low statistical power from small sample sizes and use of inappropriate statistical tests makes it unlikely that behavioural effects of treatments could be found. Further, the use of hydrochloric acid rather than acetic acid might be a problem, because the former is much less effective than acetic acid in eliciting responses in earthworms [25]. That is, the properties of these two acids are very different. The conclusion is that the study of Puri and Faulkes (2010) [17] did not replicate that of Barr et al. (2008) [16]. |
What we would need
Considering that we currently have both conclusive and inconclusive studies regarding shrimps (although the inconclusive study arguably gives less valuable insights), replication studies and new protocols would be valuable for this criterion.
For example, as the one inconclusive study focused on low pH exposition, we could instead study the potential nociceptive effect of other stimuli on the antenna, such as how (Puri & Faulkes, 2015) explored the nociceptive effect of heat, cold or irritant chemicals (AITC, capsaicin…) on the antenna of crayfish, as mentioned in criterion 1.
To better answer criterion 4a non-invasively, I mentioned stress-induced analgesia protocols, but I could not find any on decapods. In (Gibbons, Crump, et al., 2022), two studies are cited that exposed flies to a chronic stress protocol (starvation, heat and cold stress, sleep deprivation) and showed that an anti-depressant was able to reduce the depressive-like behaviors normally induced by this protocol (Araujo et al., 2021). A similar method could be used with shrimps, ideally with the use of nociceptive tests as well (responses to heat, to capsaicin, etc).
For criterion 4b, in a more simple way, nociceptive tests could be developed for shrimps, similarly to the Tail Flick or Hot Plate tests on mice, for example (Deuis et al., 2017). These tests can then serve as a baseline to study the effect of many molecules applied or injected to shrimps. We only have one study, mentioned before (Taylor, 2004), that showed the effect of lidocaine on swimming behaviors: it would be interesting to expose shrimps to other painkillers (e.g. morphine, NSAIDs) and to use other nociceptive stimuli than eyestalk ablation to observe behavioral modulations: the difficulty is yet to suggest a relevant paradigm that is highly replicable and comparable to other species. As a personal example, that’s exactly the kind of project I worked on during my PhD on the planarian model: developing relevant and replicable nociceptive tests for new models (Reho et al., 2024).
Criterion 5: Motivational trade-offs
Definition
The animal shows motivational trade-offs, in which the disvalue of a noxious or threatening stimulus is weighed (traded-off) against the value of an opportunity for reward, leading to flexible decision-making.
Enough flexibility must be shown to indicate centralized, integrative processing of information involving an evaluative common currency.
Summary
- Current confidence: Low
- Reason: No data for shrimps and medium evidence for anomuran crabs
- Priority: High priority
Description
As (J. Birch et al., 2021) states it, “what we are looking for here is robust evidence that an animal is motivated to avoid a noxious stimulus, and that this motivation is weighed (traded off) against other motivations in a flexible decision-making system”.
“Enough flexibility must be shown to indicate centralized, integrative processing of information involving an evaluative common currency” means that simple physiological processes such as sensitisation would not be enough to explain decision-making. For example, when squids are injured, they tend to escape predatory cues earlier and for longer. While this may indicate that squids are aware of their injury, and thus that they are more cautious in the presence of predators, it may also be a simpler case of sensitized tactile and visual receptors that are somehow connected, as discussed in (J. Birch et al., 2021). Confounding factors such as this one should be especially looked for in the case of this criterion, as for the next three behavioral criteria.
How to study
This criterion is behavioral-based and does not necessarily involve invasive methods. However, exposing animals to noxious stimuli or threats should still imply ethical considerations, and individuals should always be able to escape the noxious stimuli.
As another example, if you display to an individual a reward (food, secure environments…) while simultaneously exposing them to noxious stimuli (predator cues, electric shocks, heat…), they may balance (trade-off) the value of the reward with the intensity of the threat and choose to stay or leave. By varying the amount/quality of the rewards and the amount/threat of the noxious stimuli, we can measure how much the individual weights each stimuli. The difficulty here is that the animals must have already learned what the threatening stimulus is so they can expect the value of the threat to choose if the reward is worth it. Hence, conditioning paradigms would usually be needed here (as we will discuss later). Exceptions may be predator cues as a threat and food as a reward, which can both be innate mechanisms.
Other ways to study motivational trade-offs would be through motivation itself by using Maximum Price Paid (MPP) paradigms. For example, in (Hovland et al., 2006), foxes learned to pull a chain to open a door leading to food, the “cost” being adjusted by increasing the number of chain pulls the fox needed to perform to open the door; or in (Jensen et al., 2024), where cows had to push through a gate to reach their calves, the cost being adjusted by increasing the force needed for the cow to go through the gate. These protocols are also part of operant conditioning, which will be further discussed in criterion 7.
Such paradigms could thus be done using many scenarios, but the core idea should stay the same: rewards and threats of varying intensity.
Protocols summary
- Conditioning paradigms
- Maximum Price Paid (MPP) paradigms
What we currently know
Most of the evidence we have for this criterion in decapods concerns a series of studies performed by Elwood’s lab on hermit crabs. In these studies, they showed that hermit crabs (who live in shells produced by other animals) were less likely to leave a “high quality” shell rather than a “low quality” shell when subjected to electric shocks. They were also more likely to stay in their shell when they could detect a nearby odour (whether from a predator or from a food source, curiously). (J. Birch et al., 2021) concludes, “the results show that decisions regarding shell evacuation are modulated by odour. They do not convincingly demonstrate a weighing of the relative value of shock avoidance against predator avoidance for two main reasons: the observation of an effect when the odour was not that of a predator, and the failure to find any trade-off between the voltage of the shock and the concentration of the odour. In sum, we can have only medium confidence that hermit crabs satisfy criterion 5 on the basis of these experiments”.
We don’t have any data regarding shrimps or any other decapod.
What we would need
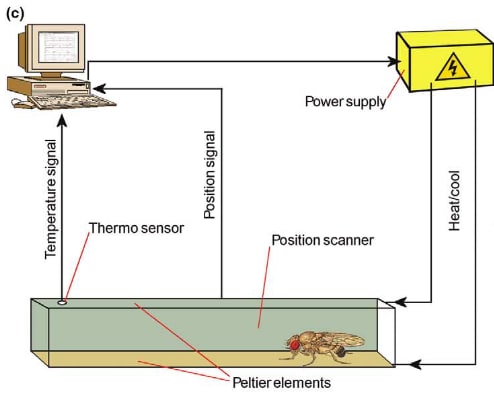
Operant conditioning paradigms in invertebrates are not common. In a review, (Brembs, 2003) showed how it can be done in some species. Here’s an example using flies:
Operant conditioning in Drosophila, from (Brembs, 2003). Cartoon of the heat-box learning paradigm. The flies walk around in small closed rectangular chambers in complete darkness. The upper and lower surfaces consist of two Peltier-elements for rapid heating and cooling. A position scanner (bar code reader) reports the position of the fly to the computer while a control circuit and a thermosensor keep the chamber at a defined temperature. Whenever the fly enters the pre-defined punished side, the whole chamber is heated to 40°C, and as soon as the fly leaves that side again the chamber is cooled down to 20°C. Within minutes flies learn to avoid the punished side during training, and maintain this side preference in the following learning test when heat is permanently switched off. |
This kind of study is more relevant to criterion 7 than criterion 5, but they could be adapted to also assess motivation towards a reward. In this example, if the fly is exposed to rapid temperature changes when they enter the left side of the chamber and we add food to the left side, then we could assess up to what temperature the fly is willing to bear to reach food.
In another example on planarians (flatworms), which is more closely related to place preference tests than operant conditioning, individuals were exposed simultaneously to a gradient of two stimuli of opposite valence (Inoue et al., 2015). Let’s look at the sub-figure D in this example: planarians were able to freely move on a gradient of light (for which they prefer darkness) and a gradient of temperature (for which they prefer cooler temperature), hence they could choose between a dark but hot side or a cool but bright side. In this case, most individuals preferred the dark but hot side, suggesting that they value the brightness more than the temperature. By doing these place preference comparisons across many other stimuli, this study was able to assess the order of predominance of each stimuli, which could also be seen as a form of motivational trade-off (the authors even state this is decision making), as nowhere on these gradients planarians would be able to reach an ideal position. This kind of protocol could also be highly insightful with shrimps.
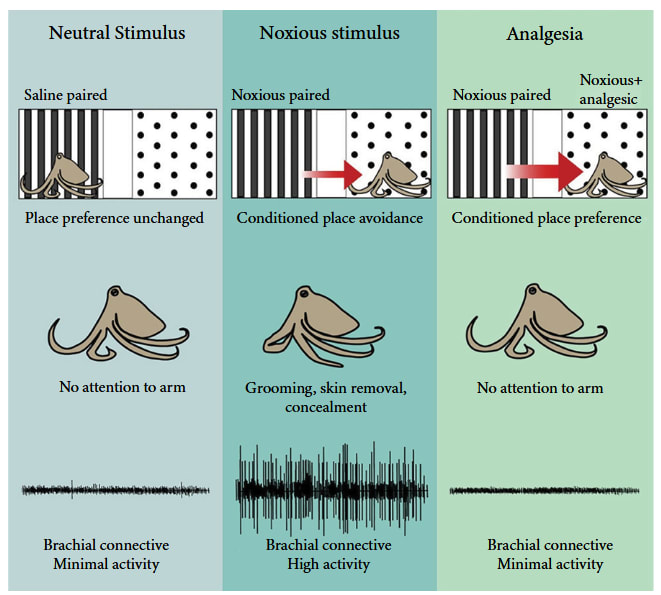
Criterion 6: Flexible self-protection
Definition
The animal shows flexible self-protective behaviour (e.g. wound-tending, guarding, grooming, rubbing) of a type likely to involve representing the bodily location of a noxious stimulus.
Summary
- Current confidence: Low
- Reason: One inconclusive study for shrimps but high confidence for crabs and medium evidence for true shrimps
- Priority: High priority
Description
(J. Birch et al., 2021) states: “What we are looking for here is robust evidence of self-protective behaviours that go beyond reflexes: to meet this criterion, the animal should be able to vary its response in a targeted way, according to where on the body the noxious stimulus is administered.”
As we humans do, animals tend to groom or rub the site of an injury. This behavior can have many benefits like cleaning the wound and preventing infections, but one reason is often pain relief. In close proximity of the site of injury, where nociceptors are usually sensitized (highly reactive), rubbing activates close non-nociceptive mechanoreceptors which, based on the gate control theory of pain, are able to locally inhibit nociceptive inputs in favor of mechanical inputs (Moayedi & Davis, 2013). In other words, as the nervous system can only process sensory inputs at a certain spatial resolution, if you activate mechanoreceptors around an injury, the mechanosensory inputs will override the nociceptive inputs from the injury, and thus the brain will receive less “pain” signals and more “touch” signals. Note that the gate control theory of pain is rather specific towards vertebrates, and I’m not aware of any similar mechanisms studied in invertebrates yet, although they may exist.
More generally, such self-protective behavior specifically oriented towards injured bodily locations are good indicators that animals are aware of where painful signals come from, and that they are motivated to act towards them, protecting themselves from further harm (Gibbons, Crump, et al., 2022).
How to study
This criterion can be observed rather easily, although strict protocols and strong controls are needed to account for the specificity of each species. Self-protective behaviors can be induced by exposing specific body parts, often limbs, to noxious (usually long-lasting) stimuli. For example, irritants such as formalin or capsaicin (targeting TRP receptors) can be injected or applied to the face, inducing face-touching or guarding, or to a limb, inducing abnormal walking (the animal will use this limb less), guarding, licking, rubbing, etc. In animals without limbs, or for those whose limbs can not reach all body parts, guarding could also be interpreted from contractions, contortions, rolling, rubbing or any behavior that will expose the injured body part significantly less to the environment or to a threat.
Protocols summary
- Behavioral cues (location-specific touching, rubbing, licking, etc) induced by noxious stimuli (irritants injections or applications, limb cut, etc).
What we currently know
Various studies on crabs clearly showed injury-oriented rubbing, scratching or guarding, either towards the mouth, the abdomen, or limbs (J. Birch et al., 2021).
For true shrimps, a few studies clearly showed injury-oriented rubbing. For example, in (Diarte-Plata et al., 2012), true shrimps that were subjected to eyestalk ablations (a common practice in commercial settings) rubbed the site of the wound, while subsequent covering of the wound (to prevent bleeding) drastically reduced the rubbing behaviors. In (Barr et al., 2008), an acidic solution applied to the antenna of another true shrimp species induced grooming and rubbing behaviours directed towards the affected antenna.
However, (Puri & Faulkes, 2010) tried to replicate these results with L. setiferus, a shrimp species closely linked to P. vannamei, with inconclusive results: the same acidic solutions did not induce any specific grooming behavior, and recordings of electrical nerve activity in crayfish exposed to this same protocol did not even show any nociceptive excitation (as discussed in criterion 4). Thus, this study suggests that these species may not be equipped to detect extremely acidic solutions and react to them, but we still don’t have relevant data for any other stimulus (thermal, mechanical, chemical agonists, etc).
What we would need
Considering that we currently have high evidence for other decapods but only one inconclusive study on shrimps, it would be highly valuable to replicate this protocol and further study this criterion, especially considering the critiques of (Elwood, 2025) on this study that we discussed in criterion 4. Considering that this criterion can be studied quite easily and the weight it has on the global consideration of sentience in the framework, it can definitely be argued as high priority.
For example, it is still not clear whether P. vannamei also consistently show oriented guarding and/or grooming after eyestalk ablation similarly to true shrimps or not at all, although it could easily be observed and reported as its still a very common practice. It would also be interesting to replicate simple exposition of irritant chemicals (or other noxious stimuli) to various body parts of shrimps to observe grooming or protective behaviors, such as how it has been done on crabs (Elwood et al., 2017).
Criterion 7: Associative learning
Definition
The animal shows associative learning in which noxious stimuli become associated with neutral stimuli, and/or in which novel ways of avoiding noxious stimuli are learned through reinforcement. Note: habituation and sensitisation are not sufficient to meet this criterion.
Summary
- Current confidence: Low
- Reason: Lack of data for shrimps but high evidence for true crabs and medium evidence for lobsters
- Priority: High priority
Description
Organisms have many ways to learn things, and even very simple organisms can learn through non-associative processes like habituation (reduction in the response to a stimulus that is delivered repeatedly) or sensitization (enhancement or augmentation of a response produced by the presentation of a strong stimulus). These non-associative learning processes are quite well understood through chemical mechanisms and neuronal circuits models (Byrne, 2013). Associative learning, on the other hand, is a broad category of learning processes that allow an animal to create relationships between stimuli and/or responses (Byrne, 2013). While associative learning implies much more complex encoding processes, their role as a proxy to sentience is still debated. Here’s how (J. Birch et al., 2021) contextualizes this issue:
Simple forms of associative learning appear to occur unconsciously in humans (Greenwald and De Houwer, 2017), and this has led to ongoing debate and inquiry as to which kinds of associative learning are linked most strongly to sentience and why (Birch et al., 2020). Instrumental learning (Skora et al., 2021), reversal learning (Travers et al., 2017), learning "incongruent” spatial relationships (Ben-Haim et al., 2021), and learning across temporal gaps between stimuli (“trace conditioning”; Clark et al., 2002) are more complex and more strongly linked to sentience than classical conditioning involving two stimuli presented at the same time. However, given the ongoing debate on this issue, we will regard all evidence of associative learning as relevant to the overall evidential picture. We stress, however, that it is only one part of that picture. |
What we are looking for in this criterion is “robust evidence that the animal is able to form associations between noxious stimuli and neutral stimuli by, for example, learning to associate a particular place, or an otherwise neutral odour, with a noxious stimulus. We are also looking for evidence that an animal can learn a novel behaviour (distinct from any preexisting reflex responses) that allows it to avoid a noxious stimulus”.
Thus, this criterion covers a quite large range of learning capabilities, and studies relevant for this criterion should be carefully interpreted. Also, as discussed in the methodology, the broad capability for associative learning can be closely linked to other criteria.
How to study
Associative learning is usually explained through the classical conditioning pavlov’s dog experiment.
Figure 47.1 from (Byrne, 2013): Classical conditioning. In the procedure introduced by Pavlov, the production of saliva is monitored continuously. Presentation of meat powder (unconditioned stimulus, US) reliably leads to salivation, whereas some “neutral” stimulus such as a bell (conditioned stimulus, CS) initially does not. With repeated pairings of the bell and meat powder, the animal learns that the bell predicts the food and salivates in response to the bell alone. |
This experiment shows that dogs are able to associate a neutral stimulus (a bell ringing, for which they don’t care) with a valenced stimulus (food, which is positively valenced) when they are exposed together enough times. In other words, dogs are able to passively associate stimuli that they normally should not care about with other stimuli that they care about if they are sufficiently correlated.
This can also be done using negatively valenced (noxious) stimuli. For example, fear conditioning is a common practice used on rodents to study learning, memory, and disorders like PTSDs. In this case, a sound or light is usually exposed to the rodent seconds before an electric shock is applied (that they are not able to escape). At first, the rodent panics or freezes after the electric shock, but after a few trials, the animal panics or freezes just after the flashing light, even if the electric shock is no longer applied, because they associated the flashing light with an incoming electric shock, just like the dog associated the bell ringing with an incoming food reward.
As mentioned, habituation and sensitization are basic learning processes that can arise without any form of sentience, so they have to be controlled for. To do so, we can usually expose the animal to each stimulus alone to check whether they elicit a different reaction (e.g. if food makes the dog salivate more or less over time, or if he gets really annoyed or aroused by the bell), or expose the dog to both stimuli but randomly, so that the bell ringing does not match food coming right after, and the dog has no reason to associate both together. If he still does, confounding factors should be looked for.
Beyond classical conditioning, associative learning can also be studied through operant (also called instrumental) conditioning; which is what “novel ways of avoiding noxious stimuli are learned through reinforcement” in the criterion’s description involved. Operant conditioning is even more closely linked to sentience, as conscious awareness seems to be required for this type of learning processes in humans (Skora et al., 2021).
Operant conditioning differs from classical conditioning in both mechanism and outcome. While classical conditioning pairs stimuli to produce involuntary, reflexive behaviors (like salivating at food), operant conditioning shapes voluntary behaviors through their consequences. Actions followed by rewards tend to be repeated, while those followed by negative outcomes diminish. (“Operant Conditioning,” 2026) |
Operant conditioning paradigms can also take many shapes and forms. For example, if we take the fear conditioning protocol from before but allow the rodent to escape the box when the light turns on, they will do so. At first, they will escape only after the electric shock, but once they associate the light with an incoming electric shock, they will escape after the light turns on, as their behavior (active escape) is reinforced by the lack of the noxious response they learned before. Another example is learned helplessness: if we expose the rodent to inescapable and unpredictable electric shocks, and only then allow them to escape, most will not escape as they learned that the shock is inevitable after the flashing light. This kind of paradigm is used to study depressive behaviors (Landgraf et al., 2015).
Protocols summary
- Conditioning paradigms
What we currently know
(J. Birch et al., 2021) argue that true crabs display high evidence of associative learning thanks notably to (Fernandez-Duque et al., 1992) and (Dimant & Maldonado, 1992). In these studies, crabs were conditioned to receive either electric shocks (negative reinforcer) or food (positive reinforcer) whenever they entered an illuminated compartment, while nothing happened if they entered another dark compartment. When given the ability to freely move towards the two compartments the next day, crabs took significantly longer to approach and enter the illuminated compartment than non-conditioned crabs if they received electric shocks, and entered the compartment significantly faster if they received food, suggesting that they indeed learned to associate the illuminated compartment with an aversive or positive stimulus. Other studies, such as from the Maldonado or the Elwood labs, either lacked sufficient controls or showed null results due to an increasing complexity of the learning tasks, which show ”the great challenges involved in rigorously demonstrating associative learning in any animal but also ingenious ways of overcoming those challenges”.
In other decapods, lobsters more specifically, a few similar studies were performed which showed encouraging signs of associative learning, but they often lacked proper controls as well. Thus, (J. Birch et al., 2021) states that “associative learning in other decapods should be regarded as a plausible, largely unexplored possibility”.
Unfortunately, we currently lack data on any species of shrimps for this criterion.
What we would need
Considering that we already have medium to high confidence for other taxa and that associative learning is a very common feature present in animals, we might argue for a low priority on this criterion. However, as we currently have no data for shrimps, and because protocols for other decapods already exist, we may argue that studying this criterion may be of at least medium priority: it could give us quick valuable insights (especially if we find out that no associative learning can be performed), even if it would not weight the most in the global framework. This criterion is, on the other hand, a quite important prerequisite for criterion 5 and 8, which would both weigh way more in the general framework. For these reasons, I argue that criterion 7 should still be highly prioritized, as it will be used as groundwork for other important criteria.
Similar conditioning protocols than for crabs could probably be easily transposed for shrimps. For example, here is the simple apparatus used in (Dimant & Maldonado, 1992):
Shrimps tend to swim way faster than crabs walk, so we may have to overcome some practical issues to adapt these types of protocols, but since the idea is so basic (contextual cues and brief stimuli), studying associative learning in shrimps is not out of reach at all.
Criterion 8: Pain-relief seeking
Definition
The animal shows that it values a putative analgesic or anaesthetic when injured in one or more of the following ways:
a. The animal learns to self-administer putative analgesics or anaesthetics when injured.
b. The animal learns to prefer, when injured, a location at which analgesics or anaesthetics can be accessed.
c. The animal prioritises obtaining these compounds over other needs (such as food) when injured.
Summary
- Current confidence: Very Low
- Reason: No data for any decapod
- Priority: Medium priority
Description
This criterion is also rather explicit. Part a. checks that the animal is able to associate an action that delivers relief to an injury (they are thus most likely aware of the painful injury and willing to act upon it, similarly to criterion 6); Part b. checks that the animal is able to associate a context where they are able to get this relief and are actively seeking to get to this context (pain relief anticipation); Part c. checks that pain relief can be actively searched for even before other vital needs, thus suggesting that pain signals are as important (if not more) and as integrated as any other needs.
How to study
Part a. is addressed using self-administration paradigms
As presented in (Gibbons, Crump, et al., 2022), “to self-administer analgesics, an injured animal must know: (1) the behavioural response necessary to administer the drug, and (2) the drug’s analgesic effect. Condition 1 is usually trained by associating a conditioned stimulus (e.g., colours, spatial locations) with the unconditioned stimulus (e.g., analgesic drug or control). For condition 2, the analgesic and control are presented successively, with a time gap when the drug’s effects can be experienced. This ensures that the animal only associates the analgesic effect with the drug conditioned stimulus, and not the control stimulus.”
One extremely important control we will need here is to know whether the effects of the analgesics or anaesthetics we use induce the activation of a reward system by themselves, besides relieving an existing pain. For example, opioids are very strong painkillers for most vertebrates, whether they are applied locally or centrally, but they are also highly rewarding and addictive by themselves. Thus, in a scenario where an animal is injured and has access to opioids, we have to make sure the animal is really searching for pain relief and not only the opioid itself. Other non-rewarding painkillers such as anti-inflammatory drugs could be used instead, but since they act way more slowly, it is much harder for the animal to associate an action (e.g. the push of a button that injects anti-inflammatory painkillers) and the resulting pain relief. In that case, it's possible to associate pain relief with a specific environment by locking the animal in the environment until the painkiller is effective. Other non-rewarding molecules used to relieve pain exist, such as clonidine or lidocaine, but their effect on different species need to be studied more thoroughly.
Practically, self-administration protocols can take many forms. The drug administration can be injected through a cannula/catheter directly in the animal for the quickest response, mixed in the food, diluted in drinks, inhaled, etc. The condition for the administration can be basically anything that the animal can learn to do: often the press of a button/lever, or the presence of the animal in a specific location. The drug administration is then performed by an experimenter or automatically (through the push of the button or via video tracking for example).
Then, once the animal learned how to get the drug and what effect it had, we can observe how healthy vs. injured animals act. For example, injured mice who can self-inject painkillers will consistently do so more often than non-injured mice and also more than injured mice that receive a non-painkiller solution instead, indicating that mice seek pain relief specifically and not the painkiller (Cabañero et al., 2022).
Part b. is addressed using Conditioned Place Preference (CPP) paradigms
CPP paradigms consist of maintaining an injured animal in a specific context while administering a painkiller (e.g. a dots-painted box) or in another context without any painkiller (e.g. a stripes-painted box). Later, when the animal can choose to go in either two boxes, they should prefer to go in the dots-painted box (even if not injured), as they previously associated this context to positive welfare (pain relief).
As a control, if a molecule (e.g. opioids) is administered to the animal in the stripes-painted box while they are not injured and they still prefer the stripes-painted box over the dot-painted one, it means that the animal is seeking the molecule, not pain relief. If this control is negative (the animal chooses stripes-painted and dots-painted boxes randomly), then we can make sure the molecule does not act by itself and is indeed inducing pain relief when associated with the injury. As another similar example, in the mice study cited before, when the painkiller was administered using ethanol as a vehicle (the carrier solution), mice showed no preference between the presence of the painkiller or the control vehicle solution (without the painkiller): they both self-administered similar quantities because they were seeking ethanol at least as much than the painkiller (Cabañero et al., 2022). They then used another neutral carrier to avoid this issue.
While CPP is done using positive reinforcement (pain relief, food or other drugs), it can also be done using negative reinforcement (noxious stimuli). It may be called Conditioned Place Aversion or Avoidance (CPA). An example is illustrated in (J. Birch et al., 2021):
Figure 3 from (J. Birch et al., 2021): A key figure from (Crook, 2021). The experiment (which is relevant to our criteria 4, 5 and 8) involved four groups of animals (with either 7 or 8 in each group): a group injected with only saline solution; a second group injected with acetic acid; a third group injected with acetic acid and, later, lidocaine; and a fourth group (not shown) injected with saline and then lidocaine. After receiving acetic acid, the affected animals showed directed self-protective behaviour, increased neural activity, and avoidance of the chamber where they had received it. Lidocaine silenced the heightened neural activity, stopped the self-protective behaviour, and led to a conditioned preference for the chamber where the effects of the lidocaine were experienced. The figure is © Robyn Crook 2021, CC-BY-NC-ND 4.0 licensed. See the original source for further methodological details. |
Part c. is addressed by combining CPP paradigms with additional variables
To observe prioritisation of pain relief rather than other vital needs, we can expose the animals to additional variables, for example by exposing them to hydric restriction and allowing them to drink in the stripes-painted box. The animal could then choose between pain relief in the dots-painted box or hydration in the stripes-painted box.
Studies relevant for criterion 8 in invertebrates are rare, but studies relevant for criterion 8c are rare even in vertebrates. Here again, we have known for a long time that most animals would choose addictive drugs (such as strong opioids like morphine or fentanyl) over food (Stewart et al., 1984; Townsend et al., 2021), but no study focuses on food vs pain relief specifically by using non-addictive painkillers in invertebrates. This is a blatant and obvious research gap, as (J. Birch et al., 2021) puts it.
One good example in vertebrates is (Martin et al., 2006), in which rats with induced chronic pain were able to press a lever to get food and another lever to self-administer clonidine, a non-addictive painkiller. Compared to healthy rats, injured rats pressed the clonidine lever significantly more and the food lever significantly less, showing that they prioritized pain relief over food over multiple days.
Protocols summary
- Self-administration paradigms
- CPP/CPA paradigms
What we currently know
There are no studies relevant for this criterion for any decapod.
What we would need
This criterion has a really high value in this sentience framework. For example, while (Walters, 2022) is rather critical of this sentience framework as mentioned in the methodology, he still argues that “CPA, CPP, and motivational conflict tests with and without prior nociceptive sensitization are powerful, well-described indicators of nociceptive aversive states”, which would pave the way for robust evidence of affective components of pain in these species, and that they “are feasible for many species, including decapod crustaceans”.
However, this criterion is also very demanding, as both protocols and results should be carefully discussed and replicated to avoid any bias, confounding factor or weird interpretations. Also, as no study has ever been done in decapods for this criterion, considerable amounts of groundwork would be necessary first (protocol development, validation, replications, etc). As mentioned, criterion 8 also relies a lot on criterion 7, as these conditioning protocols require associative learning to work. We still don’t have any associative learning protocol available for shrimps, so criterion 7 would be part of the groundwork needed to study criterion 8. Then, CPP protocols could probably be easily adapted for shrimps as well. For these reasons, I would classify this criterion as medium (leaning towards high) priority.
As presented in (J. Birch et al., 2021), (Datta et al., 2018) developed a self-administration protocol for crayfish. Their study was irrelevant for this criterion as the animals were not injured, but it may be an interesting protocol to adapt. Their method was to insert and glue a cannula to the animal’s head (similarly than for electrophysiology, except here its not electrodes) while drug administration would be automatically triggered by the position of the animal via video tracking. This a great example of operant conditioning via automatically-triggered drug rewards that we could adapt to shrimps as well. To be relevant for criterion 8, animals should also be exposed to noxious stimuli to seek pain relief, and allow them to administer non-rewarding, non-addictive painkillers.
Conclusion
In this report, I argued the (within-cause) prioritization of research for each of the eight sentience criteria for shrimps. Here is what the final prioritization table for each criteria looks like:
Criteria | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| A priori confidence | High | High | High | Medium | Low | Low | Medium | Low |
Weight | Low | Low | Low | Medium | High | Medium | High | High |
Priority | Low | Low | Low | Medium | High | High | High | Medium |
I argue that criteria 5 to 7 are of highest priority to study in shrimps, considering all variables mentioned in the methodology. Each has their own advantages and disadvantages: while criterion 6 may be quick and cheap to study but not weigh the most in the global consideration of shrimps as sentient individuals, criterion 5 and 7 would weigh more but also cost more. Criterion 8 might be the most convincing criterion of the whole sentience framework - almost convincing of sentience by itself - but it relies heavily on groundwork that we don’t yet have from other criteria, especially criterion 7. Criteria 1 to 4, on the other hand, can already be more expected to be present in shrimps even though we do not have direct evidence, and would weigh much less on the overall consideration of shrimps as sentient individuals.
What I also hope this report brings is a sense of how much studying shrimp sentience costs and how difficult it is (and isn’t!). Sentience research involves protocols and paradigms that are very commonplace and routinely used in many other fields: it can easily be studied in existing labs. It may cost more than we’d expect (science IS expensive), but it also may cost way less than we’d expect: we only need a few projects to make highly significant progress on this matter. I also discussed that a rule-of-thumb for the cost of such research would be roughly $100k per criteria (per species). While the scientific process may be unpredictable and we could expect research projects to cost a bit more than that, we can still get a glimpse on the order of magnitude it involves. Consider that studying a few more criteria in shrimps would provide significantly more advocacy power: within a year or a few years and less than $1m (as a rough example), we could substantially accelerate the protection efforts of billions (if not trillions) of shrimps every year, similarly than how sentience research in cephalopods allowed them to be better considered and protected (Schnell et al., 2026).
“Should we focus on welfare interventions instead?”
I argued in the introduction why I think advancing the science of sentience in shrimps is useful, critical and urgent. We could, however, still argue that money and efforts should be put towards shrimp welfare interventions rather than shrimp sentience research. I would agree that we have enough reasons to put more efforts towards welfare interventions rather than sentience research, but I would still argue that shrimps have a very particular place on the decapods consideration of sentience and that sentience research should be explored more than it currently is. As we have to work this question under high uncertainty, the decision is still highly based on our own risk aversion (Clatterbuck, 2023). Opposite views, of those that currently do not advocate for shrimp welfare interventions, are legitimate as well considering the current level of evidence we have. More shrimp sentience research would reduce these uncertainties, and would allow to orient money and efforts towards those who need it most - whether those are shrimps or not.
I also think that shrimp sentience research has the potential to reach people, researchers and institutions way beyond the EA ecosystem of donors, grantmakers, and animal welfare advocates.
“What about welfare ranges?”
I took the assumption, in the introduction, that the moral and legal foundation for animal welfare is sentience no matter to which degree and intensity. This assumption was made to make this report possible without having to discuss the entire field of consciousness science, but I also believe that exploring sentience and welfare research are two very similar faces of the same coin. For example, in his Welfare Range Estimates, (Fischer, 2023) argues that all invertebrates probably have welfare ranges “within two orders of magnitude of the vertebrates nonhuman animals [presented in his report]”, so more sentience research would probably carry as much weight as more welfare research in expected values maximizations (Clatterbuck, 2023). And more generally, if you want to carry on welfare research, you’ll probably end up doing the same exact types of studies relevant to study sentience. Most studies suggested in this report will also be highly valuable to value welfare, especially those of the higher criteria, which I argue should be highly prioritized.
It’s true that this report relies on a very pain-centric framework. As agreed by their authors in (Browning & Birch, 2022), it is possible that this traditional focus on pain does not serve sentience research well for some species. For example, we have interesting evidence of bumble bees playing with wooden balls “just for fun” (Galpayage Dona et al., 2022), or other optimistic and pessimistic judgement biases in bees (Browning & Birch, 2022). Then, in a sense, I agree that welfare research goes a bit beyond sentience research in the context of this framework. However, the definition of sentience does account for positively valenced states, and (J. Birch, 2024) recalls that “Sentience is not pain. Although there are pragmatic reasons for the focus on pain in debates about animal sentience, we must be open to the possibility that the class of sentient animals is bigger than the class of animals that feel pain”. Thus, both sentience and welfare research do work perfectly hand in hand, and both are needed.
“I want to support research. What should I do?”
You are a grantmaker: consider ambitious call proposals for shrimp sentience research based on criteria that have the potential to most advance our comprehension of shrimp sentience, as presented in this report.
You are a donor or an animal advocate interested in shrimps: consider requesting grantmakers for more shrimp sentience research funding. If you would like to donate specifically to this field, grantmakers need to know; and they can make it happen! Also consider expressing your interest in these questions to scientists already working with shrimps.
You want to support shrimp sentience research directly: consider expressing your interest and donating to Arthropoda Foundation.
You want to support shrimp sentience and welfare desk research: consider expressing your interest and donating to Rethink Priorities’ Animal Welfare Department.
You are convinced that shrimps are most likely sentient and that their welfare should be improved: consider donating to Shrimp Welfare Project for better farming practices, or Crustacean Compassion and Aquatic Life Institute for holding retailers and wholesalers accountable for the animals they sell. Feel free to share other charities that work towards better shrimp welfare practices.
Here are a few other grantmakers that might be interested in funding such research or welfare interventions: Animal Charity Evaluators, Animal Welfare Fund from EA Funds, Animal Welfare Fund from Founders Pledge, and Farm Animal Welfare fund from Coefficient Giving. Also feel free to comment or tag other grantmakers or funds that would be interested in shrimp sentience research.
Acknowledgements
This work was part of Sentient Futures’ Project Incubator program.
This report was entirely and carefully crafted by Guillaume Reho, with recurrent reviews and discussions with Aaron Boddy, whom I deeply thank for his time and help on this project.
Mistral AI’s “Le Chat” was punctually used as a research assistant, and only generated a negligible amount of text directly present in this report.
I also want to thank Bob Fischer and Benjamin Ransom for their prompt help and discussions; as well as the entire Sentient Futures team for their support throughout the program.
Bibliography
Araujo, S. M., Bortolotto, V. C., Poetini, M. R., Dahleh, M. M. M., Couto, S. de F., Pinheiro, F. C., Meichtry, L. B., Musachio, E. A. S., Ramborger, B. P., Roehrs, R., Guerra, G. P., & Prigol, M. (2021). γ-Oryzanol produces an antidepressant-like effect in a chronic unpredictable mild stress model of depression in Drosophila melanogaster. Stress, 24(3), 282–293. https://doi.org/10.1080/10253890.2020.1790519
Barr, S., Laming, P. R., Dick, J. T. A., & Elwood, R. W. (2008). Nociception or pain in a decapod crustacean? Animal Behaviour. https://doi.org/10.1016/j.anbehav.2007.07.004
Birch, J. (2024). The Edge of Sentience: Risk and Precaution in Humans, Other Animals, and AI (1st ed.). Oxford University PressOxford. https://doi.org/10.1093/9780191966729.001.0001
Birch, J., Burn, C., Schnell, A., Browning, H., & Crump, A. (2021). Review of the evidence of sentience in cephalopod molluscs and decapod crustaceans. London School of Economics and Political Science. https://www.lse.ac.uk/business/consulting/assets/documents/Sentience-in-Cephalopod-Molluscs-and-Decapod-Crustaceans-Final-Report-November-2021.pdf
Birch, S., & Plachetzki, D. (2023). Multisensory integration by polymodal sensory neurons dictates larval settlement in a brainless cnidarian larva. Molecular Ecology, 32(14), 3892–3907. https://doi.org/10.1111/mec.16968
Brembs, B. (2003). Operant conditioning in invertebrates. Current Opinion in Neurobiology. https://doi.org/10.1016/j.conb.2003.10.002
Browning, H. (2026). Mapping profiles of animal affect. Biology & Philosophy, 41(2), 16. https://doi.org/10.1007/s10539-026-10019-7
Browning, H., & Birch, J. (2022). Animal sentience. Philosophy Compass, 17(5), e12822. https://doi.org/10.1111/phc3.12822
Browning, H., & Veit, W. (2022). The sentience shift in animal research. The New Bioethics, 28(4), 299–314. https://doi.org/10.1080/20502877.2022.2077681
Byrne, J. H. (2013). Learning and Memory. Fundamental Neuroscience. https://doi.org/10.1016/B978-0-12-385870-2.00047-0
Cabañero, D., Álvarez-Pérez, B., Martín-García, E., & Maldonado, R. (2022). Operant Self-medication for Assessment of Spontaneous Pain Relief and Drug Abuse Liability in Mouse Models of Chronic Pain. Bio-Protocol, 12(5), e4348. https://doi.org/10.21769/BioProtoc.4348
Clatterbuck, H. (2023, October 16). Prioritizing animals of uncertain sentience. Rethink Priorities. https://rethinkpriorities.org/research-area/the-risks-and-rewards-of-prioritizing-animals-of-uncertain-sentience/
Crook, R. J. (2021). Behavioral and neurophysiological evidence suggests affective pain experience in octopus. iScience, 24(3). https://doi.org/10.1016/j.isci.2021.102229
Datta, U., van Staaden, M., & Huber, R. (2018). Crayfish Self-Administer Amphetamine in a Spatially Contingent Task. Frontiers in Physiology, 9. https://doi.org/10.3389/fphys.2018.00433
de Oliveira, G. B., Griczinski, P., Pedrazzani, A. S., Quintiliano, M. H., & Molento, C. F. M. (2024). Brazilians’ perception of shrimp sentience and welfare. Journal of Veterinary Behavior, 71, 41–56. https://doi.org/10.1016/j.jveb.2023.12.006
Deuis, J. R., Dvorakova, L. S., & Vetter, I. (2017). Methods Used to Evaluate Pain Behaviors in Rodents. Frontiers in Molecular Neuroscience, 10. https://www.frontiersin.org/articles/10.3389/fnmol.2017.00284
Diarte-Plata, G., Sainz-Hernández, J. C., Aguiñaga-Cruz, J. A., Fierro-Coronado, J. A., Polanco-Torres, A., & Puente-Palazuelos, C. (2012). Eyestalk ablation procedures to minimize pain in the freshwater prawn Macrobrachium americanum. Applied Animal Behaviour Science, 140(3), 172–178. https://doi.org/10.1016/j.applanim.2012.06.002
Dimant, B., & Maldonado, H. (1992). Habituation and associative learning during exploratory behavior of the crab Chasmagnathus. Journal of Comparative Physiology A. https://doi.org/10.1007/BF00198986
Elwood, R. W. (2025). A History of Pain Studies and Changing Attitudes to the Welfare of Crustaceans. Animals, 15(3), 445. https://doi.org/10.3390/ani15030445
Elwood, R. W., Dalton, N., & Riddell, G. (2017). Aversive responses by shore crabs to acetic acid but not to capsaicin. Behavioural Processes. https://doi.org/10.1016/j.beproc.2017.03.022
Faulkes, Z. (2015). Motor neurons in the escape response circuit of white shrimp (Litopenaeus setiferus). PeerJ, 3, e1112. https://doi.org/10.7717/peerj.1112
Fernandez-Duque, E., Valeggia, C., & Maldonado, H. (1992). Multitrial inhibitory avoidance learning in the crab chasmagnathus. Behavioral and Neural Biology. https://doi.org/10.1016/0163-1047(92)90136-r
Fischer, B. (2023, January 23). Rethink Priorities’ Welfare Range Estimates. Rethink Priorities. https://rethinkpriorities.org/research-area/welfare-range-estimates/
Fischer, B., Gottlieb, J., Schnell, A. K., & Barrett, M. (2025). Defending and refining the Birch et al. (2021) precautionary framework for animal sentience. Animal Welfare, 34, e28. https://doi.org/10.1017/awf.2025.27
Galpayage Dona, H. S., Solvi, C., Kowalewska, A., Mäkelä, K., MaBouDi, H., & Chittka, L. (2022). Do bumble bees play? Animal Behaviour, 194, 239–251. https://doi.org/10.1016/j.anbehav.2022.08.013
Gibbons, M., Crump, A., Barrett, M., Sarlak, S., Birch, J., & Chittka, L. (2022). Can insects feel pain? A review of the neural and behavioural evidence. In Advances in Insect Physiology (Vol. 63, pp. 155–229). Elsevier. https://doi.org/10.1016/bs.aiip.2022.10.001
Gibbons, M., Sarlak, S., & Chittka, L. (2022). Descending control of nociception in insects? Proceedings of the Royal Society B: Biological Sciences, 289(1978), 20220599. https://doi.org/10.1098/rspb.2022.0599
Giniatullin, R. (2020). Ion Channels of Nociception. International Journal of Molecular Sciences, 21(10), 3553. https://doi.org/10.3390/ijms21103553
Hovland, A. L., Mason, G., Bøe, K. E., Steinheim, G., & Et., A. (2006). Evaluation of the ‘maximum price paid’ as an index of motivational strength for farmed silver foxes (Vulpes vulpes). Applied Animal Behaviour Science. https://doi.org/10.1016/j.applanim.2005.11.006
IASP. (2020). Revised definition of Pain. International Association for the Study of Pain (IASP). https://www.iasp-pain.org/resources/terminology/
Inoue, T., Hoshino, H., Yamashita, T., Shimoyama, S., & Agata, K. (2015). Planarian shows decision-making behavior in response to multiple stimuli by integrative brain function. Zoological Letters, 1(1), 1–15. https://doi.org/10.1186/s40851-014-0010-z
Jensen, E. H., Bateson, M., Neave, H. W., Rault, J.-L., & Jensen, M. B. (2024). Dairy cows housed both full- and part-time with their calves form strong maternal bonds. Applied Animal Behaviour Science, 272, 106182. https://doi.org/10.1016/j.applanim.2024.106182
Kasiouras, E., Rotllant, G., Gräns, A., Hjelmstedt, P., & Sneddon, L. U. (2026). Effects of analgesia on the response to a noxious stimulus in Norway lobsters (Nephrops norvegicus). Scientific Reports, 16(1), 12190. https://doi.org/10.1038/s41598-026-41687-w
Landgraf, D., Long, J., Der-Avakian, A., Streets, M., & Welsh, D. K. (2015). Dissociation of Learned Helplessness and Fear Conditioning in Mice: A Mouse Model of Depression. PLOS ONE, 10(4), e0125892. https://doi.org/10.1371/journal.pone.0125892
Machon, J., Krieger, J., Zbinden, M., Ravaux, J., & Harzsch, S. (2020). Exploring brain diversity in crustaceans: Sensory systems of deep vent shrimps. Neuroforum, 26(2), 73–84. https://doi.org/10.1515/nf-2020-0009
Machon, J., Ravaux, J., Zbinden, M., & Lucas, P. (2016). New electroantennography method on a marine shrimp in water. Journal of Experimental Biology, 219(23), 3696–3700. https://doi.org/10.1242/jeb.140947
Martin, T. J., Kim, S. A., & Eisenach, J. C. (2006). Clonidine maintains intrathecal self-administration in rats following spinal nerve ligation. Pain. https://doi.org/10.1016/j.pain.2006.05.027
Meth, R., Wittfoth, C., & Harzsch, S. (2017). Brain architecture of the Pacific White Shrimp Penaeus vannamei Boone, 1931 (Malacostraca, Dendrobranchiata): Correspondence of brain structure and sensory input? Cell and Tissue Research, 369(2), 255–271. https://doi.org/10.1007/s00441-017-2607-y
Moayedi, M., & Davis, K. D. (2013). Theories of pain: From specificity to gate control. Journal of Neurophysiology, 109(1), 5–12. https://doi.org/10.1152/jn.00457.2012
Operant conditioning. (2026). In Wikipedia. https://en.wikipedia.org/w/index.php?title=Operant_conditioning&oldid=134435639
Puri, S., & Faulkes, Z. (2010). Do Decapod Crustaceans Have Nociceptors for Extreme pH? PLOS ONE, 5(4), e10244. https://doi.org/10.1371/journal.pone.0010244
Puri, S., & Faulkes, Z. (2015). Can crayfish take the heat? Procambarus clarkii show nociceptive behaviour to high temperature stimuli, but not low temperature or chemical stimuli. Biology Open, 4(4), 441–448. https://doi.org/10.1242/bio.20149654
Purves, D., Augustine, G. J., Fitzpatrick, D., Hall, W. C., LaMantia, A.-S., Mooney, R. D., Platt, M. L., & White, L. E. (Eds.). (2017). Neuroscience (6th edition). Sinauer Associates is an imprint of Oxford University Press.
Reho, G., Menger, Y., Goumon, Y., Lelièvre, V., & Cadiou, H. (2024). Behavioral and pharmacological characterization of planarian nociception. Frontiers in Molecular Neuroscience, 17. https://doi.org/10.3389/fnmol.2024.1368009
Schnell, A. K., Browning, H., Crump, A., Burn, C. C., & Birch, J. (2026). Sentience in cephalopod molluscs: An updated assessment. Biological Reviews. https://doi.org/10.1002/brv.70125
(Shr)Impact. (n.d.). Shrimp Welfare Project. Retrieved April 15, 2026, from https://www.shrimpwelfareproject.org/shrimpact
Skora, L. I., Yeomans, M. R., Crombag, H. S., & Scott, R. B. (2021). Evidence that instrumental conditioning requires conscious awareness in humans. Cognition. https://doi.org/10.1016/j.cognition.2020.104546
Smith, E. St. J., & Lewin, G. R. (2009). Nociceptors: A phylogenetic view. Journal of Comparative Physiology A, 195(12), 1089–1106. https://doi.org/10.1007/s00359-009-0482-z
Sneddon, L. U. (2018). Comparative Physiology of Nociception and Pain. Physiology, 33(1), 63–73. https://doi.org/10.1152/physiol.00022.2017
Somerville, J., Albalat, A., Ellis, M., Putyora, E., Ayaril, N., & Planellas, S. R. (2026). Behavioural and neurological profiles after electrical stunning and cold shock in Pacific whiteleg shrimp (Penaeus vannamei) to inform humane slaughter (SSRN Scholarly Paper No. 6315379). Social Science Research Network. https://doi.org/10.2139/ssrn.6315379
Stewart, J., de Wit, H., & Eikelboom, R. (1984). Role of unconditioned and conditioned drug effects in the self-administration of opiates and stimulants. Psychological Review. https://doi.org/10.1037/0033-295X.91.2.251
Taylor, J. (2004). Minimizing the effects of stress during eyestalk ablation of Litopenaeus vannamei females with topical anesthetic and a coagulating agent. Aquaculture. https://doi.org/10.1016/j.aquaculture.2003.09.034
Tookey, H. (2024, October 23). Uncovering neglected issues: Advancing shrimp welfare. Rethink Priorities. https://rethinkpriorities.org/impact-area/uncovering-neglected-issues-advancing-shrimp-welfare/
Townsend, E. A., Schwienteck, K. L., Robinson, H. L., Lawson, S. T., & Et., A. (2021). A drug-vs-food “choice” self-administration procedure in rats to investigate pharmacological and environmental mechanisms of substance use disorders. Journal of Neuroscience Methods. https://doi.org/10.1016/j.jneumeth.2021.109110
Utting, M., Agricola, H.-J., Sandeman, R., & Sandeman, D. (2000). Central complex in the brain of crayfish and its possible homology with that of insects. The Journal of Comparative Neurology. https://doi.org/10.1002/(SICI)1096-9861(20000110)416:2%253C245::AID-CNE9%253E3.0.CO;2-A
Waldhorn, D. R., & Autric, E. (2023, August 11). Shrimp: The animals most commonly used and killed for food production. Rethink Priorities. https://rethinkpriorities.org/research-area/shrimp-the-animals-most-commonly-used-and-killed-for-food-production
Walters, E. T. (2018). Nociceptive Biology of Molluscs and Arthropods: Evolutionary Clues About Functions and Mechanisms Potentially Related to Pain. Frontiers in Physiology, 9. https://doi.org/10.3389/fphys.2018.01049
Walters, E. T. (2022). Strong inferences about pain in invertebrates require stronger evidence. Animal Sentience, 7(32). https://doi.org/10.51291/2377-7478.1731
Walters, E. T., & Williams, A. C. de C. (2019). Evolution of mechanisms and behaviour important for pain. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1785), 20190275. https://doi.org/10.1098/rstb.2019.027
Yang, C.-H., Ma, K. Y., Chu, K. H., & Chan, T.-Y. (2023). Making sense of the taxonomy of the most commercially important shrimps Penaeus Fabricius, 1798 s. l. (Crustacea: Decapoda: Penaeidae), a way forward. Aquaculture, 563, 738955. https://doi.org/10.1016/j.aquaculture.2022.738955
Zhai, R., & Wang, Q. (2023). Phylogenetic Analysis Provides Insight Into the Molecular Evolution of Nociception and Pain-Related Proteins. Evolutionary Bioinformatics, 19, 11769343231216914. https://doi.org/10.1177/11769343231216914