Summary
Understanding the life history of animals is important for understanding wild animal welfare, but has been understudied by animal welfare advocates. In particular, life history generalizations have been used to claim that the lives of most wild animals are net negative (see discussion of this position in Brennan 2017 ). However, there are several methods of life history classification in use in ecology and evolutionary biology. The theoretical foundations for r-K selection referred by some advocates have been discredited, and in addition some large species groups cannot be placed on this continuum. However, a related form of this classification, fast-slow is still in use in the sciences. Tripartite classification schemes seem to be more appropriate for plant, insect and fish species, which do not easily fit into a single axis. More generally, large scale reviews usually come to the conclusion that a single composite axis of variation is not sufficient to explain the wide range of life history variation. One very important point is that all classification methods are considered continuums; that is, most species will lie somewhere in the middle of axes of variation rather than at the extremes.
What is life history?
Non-human organisms have a bewildering array of modes of life. While some species have lives quite similar to our own, the majority are quite different. For example, some species have a similar body form throughout their lives, while other species, like butterflies, have quite different body forms at different stages. Some, like humans and aphids, are iteroparous (reproducing multiple times during their lifetime) others, like salmon and giant Pacific octopus, reproduce only once (semelparous). Some species, like whales and dung beetles, have very few offspring while others, like sea turtles and rabbits, have many. Some, like termites, provide extensive care for these offspring, but some, like harp seals, do not. Some species, like quahog clams, live a very long time, others, like mayflies do not. Some, like monarch butterflies, travel long distances, others stay in one location at all times. Collectively, these various aspects of organism life are termed life history.
r- and K-selection theory
Early on in ecology and evolutionary biology, efforts were made to both classify and understand this large array of life history. Scientists focused on the fact that organisms could allocate different amounts of energy and time to survival, growth, and reproduction depending on selection pressures. MacArthur and Wilson (1967), expanding on ideas from Dobzhansky (1950), coined the terms r and K-selection while attempting to explain the selection forces that determined life history tradeoffs in uncrowded and crowded environments, respectively.
These terms, r and K-selection, refer to the logistic model of population growth (Fig 1). This simple heuristic model suggests that the population growth rate is density dependent, so that per capita population growth rates are large and positive when the population density is low, and negative when the population is large and exceeds some natural limit of environmental resources (the carrying capacity, K: see Lotka 1925). MacArthur and Wilson (1967) equate r-selection with strongly seasonal environments in temperate regions, where resources are periodically available in great abundance, so that selection favours great productivity. Since the population is at low values relative to the carrying capacity of the environment and is growing exponentially at rate r, this is termed r-selection. K-selection is defined as occurring in relatively constant environments, characterized by limited resources and strong competition like regions in the tropics, where the population is at its maximum sustainable size (or carrying capacity), K. This environment is predicted to favour selection for efficient resource use, strong competitive ability, and predator avoidance.
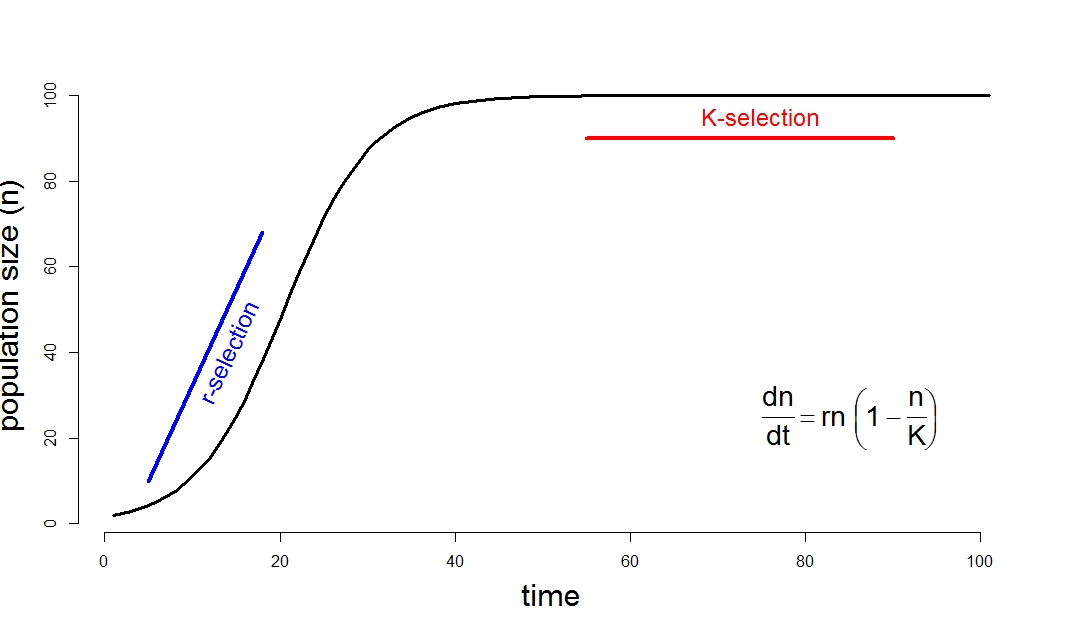 Figure 1: The relationship between population density, as predicted by the logistic equation, and r- and K-selection pressures.
Figure 1: The relationship between population density, as predicted by the logistic equation, and r- and K-selection pressures.
Pianka (1970) noted that these types of selection pressures were not restricted to the temperate and tropical zones and so expanded the scope of MacArthur and Wilson’s ideas to many more species. He further commented that no organism would ever be completely r- or K-selected, and describes an r-K continuum where various species would be placed in between these two selection extremes. He suggests that terrestrial vertebrates tend to fall on the K-selection side, while terrestrial insects tend toward the r-extreme, but notes that there are of course exceptions in each case (e.g., 17-year cicadas). He indicates that aquatic organisms do not follow this generalization, and in particular notes that fish span the entire r-K spectrum
Pianka then provides a table of life history traits that do not come directly from MacArthur and Wilson’s brief presentation, but rather represent traits which he associates with each of these extremes (Table 1). Since extreme r-selection would occur in the presence of freely available resources, when environmental conditions keep the population consistently lower than the carrying capacity, even the most unfit can acquire them. As a result, it is more advantageous from an evolutionary point of view to produce as many offspring as possible, as quickly as possible, regardless of fitness. An extreme K-strategist lives in a stable environment near the carrying capacity K, and its selection pressures are characterized by competition and threat of starvation because of severely limited resources. Since there is not enough food to go around, the offspring’s success in competition is what is most important. Here, wasting time producing numerous offspring that are not as fit as possible will doom the species to failure (see also Table 1).
Table 1: Characteristics of r- and K-selection. Modified from Pianka (1970).

The r-K selection theory was popular in the mid 1970s to 1980s (see Fig 2, and also Table 1 in Blute 2016 ) as a heuristic device. It lost importance in the early 1990s, when it was criticized because of a disparity between theoretical concepts and empirical studies (e.g., Stearns 1992, Roff 1993). Problems included the oversimplification of life history strategies along a single axis that combines both disturbance and resource availability, and the fact that many species do not match the classification (Stearns 1992). For example marine turtles have both longevity and prolificacy, and as a result, the assumption of a trade-off between r and K characteristics is not valid for these species. In addition, the traits attributed to K-selection by Pianka (1970) are not readily justifiable as a result of a constant environment, but instead merely represent a contrast to traits attributed to r-selection (Reznick et al. 2002). Moreover, this simple classification approach neglected other important factors that determine natural selection. In particular, patterns of age-specific mortality, while included in Pianka’s original classification, were not really considered as an important axis of selection (Reznick et al. 2002), nor was dispersal, nor ability to weather harsh abiotic conditions (Grime 1977).
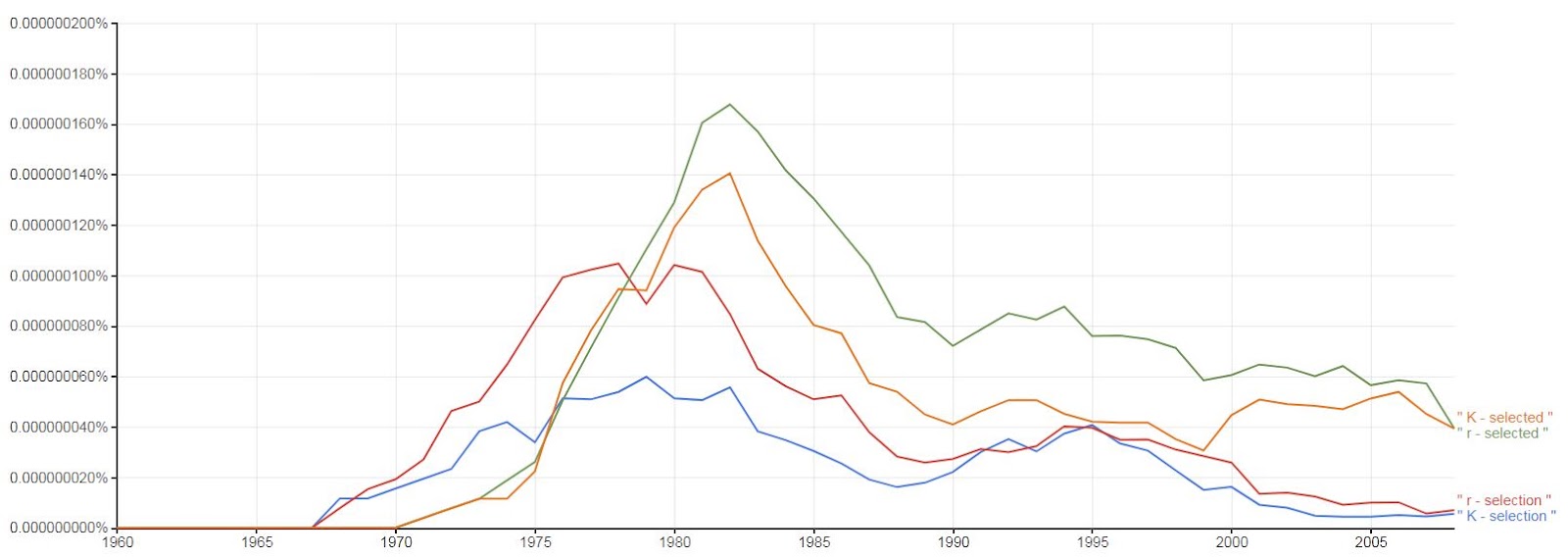 Figure 2: The use of the terms r-selection and K-selection have declined in the ecology and evolutionary biology books (measured as percent occurrence in published works), after a peak in the 80s, although they have enjoyed a resurgence in the psychology literature (produced by google ngram viewer).
Figure 2: The use of the terms r-selection and K-selection have declined in the ecology and evolutionary biology books (measured as percent occurrence in published works), after a peak in the 80s, although they have enjoyed a resurgence in the psychology literature (produced by google ngram viewer).
Reznick et al. (2002) suggest that “demographic theory” has replaced the old r- and K-selection approach, and the focus is now on determining how specific aspects of life history will affect selection. For example, increased adult mortality rates relative to juvenile rates are predicted to favor genotypes that invest in current rather than future reproduction, with early maturity and high fecundity, even if this investment comes at a cost of also producing smaller offspring (Stearns 1992; Roff 2002). Conversely, when juvenile stages experience high mortality rates relative to adult mortality rates, larger offspring may be more robust to unfavourable conditions and maternal investment in larger, but potentially fewer offspring are favoured (Stearns 1992; Roff 2002). Reznick et al. (2002) conclude that the simple r-K continuum has given way to a multitude of alternatives that can only be correctly applied if a great deal is known about an organism and its environment. However some authors still seem focused on simpler classification approaches.
Fast-slow
With the demise of r and K-selection another life history classification scheme a new but strongly related conceptual map arose in the literature. This fast-slow continuum is primarily focused on reproductive life history traits (e.g., Sæther 1987). Reynolds (2003; cited in Jeschke & Kokko 2009) describes a fast life history as characterized by early reproduction, short generation time, short lifespan, small adult body size, small offspring size, and high fecundity, while a slow life history has the opposite characteristics.
This fast-slow classification uses the vast variation in body size among organisms as a basis for comparison. Some of the most basic changes in life history are due to the fact that it takes longer for larger organisms to develop than it does for smaller organisms, and small organisms have faster metabolic rates. Thus, lifespans are longer for larger species. Jeschke and Kokko (2009) note that there are issues with this classification method in that the definition of fast and slow is not consistent among researchers. In particular, some researchers have included adult body size as a covarying trait, while others factor it out and examine the residuals of the traits. Dodson (2007) defines the fast-slow continuum as the placement of species along this scale after the effects of body size are removed, and notes that species position along this scale differs from that of body size (Dodson 2007). Therefore, small short-lived species like hummingbirds may be grouped together with large-bodied, long-lived petrals as both having low reproductive output, if one focuses on reproductive effort per unit mass.
Of those analysis that have examined the residuals of body mass, there is evidence that there may not be a single axis of life history variation that fits most species. In a large multi-species analysis of over 2000 fish, mammal and bird species, Jeschke and Kokko (2009) conclude that there is no universal fast-slow definition of life histories across these categories. For example, the direction of the relationship between fecundity and other life-history traits is the opposite in fish than in mammals. However, even within relatively tightly related groups there may be deviations from the classification. In their analysis of 267 mammal species, Bielby et al. (2007) note that some species show a mixture of fast and slow reproductive traits, and identify two-axes of variation to explain the data. The authors note in addition to their work, the foundational papers in this area (e.g., Stearns 1983, Gaillard et al. 1989 ) also identified two axes of variation: one that may be related to fast-slow classification, and another related to various aspects of the timing or energy of reproductive effort. Therefore, there are questions about the the reality of a single fast-slow continuum of life-history variation. Moreover, several studies report variation from fast to slow traits within single species across resource gradients (e.g., Singh & Mishra 2016 , Tabek et al. 2018).
In response to these challenges, some researchers have factored in the evolutionary history of the examined species as well as ease of resource acquisition in various environments to demonstrate that life history is simultaneously related to body size (physical constraints), phylogeny (evolutionary history), and more recent selection pressures associated with “lifestyle” (see Dobson 2012 for an overview). For example, in an analysis of over 600 placental mammals, Silby and Brown (2007) find that those species with lifestyles that lead lower predation rates (flighted or arboreal species) have lower reproductive output per unit mass. Nevertheless, even with these additional axes of variation, it seems clear that the fast-slow classification applies much more easily to mammals and birds than to insects and fish. However, much like r and K-selection, there are still references to a single fast-slow axis of life history variation in the literature (Nettle & Frankenhuis 2019).
Tripartite classification schemes
Three-strategy frameworks resolve some difficulties of a single axis of life history classification (e.g., r-K selection or fast-slow classification) for groups like fish and insects. There is the competition-disturbance-stress classification (Grime 1977) originally based on plants, while for fish communities, Winemiller and Rose (1992) developed the equilibrium-opportunistic-periodic model (Fig 3). Both have endpoint strategies associated with colonising vs. competitive ability, but they differ in terms of the suite of attributes associated with the third strategy. Life-history models with three strategies are generally recognised as having more predictive power than the two-strategy frameworks which can fail to recognise additional axes of life-history variation (Stearns 1977; Winemiller and Rose 1992)
Grime (1977) described three life-history strategies:competitive, stress-tolerant or ruderal (i.e., weedy) (Grime 1977; and Table 2 in Grime & Pierce 2012). This classification has the advantage of describing species with life histories that allow them to weather extremely harsh environments. Tolerant species are long-lived, with low growth and reproductive rates because of the trade-off in energy devoted to defense and survival. Competitive species are found in low disturbance environments and therefore devote less energy to reproduction and more resource acquisition, while ruderal species are found in low stress, disturbed environments with ample resources, and allocate much more energy to reproduction. Similar triangular continuums of life-history strategies were subsequently proposed for insects (e.g., Greenslade 1983).
Working with data from fish, Winemiller and Rose (1992) identify a periodic endpoint instead of the stress-tolerant strategy identified by Grime (1977). Periodic species are characterized by long lifespan, high fecundity, periodic reproduction and low investment in individual propagules favoured in habitats with large‐scale environmental variation that influences early life stage survival (sometimes called ‘bet hedging’). Under this scheme, many trees, invertebrates and fishes would be classified as periodic strategists that have large interannual and spatial variation in recruitment. This model, or similar tripartite classifications, have been applied mostly to fishes (Winemiller 1992, Juan-Jordá et al 2013), a group that has extreme variation in life history attributes relative to other animal groups. Flowering plants and arthropods are other groups that span large areas within the three part continuum identified by Winemiller and Rose (1992), but some groups, such as birds and mammals, occupy small zones (Winemiller 1992).
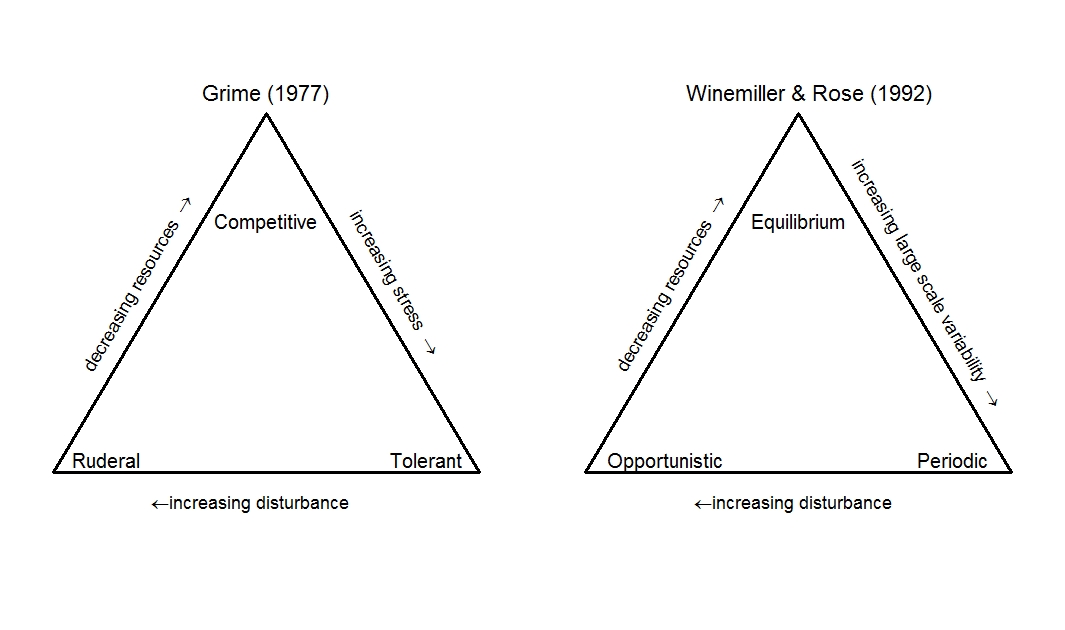 Figure 3: Tripartite axes describing selection pressure on life history for classifications from Grime (1977) and Winemiller and Rose (1992)
Figure 3: Tripartite axes describing selection pressure on life history for classifications from Grime (1977) and Winemiller and Rose (1992)
Trait-based approaches
Most recently, and somewhat outside the focus of evolutionary selection pressures, ecologists have described communities as composed of individuals with differing features. This approach is presented as an alternative to generalizing across species, which may include large variation in strategies between individuals and populations. Such trait-based ecology seeks to explain ecological patterns where individual traits will include features of life history such as lifespan and reproduction details, but also information about feeding, anatomy, social dynamics etc (e.g., Table 2 modified from Moretti et al. 2017). Such approaches have been applied primarily to plants, but interest in using these ideas for animals has steadily increased (e.g., Brousseau et al. 2018)
Table 2: Examples of terrestrial invertebrate traits important in responding to the environment. Partial list modified from Table 1 in Moretti et al. 2017.

Literature Cited
Bielby, J., Mace, G. M., Bininda-Emonds, O. R., Cardillo, M., Gittleman, J. L., Jones, K. E., ... & Purvis, A. (2007). The fast-slow continuum in mammalian life history: an empirical reevaluation. The American Naturalist, 169(6), 748-757. https://doi.org/10.1086/516847
Blute, M. (2016). Density-dependent selection revisited: mechanisms linking explanantia and explananda. Biological Theory, 11(2), 113-121.https://doi.org/10.1007/s13752-016-0241-7
Brennen, O. 2017. Infant mortality and the argument from life history. Retrieved 02.05.19 https://was-research.org/blog/infant-mortality-argument-life-history/
Brousseau, P. M., Gravel, D., & Handa, I. T. (2018). On the development of a predictive functional trait approach for studying terrestrial arthropods. Journal of Animal Ecology, 87(5), 1209-1220. https://doi.org/10.1111/1365-2656.12834
Dobson, F. S. (2007). A lifestyle view of life-history evolution. Proceedings of the National Academy of Sciences, 104(45), 17565-17566. https://doi.org/10.1073/pnas.0708868104
Dobson, F. S. (2012). Lifestyles and phylogeny explain bird life histories. Proceedings of the National Academy of Sciences, 109(27), 10747-10748. https://doi.org/10.1073/pnas.1207932109
Dobzhansky, T. (1950). Evolution in the tropics. American Scientist, 38(2), 209-221. Retrieved 23/05/2019 www.jstor.org/stable/27826306
Gaillard, J. M., Pontier, D., Allaine, D., Lebreton, J. D., Trouvilliez, J., & Clobert, J. (1989). An analysis of demographic tactics in birds and mammals. Oikos, 59-76. https://www.jstor.org/stable/pdf/3566088.pdf
Greenslade, P. J. (1983). Adversity selection and the habitat templet. The American Naturalist, 122(3), 352-365. https://doi.org/10.1086/284140
Grime, J. P. (1974). Vegetation classification by reference to strategies. Nature, 250(5461), 26. https://doi.org/10.1038/250026a0
Grime, J. P. (1977). Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. The American Naturalist, 111(982), 1169-1194. https://doi.org/10.1086/283244
Grime, J. P., & Pierce, S. (2012). The evolutionary strategies that shape ecosystems. John Wiley & Sons. https://books.google.ca/books/about/The_Evolutionary_Strategies_that_Shape_E.html?id=kDnGU-elg3kC&redir_esc=y
Jeschke, J. M., & Kokko, H. (2009). The roles of body size and phylogeny in fast and slow life histories. Evolutionary Ecology, 23(6), 867-878. https://doi.org/10.1007/s10682-008-9276-y
Juan-Jordá, M. J., Mosqueira, I., Freire, J., & Dulvy, N. K. (2013). Life in 3-D: life history strategies in tunas, mackerels and bonitos. Reviews in Fish Biology and Fisheries, 23(2), 135-155. https://doi.org/10.1007/s11160-012-9284-4
Lotka, A. J. (1925). Elements of Physical Biology. Baltimore: Williams and Wilkins. Retrieved 23/05/2019 https://books.google.ca/books/about/Elements_of_Physical_Biology.html?id=B_KTwgEACAAJ&redir_esc=y
Pianka, E. R. (1970). On r-and K-selection. The American Naturalist, 104(940), 592-597. https://www.jstor.org/stable/2459020
MacArthur, R. H., & Wilson, E. O. (1967). The theory of island biogeography. Princeton: Princeton University Press. Retrived 23/05/2019 https://books.google.ca/books/about/The_Theory_of_Island_Biogeography.html?id=a10cdkywhVgC
Moretti, M., Dias, A. T., Bello, F., Altermatt, F., Chown, S. L., Azcárate, F. M., ... & Ibanez, S. (2017). Handbook of protocols for standardized measurement of terrestrial invertebrate functional traits. Functional Ecology, 31(3), 558-567. https://doi.org/10.1111/1365-2435.12776 Supporting Information for Handbook of protocols for standardized measurement of terrestrial invertebrate functional traits Moretti et al. (2017).
Nettle, D., & Frankenhuis, W. E. (2019). The evolution of life-history theory: a bibliometric analysis of an interdisciplinary research area. Proceedings of the Royal Society B, 286(1899), 20190040. http://dx.doi.org/10.1098/rspb.2019.0040
Reynolds, J.D. (2003) Life histories and extinction risk. In: T. M. Blackburn & K. J. Gaston (Eds) Macroecology: concepts and consequences (pp. 95–217). Oxford: Blackwell https://books.google.ca/books/about/Macroecology_Concepts_and_Consequences.html?id=3vUUii37DEQC&redir_esc=y
Reznick, D; Bryant, MJ; Bashey, F (2002). r-and K-selection revisited: the role of population regulation in life-history evolution. Ecology. 83 (6): 1509–1520. https://esajournals.onlinelibrary.wiley.com/doi/pdf/10.1890/0012-9658%282002%29083%5B1509%3ARAKSRT%5D2.0.CO%3B2
Roff, D. A. (1993). Evolution Of Life Histories: Theory and Analysis. Springer https://books.google.ca/books?isbn=0412023911
Roff, D. A. (2002). Life history evolution Sunderland: Sinauer https://books.google.ca/books/about/Life_History_Evolution.html?id=M_ZzQgAACAAJ&redir_esc=y
Sæther, B. E. (1987). The influence of body weight on the covariation between reproductive traits in European birds. Oikos, 79-88. https://www.jstor.org/stable/3565691
Sibly, R. M., & Brown, J. H. (2007). Effects of body size and lifestyle on evolution of mammal life histories. Proceedings of the National Academy of Sciences, 104(45), 17707-17712. https://doi.org/10.1073/pnas.0707725104
Singh, N., & Mishra, G. (2016). Slow and fast development in two aphidophagous ladybirds on scarce and abundant prey supply. Bulletin of Entomological Research, 106(3), 347-358.
https://doi.org/10.1017/S0007485316000080
Stearns, S. C. (1977). The evolution of life history traits: a critique of the theory and a review of the data. Annual Review of Ecology and Systematics, 8(1), 145-171. https://doi.org/10.1146/annurev.es.08.110177.001045
Stearns, S. C. (1983). The influence of size and phylogeny on patterns of covariation among life-history traits in the mammals. Oikos, 173-187. https://www.jstor.org/stable/3544261
Stearns, Stephen C. (1992). The Evolution of Life Histories. Oxford: Oxford University Press. Retrieved 23/05/2019 https://books.google.ca/books/about/The_Evolution_of_Life_Histories.html?id=-NcNAZ06nNoC&redir_esc=y23/05/2019
Tabak, M. A., Webb, C. T., & Miller, R. S. (2018). Propagule size and structure, life history, and environmental conditions affect establishment success of an invasive species. Scientific Reports, 8(1), 10313. https://doi.org/10.1038/s41598-018-28654-w
Winemiller, K. O. (1992). Life-history strategies and the effectiveness of sexual selection. Oikos, 318-327.https://www.jstor.org/stable/3545395
Winemiller, K. O., & Rose, K. A. (1992). Patterns of life-history diversification in North American fishes: implications for population regulation. Canadian Journal of Fisheries and Aquatic Sciences, 49(10), 2196-2218. https://doi.org/10.1139/f92-242
Credits

This essay is a project of Rethink Priorities. It was written by Kim Cuddington. Thanks to Jason Schukraft, Daniela Waldhorn , David Moss, Marcus Davis and Peter Hurford for comment. If you like our work, please consider subscribing to our newsletter. You can see all our work to date here.

You've done a good job at reporting the trends in thought and terminology here. I'm not directing the following at you, but at the trend in the field you're describing.
I'm an evolutionary biologist and I'm tired of people saying r/K has been discredited. I think what really happened is that people realized r/K was a generalization without realizing that every other useful principle in evolutionary biology is also a generalization.
I use r/K parlance and I never get any complaints from the evolutionary theorists and population geneticists around me. It's just a heuristic. Would you say the logistic model of population dynamics has been debunked because someone points out that it's doesn't capture every variable that affects population growth? No, because it's just a model, so that was obvious from the start. Hence I don't see why people pointing out that there are other dimensions to life history somehow invalidates using the r/K spectrum as a knowing simplification. I'm all for clarifying that r/K is just a heuristic and educating people about the fundamentals of life history theory, but I don't think the fact that there's more to it invalidates r->K as a useful dimension.
There's never going to be a life history theory that's both 100% accurate and can provide generalizations at the gross level at which we typically consider life history traits. In order to make any useful statements about the relationship between offspring number and life span, for example, we're going to have to allow for exceptions.
Yes, I hope I made it plain that r- and K- classification is still in use, and that there were a variety of critiques, not just the fact there were are exception to the generalization.
I'm curious tho, some of Pianka's associated traits have opposite relationships to those stated in big taxonomic groups. Notably for insects, reptiles and fish, "generally" reproductive output increases with body size as compared to mammals and birds where it decreases. As an evolutionary biologist what is your take here? I can think of half a dozen explanations, but never found a literature consensus (e.g., Pianka just got the traits wrong for r - and K and now we use...., r and K- not good for these groups, body size relationships within groups not so important etc etc)
Kim: This is an interesting and well-structured post, but as far as I can tell, it doesn't include any information about how the material relates to any EA cause areas. I can guess at how thinking about life history might influence our views about, say, wild animal suffering, but I may be wrong, and I think many readers will be confused to see this on the EA Forum without further explanation.
Would you mind adding a brief explanation of how someone interested in animal issues within EA might benefit from learning this? Or some other description of how this material relates to EA issues? I'll update the post to the "Frontpage" category if an explanation is added.
I have added some context at the beginning of the piece, since you are quite right that it might not be obvious that this post is about determining the grounds for some wild animal welfare claims.
Thanks for the edit! Moved to Frontpage. I'm still curious how moving away from generalizations lets us push against the idea that most wild animal lives are net-negative, but I have a better sense of where you're coming from now.
Essentially, the life histories of most species are not well captured by the classification schemes used in animal welfare arguments (e.g., most species are neither r- nor K-selected). As a result, it seems much more difficult to argue that the welfare generalizations based on these schemes correspond to the actual affective experiences of most individuals.
I still don't understand which claim you're making, exactly.
Are you saying:
1) "most animals don't have as many offspring as was previously stated (mistakenly, based on the r-/K-selection model), and therefore we can't be as sure that most animals live short and gruesome lives,"
or 2) "most animals don't have as short a life span as was previously stated (mistakenly, based on the r-/K-selection model), and therefore we can't be as sure that most animals live short and gruesome lives,"
or 3) something else?
I thought the claim about r-/K-selection was always about number of offspring and lifespan, rather than other aspects of the model (competition, body size, etc.), and your article doesn't seem to suggest that these are very different from what was previously argued.
r-K selection theory suggests that some particular life history characteristics, such as short lifespan and many offspring, are tied together by selection forces. This is not true for some large groups.
Just because you belong to a group with high fecundity does not mean that either 1. total expected adult lifespan is relatively short or 2. that the highest mortality occurs at the youngest ages. For example, see our post on insect life history and in particular our reply to Tomisk regarding variability in survivorship curves.
I think we need to dig into the data to get stats on things like number of offspring. We are saying that not all species that people might describe as r-selected have many offspring, and it would be better to look at the data for different species or species groups than to use life history generalizations.
Thanks for the response!
Some recent work on how life history relates to trade-offs between traits and performance:
https://academic.oup.com/icb/search-results?f_TocHeadingTitle=Integrative%20Life-History%20of%20Whole-Organism%20Performance%20(SICB%20wide)
A lot of the initial life history work was only qualitative, but it is definitely moving in a more quantitative direction.