This essay was submitted to Open Philanthropy's Cause Exploration Prizes contest.
If you're seeing this in summer 2022, we'll be posting many submissions in a short period. If you want to stop seeing them so often, apply a filter for the appropriate tag!
Summary
- Tuberculosis kills on a scale of around 1.5 million people per year.
- The Global Plan to End TB established that 13.6 billion USD would be needed to reach SDG elimination targets by 2030, but reaching this level is being missed annually, meeting only 5.3 billion (39%) since being established. Silva et al. estimate that missing these targets and continuing our current course would represent an additional 15 years and 5.7 million lost lives. On a macro scale, bringing TB funding up to the required level would represent a cost-effectiveness of 1456 USD per life saved (8.3 billion USD / 5.7 million lives ≈ 1456).
- Specific interventions corroborate this figure, with some interventions such as mass case detection reaching a cost-effectiveness of 877 USD per life saved.
- Silva et al. also estimate a financial cost of 3 trillion USD as a result of this 15 year delay, which could be averted with present funding.
Major sources of uncertainty
The main sources of uncertainty for this proposal are:
- Latent tuberculosis infection is so widespread that exact prevalence can be difficult to estimate, with an innate need for mathematical modelling over medical record data. Mortality and incidence are more reliable (derived from IMHE data) and are therefore used in calculations
- Given the number of ways to approach TB elimination and the scale of TB infection globally, there are likely many more cost-effective routes to take and novel approaches that are not considered in this proposal. Regional variation also makes it critical to recruit local expertise in implementation of programmes and to determine the generalisability of any research results.
- Salaries for community health workers, in particular, seem to vary hugely according to sources. Dey et al. list a salary as low as 3000 INR (43 USD) per month for a real world programme, and note that this low pay was one of the major sources of attrition of health workers on the programme. Instead, a salary average based on the National Health Mission in India of 20 965 INR (265 USD) per month was used for this proposal. Note that even if salaries were 80 000 INR (1011 USD) per month, it would still rank among the most cost effective charities currently listed by GiveWell.
- Co-ordinating the wide variety of actors in this space might present difficulties or cause directed funding to get “lost in the shuffle” of many different efforts.
Importance
Tuberculosis is a disease that, if left untreated, typically kills around 70% of people that develop active symptoms. It is estimated that up to a third of the world’s population is infected with tuberculosis (TB). With 10 million new infections causing 1.5 million deaths per year, this ancient disease has been infecting humans since at least the development of agriculture and the domestication of bovine animals. While Europe has steadily moved towards elimination since the 19th century, aided in part by the creation of effective antibiotics and improved health infrastructure, TB remains at high levels of endemicity across much of the world.
Despite successes in India and China in establishing universal directly observed therapy, short-chouse (DOTS) coverage and reduction in overall numbers, hotspots of high infectivity and multidrug-resistant TB threaten to undermine these achievements. A combination of relative underfunding in comparison to other infectious diseases and the recent COVID-19 pandemic have led to a potential reversal of a decade or more of progress.
There are hopes to rapidly reestablish disrupted TB programmes and utilise the first new generation of anti-TB drugs in 40 years in shortened treatment regimens to more efficiently reach the world’s population. With public attention directed toward infectious disease and a proven willingness to rapidly develop treatments, as well as an upheaval of many health services globally, this might be considered a decisive moment (or a ‘hingey’ moment in EA terms) in the course of TB elimination; according to models created by Silva et al., actions taken within the next five years could change trajectories of elimination by up to 15 years, representing a loss of 5.7 million lives.
There are a range of interventions with high cost effectiveness, potentially <$100 per DALY averted / <$1000 per life saved. Furthermore there is significant scalability and need for large amounts of funding in the region of $4bn per annum. This makes the area an ideal target for OpenPhilanthropy or GiveWell donations, with several interventions showing potentially higher cost-effectiveness than the current suite of global health interventions.
In addition to the pervasiveness and deadliness of TB described in the summary, progress towards TB elimination has faced several challenges in the last decades. Few countries met the expressed targets for funding at the 2014 World Health Assembly, and COVID-19 brought about considerable interruptions to services for a variety of diseases and conditions, including TB care. There was an 18% decrease in the number of cases diagnosed and reported, a 15% reduction in the number of individuals treated for drug-resistant TB, and a 21% decrease in the number receiving preventive treatment in 2020.
Mortality cost
In 2020, the number of TB deaths rose for the first time in over a decade. A total of 1.5 million people died from TB in 2020 (including 214 000 people with HIV). Worldwide, TB is the 13th leading cause of death and the second leading infectious killer after COVID-19 (above HIV/AIDS). The estimated DALYs reaches 122 million per year, and even among those that undergo successful treatment, post-tuberculosis sequelae account for 58 million (47%) of that figure.
The issue is not only achieving reduction, but a better rate of reduction; the rate at which TB declines translates to differences of mortality figures in the millions, and DALYs in hundreds of millions. Silva et al. estimate the difference between reaching the 2030 SDG target versus 2045 represented a loss of 5.7 million lives.
By Silva et al.’s estimate, simply making up the estimated funding needed to reach the SDG target to prevent the loss of life in that 15 year delay would represent a cost-effectiveness of 1456 USD per life saved (8.3 billion USD / 5.7 million lives ≈ 1456).
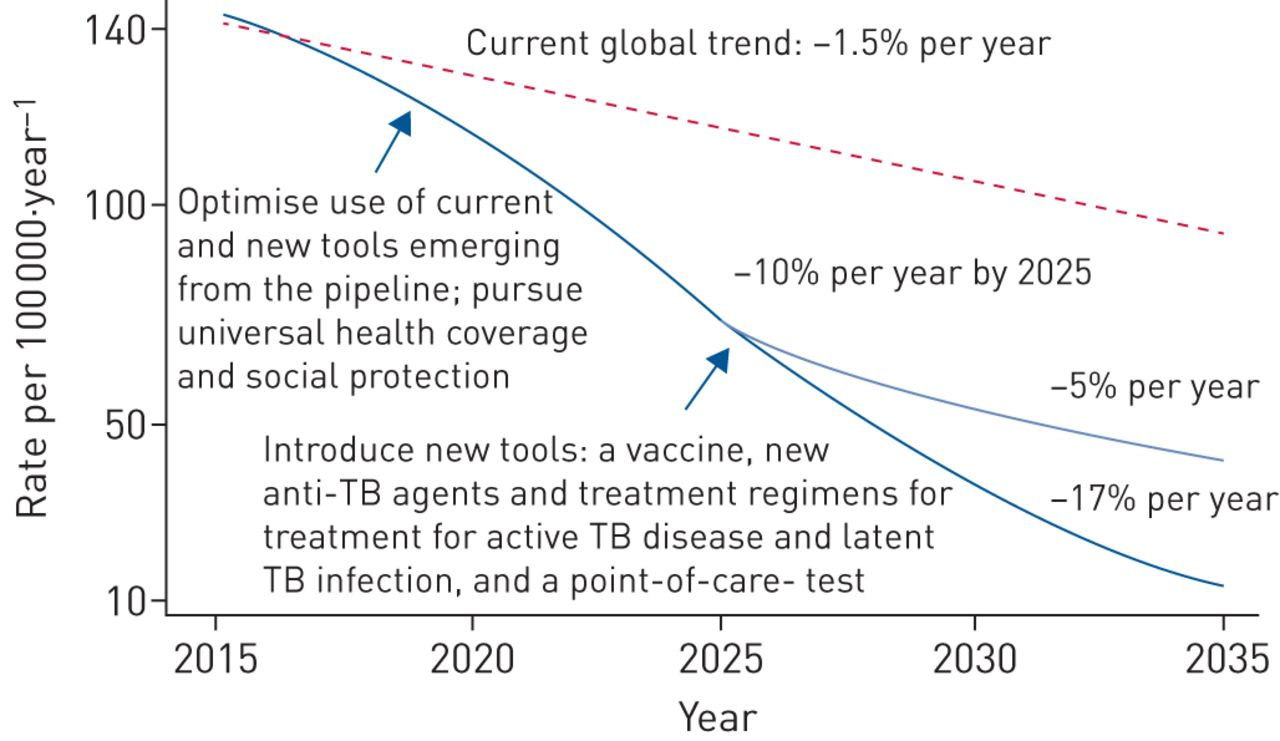
Fig. 1: Projected trend in the decline of global tuberculosis incidence, from 110 cases per 100 000 in 2015 to 10 cases per 100 000 or less by 2035, reproduced from the World Health Assembly resolution on global strategy and targets for tuberculosis prevention, care and control after 2015. Note the End TB target differs slightly from the SDG target (with complimentary reduction tby years 2035 and 2030, respectively).
Financial cost
In a study of 120 countries, Silva et al. found that the cost of inaction of not meeting the SDG tuberculosis mortality target until 2045 versus 2030 would cost $3.0 trillion in economic losses, with those effects falling disproportionately on Sub-Saharan Africa.
A study published in 2013 found that TB inflicts annual direct health costs of more than 500 million euros ($670 million) on European governments, and costs another 5.3 billion euros in productivity losses. This shows that selective inattention in important areas of disease burden, particularly those that exacerbate existing problems of inequality, presents a serious drain even in those countries with a lower TB incidence.
Finally, on a microeconomic level, a systematic review conducted by Taminura et al. found that the financial burden on families affected by TB can be catastrophic, with the greatest effects coming via income loss.
Neglectedness
Current actors
A broad coalition of actors currently work together on TB efforts. The World Health Organization’s End TB strategy brings together national programmes (via the Stop TB Partnership) to co-ordinate TB prevention and control efforts across countries. While member states committed to mobilising at least 13 billion USD year by 2022, and an additional 2 billion USD per year for TB research in the 5-year period 2018–2022, spending fell from 2020 to 5.3 billion USD. This is less than half (39%) of the amount estimated to be required in the Global Plan and less than half (41%) of the global target set at the UN high-level meeting on TB. A part of the reason for this underfunding may be due to a combination of the lack of scope for advance market agreements for TB drugs in LMICs and the perceived scale of the problem taking precedence over the evident cost-effectiveness of TB interventions. The 2021 Report on TB Research Funding Trends from the Treatment Action Group puts this shortfall in stark terms, illustrating recent funding versus targets:
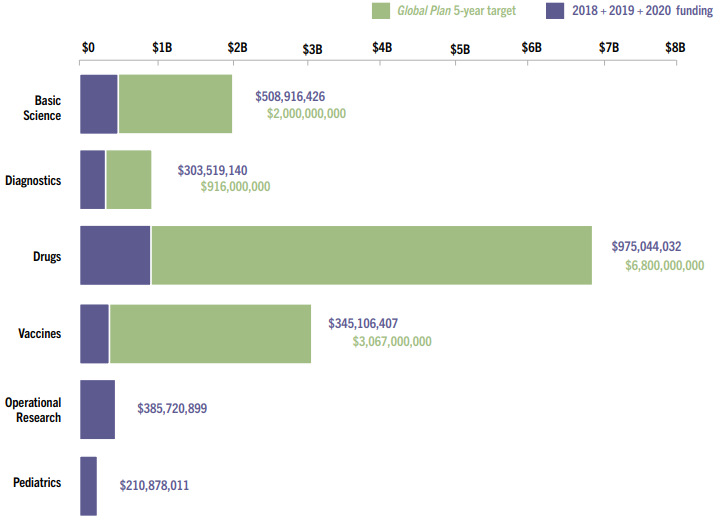
USAID and The Global Fund are the leading international contributors to TB prevention and control efforts, spending US$ 0.8 billion in 2020, 15% of the total of US$ 5.3 billion spent globally. As a portion of the total funding needed for full implementation of national TB plans, the Global Fund covered 39% of the need in LICs, 33% of the need in lower-middle income countries, and 2% of the need in upper middle-income countries. Unlike HIV/AIDS and malaria, the majority of TB funding comes from domestic sources.
Scientific organisations such as the TB Alliance, the TB Vaccine Initiative (TBVI), FIND, and the European and Developing Countries Clinical Trials Partnership share and partially fund research into new technologies for TB diagnosis, prevention, and treatment. For example, the TBVI published in their 2021 report that several vaccines (namely VPM1002, GamTBvac, M72/AS01E, MIP, and MTBVAC) are in phase III trials; typically the last phase of testing before data for a medical intervention is sent away for approval to a regulatory body. Ensuring regulatory approval of such upcoming vaccines is one of the suggested areas of possible intervention.
Direct action groups such as Médecins Sans Frontières, GAVI, and Partners In Health provide logistical support and/or direct care in some of the more difficult settings to treat TB. Finally, advocacy groups such as TB Alert, the Treatment Action Group, and the MSF Access Campaign pressure state and private actors to invest more in research and development for neglected diseases, including TB.
Funding shortfalls and comparisons
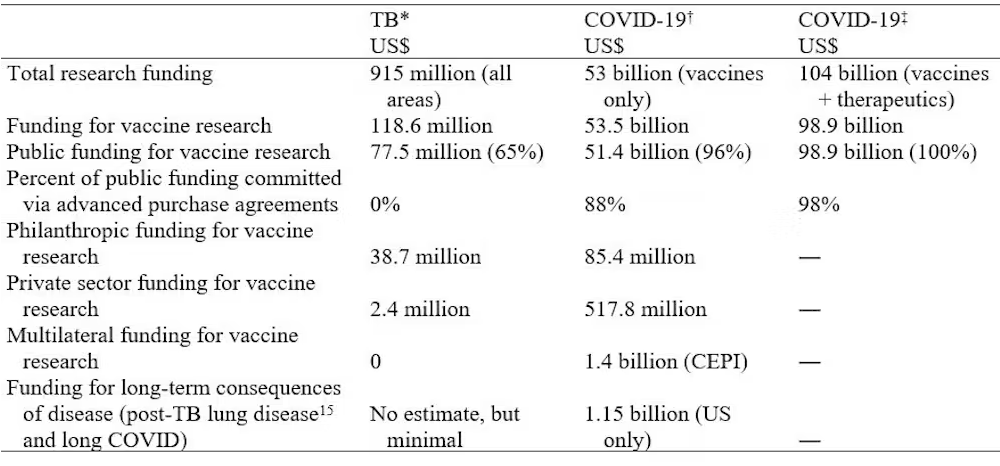
Professor Richard Chaisson of Johns Hopkins University argues that TB warrants being treated as a pandemic on the scale of COVID-19. Not only do the two dangerously exacerbate one another, but they kill on a comparable scale; the WHO places COVID-19 deaths at 6.4 million as of this competition entry, whereas even a low estimate of TB deaths since 2019 would reach 5.3 million[1] , though both are likely underestimates. Despite this, the former has received more than 112 times more in terms of R&D funding, with the benefit of advanced market commitments.
Table 1: A comparison of government funding for research on TB and two estimates for COVID-19 therapeutics and vaccines, 2020, taken from Chaisson et al.
Even counting only among those diseases recognised as neglected tropical diseases (NTD) by the World Health Organization, the scope of TB funding has been found not to match the relative DALY and mortality burden. The situation for NTDs has greatly improved in recent years, with DALYs lost from diseases included in the London Declaration declining 21.1% from 2010 to 2016, whereas TB progress and funding has demonstrably stalled.
Wang et al. highlight that TB closely matches an NTD through “the basis of its underlying burden of disease, influence and effect on poverty and development, and neglect through political will and funding.” The authors note that there are overarching principles for the design and implementation of NTD control programs that could be applied to ending TB.
Tractability
TB, perhaps more than any other infectious disease, presents multiple open avenues for improved prevention, treatment, and control. The recent drop in funding and stalling of health services as a result of the COVID-19 pandemic offers an obvious and urgent cause area – simply rebuilding previous health services would be an improvement. Looking beyond restoration to more ambitious targets, the epidemiology of TB indicates several intervention paths addressing social determinants, especially poverty, food security, and living environment. Since the disruption of health services, case finding has become an important aspect of recovery.
The biomedical approach offers increasingly promising interventions. In 2010, a third of TB diagnoses went undiagnosed, and countries representing the majority of TB burden have suffered significant declines in TB diagnosis and treatment ranging from 16% to 41%. Improved diagnostics would allow community health workers to successfully find and refer people to treatment for TB. After many years of a dearth in new anti-TB drugs, trials of bedaquiline, delamanid, and pretomanid showed their efficacy in the treatment of TB and MDR-TB, even on much shorter regimens. The most ambitious and experimental areas of research might even allow us to bypass drug resistance altogether through biologic therapies. Finally, multiple vaccine candidates have now reached phase III trials, and additional funding for their continued testing in different countries and national rollout programmes presents a highly impactful area.
Given the scope of TB mortality and infection, as well as the open choice of pursuing new interventions or “plugging in” to many existing programmes, direct funding and/or advocacy of any of the above could yield highly impactful results. In addition to this, tuberculosis elimination is achievable even with current tools, and the precendence set by both COVID-19 and the relative success of NTD control, appears highly tractable. The key difference is how long the path to elimination will take; indeed, Wang et al. mention neglect by political will and funding as two critical bottlenecks – areas where Open Philanthropy can help.
In terms of selection between potential options, a subject matter expert consulted for a recent article in the Metaculus journal emphasised the importance of not pitting advocacy for long-term economic development against biomedical funding and technological solutions, stating that “waiting for poverty alleviation without investing in research means accepting deaths and suffering for decades. We need poverty relief, better living conditions and new tools which also means access to them for all and especially those populations and persons most at risk”.
Therefore, I have attempted to make the interventions below cover a range of approaches with emphasis on different geographic areas of impact:
Table 2: A summary table of possible TB interventions
| Intervention | Does it work? | Cost-effectiveness | Fund Research, Fund Directly, or Advocate For? |
| Mass case detection | Yes | 877 USD per death averted | Fund Directly; the cost effectiveness of this intervention appears highly favourable |
| TB vaccine candidates | Promising | Between 149 USD per DALY recovered in adults and 1692 USD per DALY recovered in children | Fund Research or Advocate For |
| Scaling up TB preventive therapy | Promising | Between 31 USD in South Africa to 7608 USD in Brazil | Fund Directly or Advocate For |
| Improve regulatory approval times for anti-TB drugs | Difficult | 285 USD per DALY recovered | Fund Directly; non-profits largely fund these clinical trials but lack resources to get them through regulatory approval. Alternatively, Advocate For; policy changes could allow cheaper regulatory approval? |
| Biologic / peptide therapies | Difficult | Unknown, but possibly with a large favourable effect | Fund Research |
| Public manufacturing of rifapentine in Europe | Yes | 27 948 USD per QALY gained | Fund Directly or Advocate For |
Possible interventions
Multiple promising avenues for TB prevention and control exist. I recently pitched the following ideas which I believe to be potentially very high impact, with rough but favourable cost-effectiveness calculations attached:
Mass case detection via Community Health Workers (CHWs) in areas with high endemic TB
Aims to replicate the Tuberculosis Health Action Learning Initiative (THALI) programme led and funded by USAID. CHWs are trained in TB case detection and referral combined with an understanding of local communities.
Estimated impact per USD spent:
1 424 640 USD / 1624 deaths averted = 877 USD per death averted, as per below:
- 31 617 matches to case definition found in Potty et al.; 3841 tested positive of which 3812 started treatment. With an 89.9% success rate among 2580 followed up cases, 2319 TB cases were cured. Assuming a 70% case fatality rate in untreated TB cases as found by Tiemersma et al., then 1624 deaths were averted.
- Assuming CHW salary of 251 580 INR (3180 USD) per year for a 4 year programme, 112 health workers could be employed for 1 424 640 USD.
Reasoning:
Cases that aren’t detected can’t be treated. CHWs play a critical role in contacting hard-to-reach populations that often make up the majority of TB patients in high-incidence countries, such as India. Depending on the salary of the CHWs and assumptions used, simply detecting and referring cases to existing services in areas of high TB prevalence could have a profound impact of 847 USD per death averted. This has high potential for replication across ‘slum’ areas in other Indian cities.
Investment in TB vaccine candidates
Several TB vaccines are in development, with useful overviews of ongoing clinical trials given as of October 2021 by the TB Vaccine Initiative and as of February 2020 given in Table 1 of Li et al.. In this case, advocacy could be directed toward existing efforts, the most promising vaccine candidate, or new potential candidates such as an mRNA vaccine.
Estimated impact per USD spent:
Highly variable depending on the final clinical properties of each vaccine. Knight et al. suggest that, between 2024 - 2050, a vaccine with 10 year duration and 60% efficacy would cost 149 USD per DALY recovered if effective in adults and 1692 USD per DALY recovered if effective only in children.
Reasoning:
The BCG vaccine is now over a century old (first used in humans in 1921). It is effective in preventing TB in children, especially TB meningitis, but has limited efficacy in adults. With the recent increase in focus on the potential for rapid production of vaccines with COVID-19, now may be an opportune time to contribute to existing and newly emerging efforts to create a new TB vaccine.
Scaling up TB preventive therapy (TPT)
High-risk groups within the population of people with TB, such as those co-infected with HIV, are shown to benefit from TB preventive therapy. The WHO has already emphasised the importance of explanding TPT to those living with HIV and household contact persons of people with TB disease.
Estimated impact per USD spent:
Nsengiyumva et al. find that scaling up TPT to 100% coverage would hypothetically cost 7,608 USD per DALY averted in Brazil, and 31 USD per DALY averted in South Africa compared to the status quo, assumiing that scale-up can be achieved over a 3-year period.
Reasoning:
TPT could help to improve the overall picture of TB epidemiology in two ways: by reducing the number of deaths among high-risk groups, and also in reducing transmission in TB hotspots. This intervention is also set to become more effective in combination with research into new anti-TB drugs and shortening therapies.
Faster regulatory approval of future vaccines and novel antituberculosis drugs
Clinical trials like TB-PRACTECAL have shown that novel TB drugs can cut treatment times from 12 months to 6 months or less, and also eliminate the need for injectable drugs through the use of more effective oral drugs. This presents a considerable DALY-saving and cost-saving benefit, and also reduces the burden on the patient through a difficult, complicated, and often unpleasant treatment.
Estimated impact per USD spent:
While the exact cost-effectiveness of advocacy for regulatory approval is hard to measure, there are several studies of the new generation of anti-TB drugs. Cost-effectiveness figures of bedaquiline are variable between countries. Table 3 in Gomez et al. gives an indication of where it might be most effective to target advocacy efforts, with the highest DALYs recovered per USD spent in South Africa at 285 USD per DALY recovered.
Reasoning:
Many novel drugs for neglected diseases including TB are developed in the not-for-profit domain, leaving them without support in post-regulatory approval stages. This creates an administrative (and potentially rectifiable) bottleneck for life-saving drugs.
Investment in early-stage biologic/peptide therapies
According to Rivas-Santiago et al., antimicrobial peptides “have demonstrated remarkable efficacy to kill mycobacteria in vitro and in vivo in experimental models”. Khusro et al. give an overview of the biological mechanisms. Dijksteel et al. provide an overview of all clinical trials and therapeutic uses in humans in Table 1 of their article; note that none specifically target TB as of the article publication on 22 February 2021.
Estimated impact per USD spent:
Very difficult to calculate at this stage.
Reasoning:
Included as a high-risk, high-reward, biomedically-focused option for future TB treatment. Potential impact could be the dramatic reduction (or even elimination) of need for traditional TB drugs and replacement of their use with peptide therapies, bypassing drug-resistance. Note that this is very early stage research and it is almost certain that there would be difficulties in testing for human use, wide-scale manufacture and rollout.
Public manufacturing of rifapentine and/or registry of rifapentine with the UK Medicines Regulatory Agency and European Medicines Agency
Rifapentine is a synthetic derivative of rifampicin with a longer half-life and promising anti-microbial effects on TB. Sanofi is currently the only manufacturer of rifapentine in Europe, but since this is not currently registered with either the UK or EU Medicines Agencies, this is not available to the staff attending the 260 000 annual TB cases reported in the EU or the 4000 annual TB cases reported in the UK.
Estimated impact per USD spent:
A global model of all treatment regimens utilising rifapentine found them to be more cost-effective than previous regimens at 27 948 USD per QALY gained.
Reasoning:
“Low-hanging fruit” in terms of the straightforwardness of the intervention, the local intervention setting (UK and EU), wide array of supporting agencies (including WHO), and ability to manufacture. Some European organisations are already discussing possibilities for public manufacturing of the drug.
Acknowledgements
Many thanks to Sam Hilton and Zachariah Baker for their review and suggestions on this proposal.

This post/cause seems sorely underrated; e.g. what org exists can someone donate to, for mass case detection? It has such a high potential lives-saved-per-$1,000!
Thanks Nicholas. I'm still advocating for this and submitted a more specific project proposal to several EA-affiliated organisations in late 2022. I understand at least two of these organisations are exploring TB as a potential cause area.
I would love to join the Charity Entrepreneurship Incubation Programme with this idea, and have also considered founding a charity independently. Perhaps one day I can direct you to an organisation I'm directly involved in.
Until then, a good first reference might be the grantees of the TB REACH programme, listed (alongside the results of their grants) in this PDF: https://stoptb.org/assets/documents/resources/publications/technical/TB_Case_Studies.pdf. Another idea would be to donate directly to the Stop TB Partnership, part of UNOPS.
These are mostly larger organisations that have directed some of their resources to case detection, rather than dedicated charities. As far as I know, no such single-focus charity exists (yet).
"directly observed therapy, short-chouse (DOTS) coverage" looks like a somewhat confusing typo of "directly observed therapy, short-course (DOTS) coverage", unless I'm about to learn a new medical term.
Great essay - thanks for writing. I think one interesting element to explore is the race against drug resistant TB, and how that could set back progress/increase costs in a way that narrow cost-effectiveness analysis can't really show.
When thinking about funding - I think it's clearer to see how philanthropy can/should support in low income settings. I'm not convinced that charitable money should be needed to set up new manufacturing in Europe, especially if that's likely to happen anyway.
Thanks for your comment. Just a sidenote to your point: I think it's a curious element of TB history that we've known about the dangers of drug-resistant strains since at least the 1950s, not just as a potential future threat but right away in real world practice; the reason streptomycin is combined with other powerful antibiotics (e.g. isoniazid and others) as part of standard TB treatment is because that's the only way to completely clear all bacilli. I wrote a bit about this in my piece for Metaculus: https://www.metaculus.com/notebooks/12130/from-reduction-to-elimination-replicating-the-successes-of-historical-tuberculosis-control/
I haven't looked into projections of MDR-TB specifically, but this would definitely be a worthwhile line of inquiry. Interestingly, a quick search shows some advocacy for combined TB and AMR control efforts in LMICs: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5840628/
https://lshtm.ac.uk/newsevents/news/2020/where-and-how-does-tb-fit-amr-agenda
I agree, I would also choose other interventions over manufacture of rifapentine in Europe, though I am not sure it's likely to happen without incentives for Sanofi. As helpful as it might be in countries like Moldova, Estonia, etc., I think the preceding cause areas would have a far greater impact (they're listed in order of my best guess at their value).
I added this mainly because I wanted to include a variety of options. Open Philanthropy may opt for a less effective but more tractable option, for example. It also shows just how many possible avenues there are to contribute to TB efforts.