This essay was submitted to Open Philanthropy's Cause Exploration Prizes contest and posted to the Forum with the author's permission.
Executive summary and transformational goal
Increasing access to infertility services in LMICs directly matches the Open Philanthropy’s criteria of importance. Between 48 million couples and 186 million individuals live with infertility globally (WHO, 2022), a condition which has tremendous impact on their social and mental wellbeing, especially in LMICs. Africa hosts the countries with the highest infertility prevalence in the world, spiking at 20-30% of couples of child-bearing age. For these couples, infertility leads to a form of “social death”, with the most deprived populations being the most prone to stigmatization. Over 50% of these cases of infertility could be easily prevented, and ~66% could be treated. Last but not least, evidence suggests integration of infertility prevention and treatment in Reproductive Health services increases the acceptability of other family planning services (e.g. modern contraception).
Increasing access to infertility services in LMICs is a highly neglected clause. “Family planning is about deciding how many children you choose to have and when you want to have them” (WHO, 2013). Although access to contraceptives in Low and Middle Income Countries (LMICs) has significantly improved over the past ten years, the other side of the reproductive healthcare equation – i.e. access to infertility care – remains reserved to ~1.5% of the population[1] in most LMIC countries.
Moreover, tractability is high: Open Philanthropy has a unique opportunity to radically transform the state of access to infertility services in African countries through a two-pronged approach: 1) partnering with NGOs and Ministries of Health in LMICs to transform their current ambition to act into evidence-based policies and cost-efficient patient pathways; and 2) a Market shaping intervention, which would increase the accessibility of drugs and equipment necessary to the prevention, diagnosis and treatment of infertility.
Importance
Nine percent of child-bearing age couples worldwide are currently experiencing infertility (Boivin et al., 2007), defined as the inability to conceive a child within one year of unprotected sex. Contrary to popular belief, the disease burden is heavily skewed towards the world’s poorest countries, with WHO (2017) reporting that “one in every four couples in developing countries had been found to be affected by infertility”. Rates vary greatly across countries; with a so-called "African infertility belt” spanning from Tanzania in the East to Gabon in the West (WHO, 2010). For instance, a 2017 study of the DHS survey of ~39,000 Nigerian women found a 31.1% prevalence of infertility (Polis et al, 2017).
In several African countries, married women are referred to by the name of their eldest child (e.g. “Mama George”). For “Mama and Papa Nothing” (Dhont et al, 2011), the consequences of infertility in developing countries range from severe economic deprivation, to social isolation, murder and suicide. Studies in Nigeria, Mozambique and South Africa revealed that life without children is perceived as unhappy and not worth living (Dyer, 2007). Psychological distress is significantly more common in infertile women in comparison to their fertile counterparts (Dyer et al., 2005). In Andhra Pradesh, India, ~70% of women with infertility report experiencing physical violence as a result (Unisa et al, 1999). In a study in Nigeria, half of infertile women evaluated were diagnosed with depression (Upkong & Orji, 2006). As explained by Professor Basil Tarlatzis, President of the International Federation of Fertility Society (IFFS): “Womanhood is defined by motherhood so infertile women can be seen as 'cursed' and are deprived of all social status”. As such, the consequences of infertility are more extreme in developing countries due to established social constructs of family and the lack of support. Daar and Merali (2002) have mapped out the consequences of infertility into a six-step scale (see Figure 1). Ombelet (2008) explains that “in developed countries, the consequences of infertility rarely extend beyond level 2, in developing countries the consequences are infrequently as mild as level 3”.
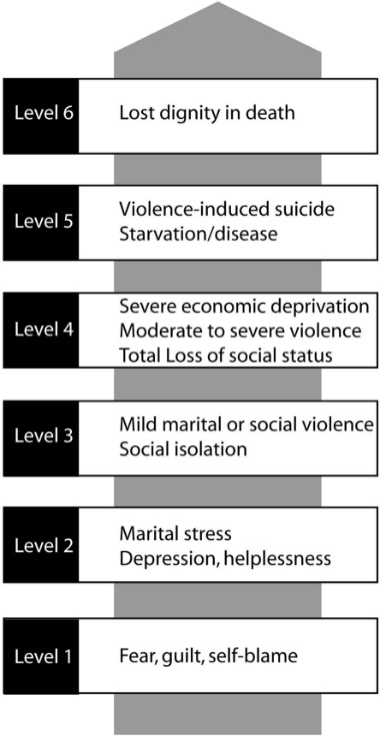
Indeed, Aleyamma et al (2011) stress that:
“In the developing world, marriage is almost universal and often occurs at an early age with the ultimate goal being childbearing. Couples bearing the burden of infertility are subjected to social stigma, community isolation and harassment from family members, to the extent of even being excluded from inheritance rights (Van Balen and Bos, 2009). Without an established social security system, infertile couples are economically vulnerable in their old age. In certain cultures, children play an important role in the rituals related to their parents’ death. The awareness that they would be deprived of this privilege adds to the infertile couple's sorrows and unease. […] Men are not unaffected and live in fear of being labelled sexually impotent, often resulting in a reluctance to come forward for investigations or treatment and/or releasing their anger on their spouse (Dyer et al., 2004; Gerrits and Shaw, 2010).”
Attitudes in high-income countries towards provision of infertility care in low-income countries have mostly been either dismissive or indifferent as it is argued that scarce healthcare resources should be directed towards restricting population growth (Hammarberg, 2013). Yet, this argument, in addition to being oblivious to the high prevalence and social stigma carried by the disease, neglects the fact that over 50% of these cases of infertility could be easily prevented, and that infertility counts many linkages with other core reproductive healthcare agendas.
In particular, prevention and treatment of infertility can increase the acceptability of family planning policies limiting population growth (Fathalla, 2002; Richards, 2002). Countries like Egypt have been highly successful in controlling population growth by integrating contraception services with infertility care for childless couples (Serour at al., 2008). Infertility helps increase the utilization of family planning by providing a service that men see as of value, especially if it tackles male infertility (Serour, 2017). In a country where primary & secondary infertility rates are as high as 30% (e.g. Nigeria DHIS study) and access to infertility treatment is lacking, delaying a future pregnancy after an early marriage or a previous birth may not be a rational decision. Education about the association of STIs and tubal infertility can also strengthen the effectiveness of public health programs that promote condom use and reduce the spread of HIV and other diseases (Asemota et. al, 2015).
Neglectedness
Ever since the 1990s, the right to have children has been recognized, in high and low resource settings alike (Ombelet, 2009). The International Conference on population and Development (iCpD) in 1994, the World Health Assembly 2004 and the World Summit in 2005, all underlined the importance of the prevention and appropriate treatment of fertility. Infertility care is now encompassed within Strategic Development Goal (SDG) 3.7, which aims for universal access to sexual and reproductive healthcare services. Because the burden of disease of infertility is incorrectly often assumed to be the fault of the woman, as pregnancy and child birth are manifested in the woman, the provision of effective infertility services is also consistent with the objectives of the Global Strategy for Women’s, Children’s and Adolescents health 2016-2030: Survive, Thrive, Transform.
With the mushrooming of high mark-up private clinics throughout Africa, awareness of infertility has risen. Several high-level African officials have spoken of their ambition to increase access (e.g. First Lady Initiatives in Cameroon and Nigeria, parliamentary groups in Uganda and Kenya, etc.), but concrete steps are lacking. National prevention and treatment guidelines are inexistent in most African countries. In addition to improving equity and prevention and treatment outcomes, increasing MoH oversight would put an end to the market failure and patient exploitation which currently occurs in the private sector as a result of major information asymmetry (Zandvoort et al., 2001).
Today, infertility care is probably the most neglected and underestimated health care issue in developing countries (Ombelet et al., 2011, 2022). Unlike for access to contraception and safe abortion, no significant donor has yet decided to prioritize this cause. In some LMICs, patient organizations and local organizations of ObGyns have formed to improve access, such as the Kenya Fertility Society, but they lack the financial means to address the issue at hand. Yet in several developing countries, infertility diagnosis is the leading reason for gynecological consultations (Murage et al., 2011, Okonofua, 1996), but unfairness in access to treatment is glaring, reflecting a large unmet need. For instance, the average cost of an IVF cycle in Uganda is $6,500, which can be afforded by only ~1% of the population. Yet, options exist to tackle this inequity and improve prevention and access to treatment.
The state of infertility treatment in Africa today resembles that of HIV in the early 2000s. There are effective tools to diagnose and treat infertility, but access to these tools is largely limited to developed countries. As with ARVs 15 years ago, the global community assumes that infertility treatment is unaffordable for developing countries. Also mirroring the early HIV treatment landscape, infertility guidelines (where they exist) tend to be highly fragmented and tailored for developed countries. There is a need to create simplified prevention and treatment guidelines for low-resource settings that consolidate around a smaller number of versatile, affordable procedures and products. This, combined with significant price reductions, and the resultant influx of donor funds, is what allowed HIV treatment to scale rapidly and effectively. Moreover, low-cost innovative solutions for infertility are showing promising results in the developed world (e.g. The Walking Egg project), but lack a clear pathway to scale in LMICs. By lowering prices, consolidating treatment protocols, and supporting countries to create effective delivery systems, there is an opportunity for Open Philanthropy help reshape the global conversation, accelerate investment in infertility treatment in LMICs, and help again demonstrate what is achievable.
Tractability
Open Philanthropy could radically transform the state of access to infertility services in Africa through a two-pronged approach; working with LMIC (Ministries of health, NGOs) to strengthen the patient cascade, and working with global suppliers to radically improve the affordability of commodities required.
On the Delivery side, infertility treatment in developing countries should be part of an integrated reproductive care unit, which would include family planning and contraception, as well maternal health. An NGO like the Clinton Heath Access Initiative (CHAI) could partner with Ministries of Health to lead the creation of Comprehensive Reproductive Health strategies that include prevention of and treatment of infertility within the African context. Due to low funding availability in MoH, priority would be given the most cost-neutral or cost-efficient solutions along the patient pathway that would pay for themselves in the medium term:
Drafting and implementing prevention guidelines in LMICs
In Sub-Saharan Africa, infertility is caused by infections in over 50% of women compared to 33% worldwide (Cates et al., 1985; WHO, 1987, 2017 interviews)[2]. Approximately 70% of pelvic infections are caused by STDs while the other 30% are attributable to pregnancy-related sepsis (Ericksen and Brunette, 1996). Similarly, many cases of male factor infertility (responsible for ~16% of infertility cases in Africa) are caused by previous infections of the male genitourinary tract (Kuku and Osegbe, 1989).
To prevent these infections, Open Philanthropy (in partnership with NGOs present locally) could support Ministries of Health to:
- Identify cost-effective mechanisms to raise awareness for these infections and how to prevent them in key populations. There is a need to increase willingness of key populations (e.g. adolescents) to seek treatment for pelvic infections at local Primary Health Care centres, as today the stigma associated with STDs can lead those concerned not to seek care or to prefer traditional medicine. Mainstreaming this communication will be a beneficial horizontal platform to prevent infertility but also other adverse effects of STDs.
- Village Health Workers could be leveraged to promote key knowledge about reproductive physiology, cases of infertility, and dispel myths and stigma about infertility and family planning (Asemota et al, 2015). Education efforts through empowered community health workers and strengthened PHC guidelines would also tackle other MNCH preventable causes such as premature sexual relationship, post-abortal or puerperal sepsis.
- One promising option would be to build on the network of Tradition Birth Attendants embedded daily in the community to popularize these messages, as well recognize infertile couples and encourage them to attend the Family planning clinic.
- Create evidence-based guidelines for all medical staff to know how to diagnose STDs and respond by systematically prescribing the right antibiotics, which are able to prevent infertility at a low cost. One issue encountered today is that the index of suspicion that STDs will cause infertility is low within primary health care centres, leading nurses and doctors to prescribe antibiotics (such as penicillin) which are ineffective in preventing infertility. While a comprehensive diagnosis of STDs such as chlamydia or gonorrhea would require sophisticated lab equipment, nurses could be trained to recognize warning signs and prescribe appropriate medication such as metronidazole, Doxycycline, or tetracycline.[3] This could potentially be complemented by Point of Care urinal tests for the most common STIs - these already exist, and a NGO experienced in market-shaping such CHAI could help decrease their price through negotiations with suppliers.
Streamlining the diagnosis of infertility
In order to implement accessible fertility services in developing countries, the next objective would be to improve the speed to and quality of infertility diagnosis, the pre-condition to identify adapted treatment options. This would include simplifying diagnostic procedures, establishing clear protocols, and enhancing the availability of diagnostic equipment.
In the West, most countries have clear guidelines regarding which steps the primary health care centre must undertake prior to referring patients for specialized care. In the UK for instance (NICE, 2017), the general practitioner must map out the couple’s history of trying to conceive, as well as their medical history focusing on periods, other significant medical problems, any previous pregnancies/miscarriages, medication they are on, smoking habits, etc. He/she must discuss methods to improve chances of conception (e.g. ovulation and timing intercourse, diet, stress reduction), provide emotional support, reinsure them on chances of conception and perform a short examination. Prior to referral to specialist care - after at least one year of trying- , the couple is expected to have done a specific set of tests: semen analysis, blood tests including progesterone on Day 21 of the cycle, FSH and LH if the periods are irregular, rubella and a pelvic ultrasound.[4]
In stark contrast, in Africa, a clear diagnosis pathway starting from the Primary Health Care (PHC) level is lacking, and infertility diagnosis procedures are not routinized in the main hospital system (e.g. in Uganda, even most tertiary clinics are not equipped to do full diagnosis). Today, because of poor referral pathways, the average patient referred for assisted reproductive technology in Kenya only seeks treatment after ten years of experiencing infertility. With the median age of consultation thus greatly increased, the likelihood of positive treatment outcomes drops.[5] Using its expertise in Family Planning, CHAI could further investigate how to leverage Family planning clinics already organized every week at PHC centers to become the first point of contact for infertility journeys. This would require creating clear guidelines for nurses in local hospitals on how to take a menstrual history, review endocrine symptoms and also 1`perform a pertinent physical examination (Asemota et. al). These new guidelines would also establish simple, reinsuring messages to be communicated to the couple and standard referral conditions to higher levels.
For couples referred onwards, one option would be for Ministries of Health to set up “one-stop clinic for the diagnosis of infertility” through one-day clinics. Ombelet (2007, 2016, 2019) suggest the work-up would be very simple - with all procedures performed by a small team of health care providers. After one day of tests - limiting the need for repeated travel to distant clinics -, in over 90% of cases, a diagnosis could be provided to the couple by a doctor/nurse,[6] coupled with clear next steps on the corresponding treatment pathway. Ombelet and Onofre (2019) offer a detailed description of how a full diagnosis, including of tubal abnormalities or semen, can be performed at high quality with reduced need for expensive equipment.
A small pilot of a similar model in Kenya (Murage, 2017) has been found to reduce costs of diagnosis by 2/3 and considerably reduce the Time To Pregnancy. Responding to the unwillingness of many African men to go to get tested in general gynecology wards as they consider them female spaces,[7] this one-stop shop clinics could provide increased confidentiality. These clinics could be fixed or mobile sites, and would have the added benefit of providing the Ministry of Health with refined data on causes of infertility, and total theoretical demand for specific drugs/processes. This information would help to estimate potential volumes for market-shaping negotiations.
Creating patient pathways to improve access to quality infertility treatment
In many developing countries, national guidelines for the treatment of infertility are inappropriate, inexistent or incomplete, and not well disseminated throughout the Health system. The WHO manual for the standardized investigation and diagnosis of the infertile couple has not been updated since 1993, despite continuing scientific progress (WHO, 1993). In general (2022 interviews in LMICs), there is no established structure in MoH for managing infertility services, and no clear way in which the public sector frames what private sector actors should be doing and encourages good practice. Increasing MoH oversight would put an end to the market failure and patient exploitation which currently occurs in the private sector as a result of major information asymmetry (Zandvoort et al., 2001).
NGOs locally present could support MoH to draft and implement new evidence-based guidelines, which would integrate cost-effectiveness considerations. These guidelines would tackle some big issues around quality of care, guiding clinicians to adopt protocols which will minimize complications and selective abortions due to multiple pregnancies through ART, as well as institutionalizing transparency on practices and results in the private and public sectors. In most European countries, clinics must publish their success rates for various interventions, whereas in Africa this is not the case, leading to laggards surviving at the expense of patients.
Treatment protocols should encompass, for instance, low-cost methods of ovarian stimulation and timed coitus. Given only 20% of Nigerian women know how to identify their fertile window (Polis et al., 2017), basic education on reproductive anatomy could likely already improve reproductive outcomes. Other low-cost interventions are equally important such as a listening ear and psychological support for infertile couples, which require health care workers to be trained to exhibit appropriate interpersonal communication for this pathology. To this effect, the use of standardized training-courses for healthcare workers would allow appropriate incorporation of infertility treatment into sexual and reproductive health-care programs, without adding substantially to existing health care costs (PATH, 1997).
Low cost, simple, intrauterine insemination (IUIs) procedure with unwashed centrifuged husband's sperm should be encompassed within these new treatment protocols. These IUIs rest on a simple intervention and qualified nurses could be trained to perform it, instead of specialized doctors, driving down the cost significantly and increasing the volumes which could be managed in country.[8] Today, IUI is not offered in any public facilities in most African countries and private clinics charge ~$400 for the procedure. Through private-public partnerships, Ministries of Health could put in place a system to train nurses in district hospitals to perform this simple procedure at 1/10th of the current price (~$50).
On the higher end of treatment options, in vitro fertilisation (IVF) is the logical treatment for tubal infertility. Because ~ 50% of infertility in Africa is attributable to tubal infertility and ~16% to male infertility (which often also requires IVF), any intervention looking to transform access to fertility services on the continent should include radically changing the game on IVF. Today, most African couples suffer from the lack of real competition between the few private providers of IVF. For instance in Kenya, no public facility offers IVF, and all ~12 IVF centres nationwide benefit from this oligopoly and all charge a high price for an IVF cycle (~$5000). NGOs present locally, experienced in Sexual and Reproductive Health (SRH) could help put in place a public -private partnership, which would feature negotiated price/volume agreements. For instance, the public sector, through its improved diagnosis patient pathway would agree to refer its more numerous diagnosed patients to a chosen private provider, who in exchange would offer a large price reduction on IVF treatment. From interviews with local practitioners, a discount of 2/3rds of the current price seems achievable.[9] Simultaneously, these NGOs could help the MoH better supervise private sector practices and institutionalize transparency on results.
Another promising option to lower the cost of IVF is to avoid the necessity of expensive CO2 based incubators and laminar-flow chamber by using a simple incubation system in tubes. For Teoh and Maheshwari (2014) “the most significant breakthrough in reducing the cost of IVF in recent years is the invention of a simplified culture system, [where] laboratory costs can be reduced by up to 90%. This innovation demonstrates that affordable IVF, especially in developing countries with very basic resources, is within reach in the not-too-distant future”. The Walking Egg project, led by researchers in Belgium has set up a low-cost IVF laboratory for ~$80K, instead of ~$500K- $1M usually required. Preliminary results from the simplified culture system in Belgium indicate good live birth rates and tremendous cost savings, with the total laboratory expenses for this system at only 10%–15% of those used by most IVF laboratories. This would mean that IVF can be performed at ~$1000 per cycle, as opposed to at least ~$5000 today. But W. Ombelet, who leads the project, admits that this initiative does not yet have a clear scale-up path in Africa. Forthcoming articles by Dr Ombelet in the Reproductive BioMedicine Online journal demonstrate parity of live birth outcomes between his low-cost IVF method and the traditional high cost IVF. But the road to scale up will require a coalition of NGOs working Ministries of Health to break the lobbying efforts of expensive private clinics who wish to maintain their mark-ups.[10] Early signals from East African doctors indicate this would be a feasible solution to allow African public hospitals themselves to offer IVF (either in the capital or in medium cities where private providers are absent).[11] NGOs present locally, partnering with the Ministry of Health, can help analyze under which conditions these low cost IVF solutions can best be introduced to Africa. Other LMIC countries’ Ministries of Health which do not yet offer IVF in the public sector, such as Cambodia, have also expressed initial interest in the new, lower cost method.
Global market-shaping
Suppliers of infertility drugs set prices in higher-income markets based on what they think the market can bear, with substantial subsidization of infertility care through insurance. As was the case with HIV anti-retrovirals, many suppliers are largely uninterested in servicing lower-income markets. To this issue, one must add the significant mark-ups imposed in country by private sector actors. For instance, in Uganda, because infertility drugs are not available in the public sector as they are not currently on the Essential Medicines list, private clinics can price them with a 300% mark-up given their monopoly.[12]
Preliminary work in this area suggests that there is the potential to apply the market-shaping approaches that were used by global health actors in reducing the cost of medicines and diagnostics for HIV, family planning, vaccines, and elsewhere to substantially reduce the cost of infertility-related drugs (e.g. Gonal F, Cetrotide, etc.) To give an example, in Kenya the price of Gonal F drugs necessary to prepare for a single intra-uterine insemination (IUI) amounts to ~$300, approximately 20% of the average GNI per capita. To this, one must add procedure costs and post-procedure drugs; in addition, several IUIs are generally necessary to conceive. Costs for IVF are exponentially higher. In order to pay for high-cost IVF treatments, many infertile couples engage in ‘catastrophic expenditure’, defined as any out-of-pocket payment that threatens household survival by exceeding 40% of annual non-food expenditures. A study in South Africa by IVF physician-activist Dyer et al. (2013) found that 22% of infertile couples attending a public-sector IVF clinic had incurred catastrophic expenditure, (Dyer and Patel, 2012; Inhorn, 2015).
Global Health NGOs such as CHAI could partner with Infertility Patient organizations to analyze the global market for infertility drugs, identify opportunities for market-shaping, and implement strategies to secure meaningful price reductions for these drugs. Staff based in Sub-Saharan Africa would gather data about drug availability, cost at importation, and cost to the patient from public and private health centers, public referral hospitals, drug distributors, and national procurement programs. This data would be used to select target products for price reduction and identify appropriate market-shaping opportunities.
For instance, these NGOs would explore tiered pricing agreements with manufacturers of these drugs (Sanofi, Merck, etc.) potentially based on volume commitments; pooled procurement to ensure timely deliveries, fair pricing, and access to quality products; improved public sector procurement rules; and support to generic manufacturers to enter the market through voluntary licensing.
Preliminary theory of change
Final Outcomes
- Unmet need for quality, patient-centered reproductive health care is met
- Family planning in its full sense can occur as per WHO 2013 definition, i.e. African countries “can decide how many children you choose to have and when you want to have them”
Intermediary Outcomes
- Prevalence of STIs causing infertility (e.g. chlamydia, gonorrhea) is decreased
- % of couples experiencing infertility decreased through successful prevention
- % of couples with primary infertility decreased
- % of hospitals that provide infertility treatment services increased
- Average Time To Pregnancy for couples seeking children decreased
- Equity strengthened:
- Gap between the % of couples able to access infertility treatment in developed world vs. developing world decrease
- Gap between the % of couples able to access infertility treatment
- Accessibility increased - # of patients accessing infertility care multiplied by 10
Select Outputs
- Guidelines and national strategy developed, including clear patient pathways
- Referral system established for quicker diagnosis
- PHC nurses trained to provide basic support to couples experiencing infertility
- Reinforced prevention of STDs and other preventable infertility cause
- Average Time to Diagnosis decreased by half and cost of infertility diagnosis reduced by at least 2/3rds
- Cost of key infertility care processes decreased by at least 2/3rds (IUI, IVF)
- ^
Interviews conducted by Soleine Scotney with Pamela Nawaggi, former embryologist in Uganda, Dr W. Ombelet and Dr. A. Murage
- ^
Interviews conducted by Soleine Scotney with Pamela Nawaggi, former embryologist and pharmacologist in a private clinic in Uganda, and Dr A. Murage, ObGyn specialized in ART at Aga Khan Nairobi
- ^
Interview with Antony Ngatia, who used to practice in the field in Kenya and and Dr A. Murage, ObGyn specialized in ART at Aga Khan Nairobi
- ^
Interviews with Gillian Fraser, General Practitioner in the UK
- ^
Interviews with Dr A. Murage, ObGyn at Aga Khan
- ^
Interviews with W. Ombelet
- ^
Interviews with Antony Ngatia and Pamela Nawaggi, who have both practiced in East Africa
- ^
Interviews with Pamela Nawaggi, former embryologist and pharmacist for private clinics in East Africa and Dr A. Murage, ObGyn specialized in ART at Aga Khan Nairobi
- ^
Interview with Dr Alfred Murage, ObGyn at Aga Khan and head of Kenya Fertility Society
- ^
Interview led by Soleine Scotney with W. Ombelet, who leads the Walking Egg project
- ^
Interview led by Soleine Scotney and Dr A. Murage, ObGyn specialized in ART at Aga Khan Nairobi, and who heads the Kenya Fertility Society
- ^
Interviews conducted by Soleine Scotney with Pamela Nawaggi, former embryologist and pharmacologist in a private clinic in Uganda

I was blown away by the quality of this entry and would recommend this as an example to other people who are trying to introduce a new cause area.
It would take more work to figure out if interventions in this area could be cost-effective to the extent of being competitive with the Against Malaria Foundation or Strong Minds, but I am convinced that it is important, neglected and tractable enough to investigate further, which I wouldn't have guessed before reading.