Epistemic status & relevant background: Two of us wrote this post. One of us works in patent law and the other has an undergraduate degree in economics and has covered some of these topics in his coursework and job. However, neither of us have significant experience with any of the topics covered. We’d love feedback/pushback!
This post is for people interested in learning a little about US patent history, patent law, and innovation economics. The goal of the post is to draw attention towards issues within patent law that warrant more attention and away from those that don’t.
We hope posts like this -- that discuss work opportunities that aren't necessarily directly related to 80,000 Hours’ focus areas -- can help supplement 80,000 Hours, enabling a broader range of people to focus on impactful work. We cover three topics within patent policy. Feel free to read all three or just the topics that pique your interest.
We assess the scale, neglectedness, tractability, and overall prioritization of each subtopic from high to low. Our assessment’s not based on quantifiable metrics and are meant to be taken with a major grain of salt. Furthermore, the prioritization is relative to each other (i.e., within patent policy) and isn’t meant as a comparison to other cause areas assessed by 80,000 hours.
Summary/TL;DR:
- Incentivizing Innovating for Developing Countries (e.g. Global Health): Overall Prioritization: Medium-High | Scale: High | Neglectedness: Medium | Tractability: Medium | Alternative innovation finance mechanisms -- such as advanced market commitments (AMCs) and the Health Impact Fund (HIF) -- can help incentivize firms to invest in R&D aimed at helping developing countries’ poorest people. The present patent system, on the other hand, provides limited incentive to create innovations for these people. We think EA may want to pay more attention to innovation funding mechanisms like AMCs and the HIF.
- Patent Trolling: Overall Prioritization: Low | Scale: Low | Neglectedness: Low | Tractability: Low | Recent US legislation and several court rulings have significantly reduced the potential for patent trolling making this issue fairly unimportant moving forward.
- Evergreening: Overall Prioritization: Low | Scale: Low | Neglectedness: Low | Tractability: Low | It does not appear that companies unfairly extend (i.e., evergreen) their patent terms using statutory strategies. However, there is reason to believe companies use other means such as the 30-month stay provision to extend effective market monopolies. We think further research in this area should explore abuse of FDA exclusivity periods and Hatch-Waxman stay periods (discussed further in the Evergreening section).
Global Health Innovation Incentives
Overall Prioritization: Medium-High | Scale: High | Neglectedness: Medium | Tractability: Medium
Summary: Given that the world’s poorest individuals have very little ability to pay for any goods/services, there’s essentially no financial incentive for global firms to innovate in markets that apply only to these people. We focus on how this problem relates specifically to R&D incentives for pharmaceutical companies to illustrate the point, but it can be applied more broadly.
Scale: High
To provide context, 10% of people in the world live on less than $1.90 worth of goods per day, adjusted for cost of living. An additional 15% of people live on less than $3.20 worth of goods per day. Both groups, but particularly the former, have nearly no ability to pay for anything (their consumption consists primarily of food, water, shelter, etc.). (Our world in data)
Easily preventable diseases, which primarily affect these extremely poor people, kill and cause suffering for billions of impoverished individuals living in developing countries (WHO). According to 80,000 hours, these easily preventable diseases lead to the loss of between 200-500 million DALYs per year in the least developed countries plus India.
Neglectedness: Medium
Patents provide minimal incentive to innovate for extremely poor individuals because patents allow firms to recoup R&D costs by charging a premium to their customers. Extremely poor people have very little ability to pay for anything, which means patents don’t incentivize innovation for these individuals. In order to incentivize innovation for these individuals, other mechanisms have been used and proposed.
The primary mechanism currently used to fund this R&D -- instead of patents -- is direct funding of R&D (by governments and charities). Via direct funding, the US federal government contributes roughly $2 billion annually to global health R&D specifically and the rest of the world contributes another $1.5 billion. Additionally, pharmaceutical firms provide pro bono research support and funding. This funding is somewhat unpredictable, not necessarily focused on the most important diseases (at least from a QALY perspective), and doesn’t necessarily reward effective distribution of the vaccine/treatment once developed (it simply funds the research).
Alternative incentive mechanisms, such as advanced market commitments (AMCs), are used infrequently, but have several key advantages over patents and direct funding of R&D. AMCs are binding promises by governments or organizations to financially reward companies for developing vaccines or treatments for particular diseases after the vaccine/treatment is developed. AMCs specify the type of disease that the vaccine/treatment to be developed is for and whether the drug is vaccine or a treatment. The key advantage that AMCs have over traditional, direct R&D funding is that they provide a predictable incentive to R&D firms (and financiers) over a long period of time and only reward firms once they’ve successfully developed and distributed the drugs. The funding tends to be rewarded, at least partially, based on the number of vaccines/treatments distributed creating the incentive to distribute the vaccine/treatment.
The most notable example of an AMC is the 2007 promise by several countries (Canada, Italy, Norway, Russia, the United Kingdom) and the Bill & Melinda Gates Foundation of $1.5 billion to organizations that develop a vaccine for pneumococcal diseases (Wikipedia). This successfully led to the production of two vaccines and an estimated 700,000 lives saved (Kremer).
Another alternative incentive mechanism is the Health Impact Fund (HIF), which promises innovators payouts based on the number of quality adjusted life years (QALYs) these innovators save. This is similar to AMCs in that it awards firms after drugs are developed. However, it's distinct from AMCs because the HIF is agnostic to the target disease and treatment mechanism -- they pay firms based on the number of QALYs saved rather than at some specific price for a specific drug.
We believe people, including the EA community, may neglect innovation incentives for extremely poor people. Despite their potential for significant impact, AMCs have been used seldomly and the HIF has never been used (only proposed). We think it’s possible that, on the margin, funding these R&D incentive mechanisms would provide a better bang for buck than additional funding for distribution of vaccines/treatments (which is what much of the EA global health community focuses on). Supporting this conjecture, Rethink Priorities did research on the cost-effectiveness of vaccine development in 2018 and concluded that funding R&D may be more cost-effective than the distribution-focused charity.
Tractability: Medium
Given that these alternative funding mechanisms (e.g. AMCs and the HIF) have support from influential individuals and countries, we believe it’s feasible they receive future financial support and lead to the successful development of impactful vaccines/treatments.
Influential governing officials, academics, scientists, and organization leaders have promoted these mechanisms.
AMCs: Michael Kremer (Nobel prize winner) developed the concept in the first place. Bill Gates and several countries have supported them financially. Tom Kalil (former Deputy Director for Policy in the White House Office of Science and Technology Policy) discusses increasing funding alternative funding mechanisms (including AMCs) in an 80,000 Hours podcast.
HIF: Peter Singer has promoted the Health Impact Fund (including in a New Yorker article earlier this year). Economist Kenneth Arrow (Nobel prize winner), economist Amartya Sen (Nobel prize winner), Judith Whitworth (former Chair of the WHO Global Advisory Committee on Health Research), and Carl Nathan (Chairman of the Department of Microbiology and Immunology at Cornell University) have also promoted the Health Impact Fund and serve on its advisory board (HIF advisory board).
Patent Trolls
Overall Prioritization: Low | Scale: Low | Neglectedness: Low | Tractability: Low
Summary: A broad range of firms -- referred to as non-practicing entities (NPEs) -- (1) own patents, (2) do NOT produce products themselves, and (3) sue companies for infringing on/violating their patent rights. Patent trolls are a specific type of NPE that use questionable legal tactics to file these infringement suits. The types of suits can be grouped into two buckets:
- Obvious/Vague Patent Lawsuits: A patent troll purposefully purchases poorly-worded, obvious, and/or vague patents and then sues firms for infringement even though the defendants are not intentionally violating patents. Obvious/vague patents aren’t supposed to exist in the first place. However, since the US Patent and Trademark Office (USPTO) has limited resources, some obvious/vague patents slip through the cracks during approval processes. Patent trolls focus on finding patents that have slipped through the cracks and then sue entities that “technically” are violating the patent.
- Frivolous Lawsuits: Patent trolls also file lawsuits on small entities (e.g. start-ups) anticipating that the small entity cannot afford to go through an entire legal case and, thus, instead, the small entity will opt to pay to avoid the case (i.e., settle) even though the small entity wasn’t doing anything illegal.
Our understanding is that there’s essentially a (non-partisan and expert) consensus that patent trolls harm innovation because they do not protect specific innovations, make firms hesitant to use technology (out of fear that they’re unwittingly violating an obvious/vague patent), and cost otherwise law-abiding firms millions of dollars (potentially billions) in legal fees each year. However, many legislative and judicial steps have been taken since 2013 to address patent trolling in the US, making the issue -- in our view -- presently low in scale, neglectedness, and tractability.
Scale: Low
Patent trolling was a more significant issue in the US before 2013. Since then, federal legislation and several court cases have curbed the ability of NPEs to “troll”. Correspondingly, costs imposed by patent trolls seem to have reduced (however, we couldn’t find great data on the topic and there’s some disagreement regarding how to best measure patent troll costs; more info here: Oxford handbook section I., paragraph 13-14, Stanford paper). The significant legal action against patent trolling followed by the potential reduction in costs imposed by trolls suggests that this issue has significantly reduced in scale.
Neglectedness: Low
In the early 2010s, the US Congress and several courts took steps that reduced the likelihood of patent trolling by reducing the number of obvious/vague patents and disincentivizing frivolous lawsuits:
- American Invents Act (“AIA”) (2011) – The AIA reduced the likelihood of obvious/vague patents from existing by making it easier and cheaper for organizations (e.g. non-profit watchdogs, corporations) to challenge patents (i.e., via post grant proceeding). These measures reduce the number of obvious/vague patents that exist, which, in turn, reduces the ability of NPEs to engage in lawsuits using obvious/vague patents.
- Several court cases reduced the likelihood of frivolous lawsuits (2016-2020) – For decades, federal and state laws stated that courts could compensate defendants for legal fees during frivolous lawsuits by requiring the plaintiff to pay the defendant’s legal fees. However, it required several court rulings (e.g. Mossberg & Sons Inc. v. Timney Triggers LLC and Gust, Inc., v. Alphacap Ventures, LLC, et al.) to help send a meaningful signal to patent trolls that courts would punish patent trolls for frivolous lawsuits. This removed the incentive for patent trolls to file frivolous lawsuits.
- TC Heartland LLC v. Kraft Foods Group Brands LLC (2017) Prevented Plaintiffs from Picking Favorable Judges – The Supreme Court ruled that patent infringement cases must be heard within the district that the defendant is incorporated, making it difficult for patent trolls to file in favorable districts. This was relevant because the US District Court for the Eastern District of Texas had a judge that was extremely favorable to plaintiffs (in fact, plaintiffs sought him out so much that he saw 25% of infringement cases filed by non-practicing entities in the United States in recent years preceding 2017).
Theoretically, the AIA and court cases, such as those listed above, significantly reduce the ability of NPEs to engage in patent trolling. Early empirical evidence suggests these measures have been effective. This leads experts, such as Paul Gugliuzza, writing in a 2017 Oxford handbook (research review), to argue that further legislative reform regarding patent trolling in the US would not be useful at this point. NBER is researching this topic further, so this could change.
Tractability: Low
The Overton window in the early 2010s has seemingly passed. This makes sense given that Congress and courts took major steps to reduce the likelihood of patent trolling. If patent trolling is observed again, perhaps, a new Overton window will open up.
Evergreening
Overall Prioritization: Low | Scale: Low | Neglectedness: Low | Tractability: Low
Under current US law, patents provide a 20 year term (measured from the filing date of the earliest U.S. non-provisional or international application for which priority is claimed). However, many argue that sophisticated firms, especially in the pharmaceutical sector, utilize a web of legal and regulatory processes to unfairly extend the length of their patent monopolies beyond this 20 year term. This practice is generally referred to as “evergreening.”
Evergreening mechanisms differ by industry, with the majority of scrutiny falling on the pharmaceutical sector. A summary of commonly cited statutory evergreening tactics is produced below:
Mechanisms relating to any invention:
- Continuing applications
- Line extensions
- Patent term adjustment (PTA) based on USPTO delay
Mechanisms relating to drugs (or medical devices, etc.):
- Patent term extension (PTE) based on FDA delays
- FDA exclusivity periods
- Hatch-Waxman stay periods related to litigation
Additionally, a litany of non-statutory strategies (e.g., (i) inter-party payments for delay in market entrance: “reverse-settlements,” (ii) brand migration, (iii) brand name generics, and many others) are often employed to extend effective market monopolies. However, these are market-based and beyond the scope of this discussion.
Scale: Low
Based on our review, there does not appear to be significant evidence of “unfair” evergreening based on statutory strategies (e.g., line extensions, PTA, etc.). It appears much ado is given to particularly aggressive strategies that, in the end, are more bark than bite (see for example Allergan’s attempt to flout the PTAB by claiming Native American sovereignty which was struck down by the Court of Appeals). In general the narrative appears to be that creative lawyers and business types will always have incentives to try to make more money by extending effective monopolies but that other market participants (in combination with the courts and the USPTO) act as a check on the excesses of these attempts. We find that claims of unfair (statutory-based) evergreening rely on “attempts” at evergreening rather than “successes,” and that, in general, the system appears to function as designed. This notwithstanding, we welcome specific examples for further analysis. (Epistemic status: 3 years working as a patent agent and ~20 hours of additional outside research.)
Below is an overview of the most common evergreening tactics we saw discussed in the literature.
Continuing Applications
Continuing applications such as a continuation, divisional, and/or continuation-in-part are sometimes cited as a mechanism for extending patent term. However, continuing applications share a priority date with the application they stem from (often called the “parent application”), and therefore have identical patent term as the parent application (without taking into account PTA based on USPTO delay, discussed below). Moreover, in practice it is not uncommon for a continuing application to have a terminal disclaimer filed, which ties the expiration date of the continuing application to the expiration date of the parent application, thereby negating any possibility of extending patent term via PTA. In short, we don’t see continuing applications as contributing to unfair evergreening.
Line Extensions
Line extensions refer to filing multiple applications, each having different patent terms, on a single product (or closely related product line, etc.). For example, a firm may file a first application on Tylenol per se (e.g., the chemical formulation of Tylenol) having a first patent term and may subsequently file a second (non-continuing) application on a formulation of Tylenol (e.g., the chemical formulation of Tylenol mixed with other ingredients, Tylenol PM for example) having a second patent term that extends beyond the first patent term. As shown below, at the expiration of the first patent, Tylenol per se becomes available while Tylenol PM remains on patent.
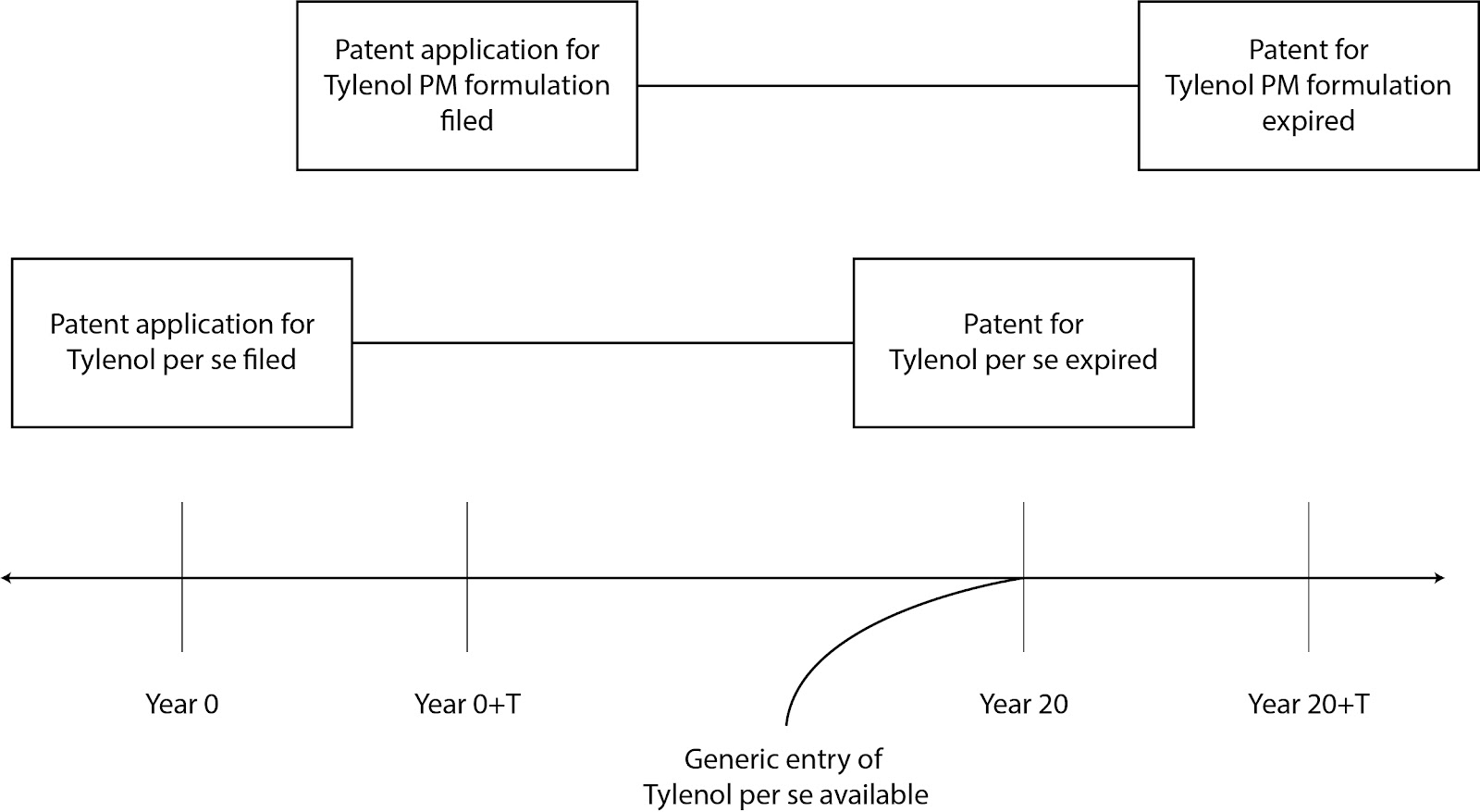
Some argue that firms abuse line extensions by patenting trivial (e.g., “obvious”) modifications of an original product thereby defacto achieving longer terms on the original product. For example, “Overpatented, Overpriced: How Excessive Pharmaceutical Patenting is Extending Monopolies and Driving up Drug Prices” from i-mak.org reports Humira patents as “blocking competition for 39 years” (e.g., far in excess of the 20 year term of a patent).

As another example, “Evergreening: A deceptive device in patent rights” describes Pfizer’s attempt to patent obvious formulations of Viagra.
However, evergreening via obvious line extensions does not appear to succeed in practice (we were not able to identify any case from the literature in which an obvious extension of a patent was used to extend the term of the patent). We suspect that the majority of attempted obvious line extensions are struck down at the USPTO level (via § 103 rejections) while remaining attempts die in litigation (although data on this is hard to find and this is admittedly speculative). However, we are confident in saying that if obvious line extensions are used to evergreen, it is only rarely - and that claims of rampant “obvious” line extensions overstate their case.
Moreover, it is worth noting that having a patent is not the same as enforcing a patent and that attempts to enforce a patent (e.g., stop a competitor from practicing your invention) will almost certainly open a patent to invalidation. For example, in the Viagra case cited above, a November 2000 decision struck down Pfizer’s patent for insufficient utility (see MPEP § 2107) as a result of a challenge from Lily ICOS.
And what about Humira’s “39 years blocking competition”? The paper’s methodology states:
“...we identified the a) earliest filing date, and b) latest potential expiration date of all patents on each drug, according to the patent landscape analysis conducted. For all drugs, the latest expiration date was on an active patent application that was not yet an issued/granted patent. As such, the later time point represents a potential time point that could be in place if/when the patent is granted. This total span of time – from earliest filing to latest potential expiration– was calculated and used as an indication of drugmakers’ intent to extend their monopoly period and thwart generic competition.”
This methodology captures the patent term of formulations, delivery methods, packaging, etc. that do not in and of themselves extend patent protection for Humira per se (e.g., the chemical formulation of Humira). Therefore, “39 years blocking competition” for Humira seems misleading at best.
Patent term adjustment (PTA) based on USPTO delay
Patent term adjustment compensates a patent applicant for delays that occur during patent prosecution. For example, an applicant may receive additional patent term if the USPTO takes longer than 14 months to examine a patent application after filing or longer than 4 months to issue a patent after an applicant pays an issuance fee. Some point to significant PTA as an example of unfair evergreening because it appears as though the applicant is arbitrarily gaining patent term. However, PTA exists because delays at the USPTO may be significant. For example, in 2019 the average time-to-examination from filing in the 3700 technology center was 19.1 months. That means that if you filed a patent application today, you could expect the USPTO to pick that patent application up in over a year and a half from now (not to mention the delays introduced in prosecution). It is worth noting however that delays caused by an applicant do not garner PTA. Therefore, applicants’ cannot purposely introduce delays to abuse PTA. Based on our review PTA does not contribute to unfair evergreening.
Patent term extensions (PTE) based on FDA delays
Patent term extension compensates a patent application for delays that occur during the FDA’s review process. For example, an applicant may receive additional patent term for each day of the FDA’s regulatory review period. Similar to PTA, some point to PTE as an example of unfair evergreening - although again PTE exists because FDA delays may be significant. In short, we are not aware of any abuses of this system and conclude that PTE does not contribute to unfair evergreening.
FDA exclusivity periods
FDA exclusivity periods, while separate from patents, overlap in some respects. FDA exclusivity periods stem from the Drug Price Competition and Patent Term Restoration Act of 1984 (e.g., “Hatch-Waxman”) and relate to intentional delays introduced by the FDA for approval of competitor drugs. For example, an applicant may receive a 5 year new chemical entity (NCE) exclusivity period in response to an FDA approval of a drug having a new active ingredient (e.g., a new chemical moiety) during which time generics may not enter the market. There are many types of exclusivity periods ranging from 7 years (Orphan Drug Exclusivity) to 180 days (Competitive Generic Therapy) in length. Patents have longer terms than FDA exclusivity periods and therefore generally set the upper bound of protection. In some cases FDA exclusivity may extend patent term (see Pediatric Exclusivity), however we are not aware of this process being abused. However, there is some evidence that brand name drug manufacturers collude with early-entrant generic manufacturers to nullify the 180 day competitive generic therapy exclusivity period. We see this as an area for further investigation.
Hatch-Waxman stay periods related to litigation
Similar to FDA exclusivity periods, Hatch-Waxman stay periods relate to intentional delays introduced by the FDA for approval of competitor drugs. Specifically, the Hatch-Waxman stay period grants a 30-month stay of regulatory approval in response to a patent owner filing an infringement suit against a generic entrant. There is some evidence that firms utilize the 30-month stay to unfairly maintain market monopoly (e.g., regardless of the merits of their infringement suit). In fact, according to a 2002 study by the FTC, approximately 72% of brand name drug manufacturers utilized the 30-month stay despite generics prevailing in 75% of those cases that were resolved. This suggests that the 30-month stay may be abused to promote evergreening. We recommend further investigation.
Neglectedness: Medium
While we believe that FDA exclusivity periods and Hatch-Waxman stay periods may be used to unfairly maintain market monopolies (e.g., evergreen, although not strictly in a patent sense), it does appear some attention has been paid to these issues. For example, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 requires notice of settlement between brand and generic firms to the FTC and Department of Justice, thereby inhibiting collusion related to FDA exclusivity periods. Moreover, the FTC has pursued a number of antitrust enforcement actions in the pharmaceutical sector. This notwithstanding, we believe outsized attention is paid to low impact “evergreening” activities such as line extensions relative to collusion-based abuses of FDA exclusivity periods and Hatch-Waxman stay periods.
Tractability: Medium
Progress can be made via legislation or enforcement actions. On the legislative side see: Preserve Access to Affordable Generics Act. More straightforward than legislation is having the FTC file enforcement actions. This could plausibly be achieved through a commission mandate.
