Summary
The article discusses the increasing recognition of the role of the environment, particularly climate change, in the emergence and spread of infectious diseases caused by fungi. It highlights the rise in emerging fungal infections, their threats to food security, and the challenges they pose to healthcare systems due to limited antifungal therapies. The text emphasizes the ecological and economic impacts of fungal diseases on crops, forests, and urban ecosystems. It also explores the potential for climate change to drive fungal evolution and the need to understand the relationship between ecology and medical mycology. The article calls for a comprehensive approach to mitigate the impacts of climate change on fungal infections and enhance our understanding of these complex dynamics.
The role of the environment in emerging and reemerging infectious diseases is increasingly recognized. Climate change, defined by the United Nations Framework Convention on Climate Change is “a change of climate which is attributed directly or indirectly to human activity that alters the composition of the global atmosphere and which is in addition to the natural climate variability observed over comparable time periods” may create environmental pressures that result in new diseases caused by fungi. The incidence and spread of emerging fungal infections have increased in the last decade. Emerging infectious diseases (EIDs) caused by fungi are increasingly recognized as presenting a worldwide threat to food security. For the vast majority of fungal species, the capacity to grow at elevated temperatures limits their ability to infect and establish in mammals. However, fungi can be trained to evolve thermotolerance, and gradual adaptation to increasing temperature caused by climate change could lead to an increase of organisms that can cause disease
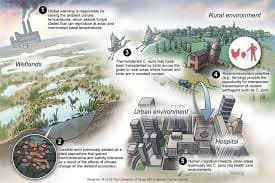
Image Credits: Science Media Centre
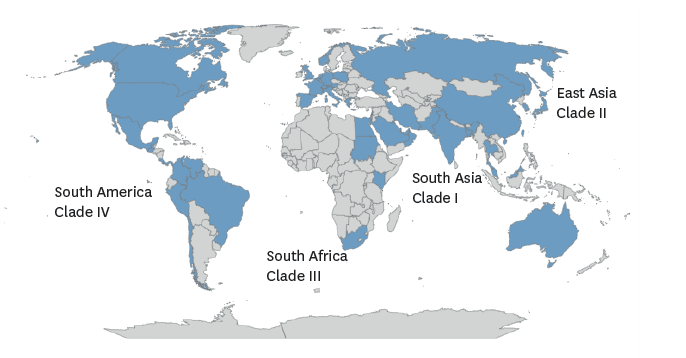
It proposed that Candida auris “is the first example of a new pathogenic fungus emerging from human-induced global warming,” and as temperatures continue to rise in the 21st century, it may be the first of other fungal lineages to become more thermally tolerant and capable of infecting humans. So far, C. auris is a multidrug-resistant (MDR) fungus, Candida auris has spread to almost all the continents of the world. The severity of these infections is aggravated in individuals with weakened immune systems enabling opportunistic fungal pathogens to be disease agents, causing serious and often fatal infections
Countries with reported Candida auris cases and clade identification by region and widespread distribution of C. auris Source
Fungi research is neglected but important:
Fungi infections majorly affect people with weakened immune systems in hospitals, between 30 and 60 percent of people infected with the fungus die, according to US Centers for Disease Control (CDC) estimates. While viral and bacterial diseases receive the most attention as the potential cause of plagues and pandemics (The rise of COVID-19 makes it even worse), fungi can arguably pose equal or even greater threats: There are no vaccines available yet for fungal pathogens, the arsenal of antifungal agents is extremely limited, and fungi can live saprotrophically, producing large quantities of infectious spores and do not require host-to-host contact to establish infection.
Indeed, fungi seem to be uniquely capable of causing complete host extinction . Pathogenic fungi have not been widely recognized as posing significant hazards to animal health. This perspective is fast shifting as a result of the recent occurrence of several high-profile wildlife reductions caused by the advent of previously identified fungus. In 2007 ascomycete fungus Geomyces destructans the causative agent of white-nose syndrome caused that caused Bat numbers across affected sites to declined by over 70% and analyses have shown that at least one affected species, the little brown bat Myotis lucifugus, has a greater than 99% chance of becoming locally extinct within the next 16 years due to this fungus. Another fungal infection has caused the greatest disease-driven loss of biodiversity ever documented. The skin-infecting amphibian fungus Batrachochytrium dendrobatidis was discovered in 1997. As a consequence of the infection, some areas of central America have lost over 40% of their amphibian species, a loss that has resulted in measurable ecosystem-level changes.
Fungal disease impacts crops visibly, and there are obvious observable economic effects linked with die-off in forest and urban ecosystems. Invasive tree diseases have caused the loss of approximately 100 million elm trees in the United Kingdom and the United States and 3.5 billion chestnut trees have succumbed to chestnut blight in the United States. In natural settings, valuations have recently estimated the losses to US agriculture that are the result of declines in bat populations at more than US$3.7 billion per year
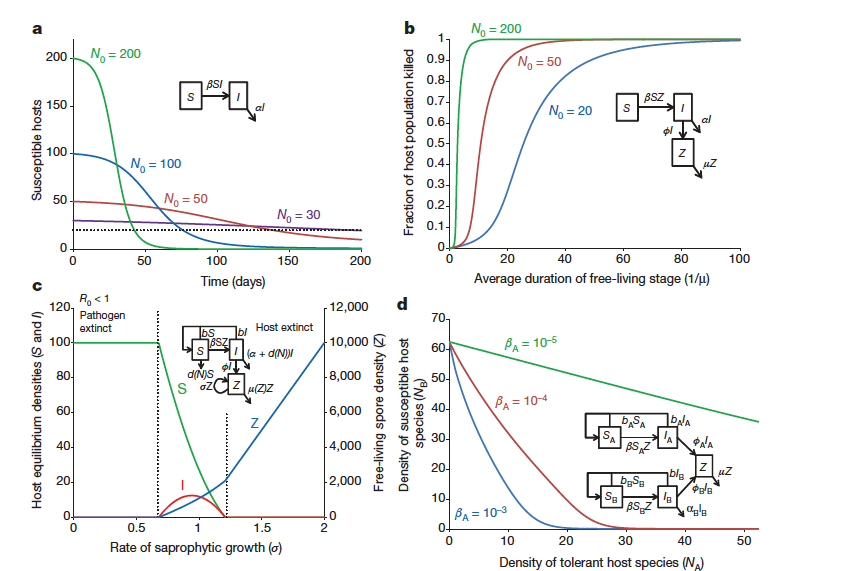
Fungal disease dynamics leading to host extinction Source
a, The presence of a threshold host population size for disease persistence does no prevent host extinction during a disease outbreak, especially in cases in which a lethal pathogen invades a large host population. In a large host population
transmission is rapid and all hosts can become infected before the host population is suppressed below the threshold (pathogen transmission rate,
b. 50.001 per individual per day; disease induced-death rate, a50.02 per day; simulations start with one infected individual and N0 susceptible individuals).
b, Long-lived infectious stages can increase the potential for host extinction. The fraction of hosts killed in a disease outbreak is shown as a function of the duration of the free-living infectious spore stage (pathogen transmission rate, b5531026; disease-induced death rate, a50.02; rate of release of spores from infected hosts, w510; outbreaks initiated with one infected host in a population of N0 susceptible individuals).
C. Saprophytic growth: equilibrium densities of susceptible and infected hosts and free-living spores as a function of the rate of saprophytic growth, s. With no (or low levels of) saprophytic growth, the basic reproductive rate of the pathogen (R0) is less than 1, the pathogen cannot invade the system and the host persists at its disease-free equilibrium density. Intermediate levels of saprophytic growth allow the pathogen to invade and persist with the host. High levels of saprophytic growth lead to extinction of the host, and the pathogen persists in the absence of the host (host intrinsic rate of increase, r5b2d050.01; density-independent host death rate, d05131023; strength of density dependence in host death rate, d15131024; pathogen transmission rate, b5131025; disease-induced death rate, a50.02; rate of release of spores from infected hosts, w510, densityindependent spore mortality rate, m051; strength of density-dependence in spore mortality rate, m15131024).
d. The presence of a tolerant host species (host species A), which can become infected and shed infectious spores can lead to the extinction of a susceptible host species (host species B). Species A does not die because of the disease, but species B has a disease-induced per-capita mortality rate of aB. Species B is driven extinct at high densities of species A. For all parameters, subscriptsAor B indicate the host species. Host intrinsic rates of increase, rA5rB50.01; density-independent host death rates, dA05dB051 x1023; host birth rates, bA5bB5rA1dA0; density-independent death rate for species, B dB151 x10-4; rate of release of spores from infected hosts, wA5wB510; aB50.05; spore mortality rate, m51. The density of tolerant species NA was varied by varying dA1 (the strength of
density-dependence in host species A), such that NA5rA/dA1.
The lack of effective antifungal therapies makes treatment of these infections extremely difficult. The rise in the incidence of pathogenic fungal infections has constituted a fundamental change in the epidemiology of invasive fungal diseases, especially in immunocompromised individuals, such as those with HIV, cancer, or subjected to transplants. Environmental fungi are globally ubiquitous and human populations are frequently exposed to spores and other fungal propagules. Systemic fungal infections account for more than 1.6 million people annually across the globe and are estimated to cost the US healthcare system $7.2 billion dollars per year.
Climate change drives fungal evolution
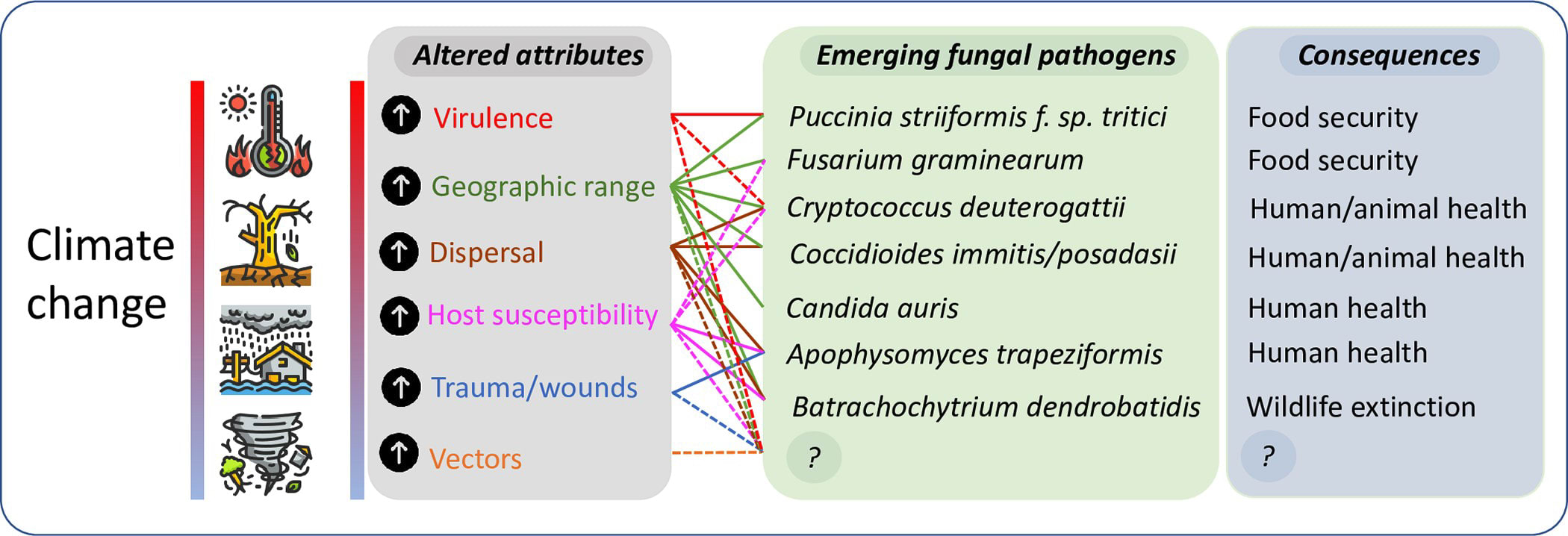
Climatic changes are a result of biotic and abiotic activities, these activities are expected to increase the mean global temperature by 2 to 5°C in the next decades, leading to the warmest period in the past 40 million years. The fungi kingdom is made up of fungi that could grow at a wide range of temperatures, some growing between 12-30oC, while others could grow at 65oC Global warming could have a significant effect on fungal populations by leading the distribution of both heat-tolerant and susceptible species, thereby favoring those that are more thermotolerant, and by creating conditions for more environmental fungi to spread and enter into closer contact with human populations. This effect is expected not only to widen the geographic area of pathogenic (i.e., intrinsically heat-tolerant) species but also to facilitate the close interaction of environmental fungi with human populations, leading to the emergence of novel fungal pathogens.
The effect of climate change on the emergence of fungal pathogens Source
In contrast to native fungi in their centre of origin, pathogen epidemics and outbreaks are studied
in great detail, and this has distorted our understanding of genetic diversity in natural populations
of fungi. The environment has a significant impact on the spread of infections and also has a lot to say about how fungi interact and adapt for survival. Climate has a significant environmental impact on a location's ecosystem, and ecosystems respond to changing climate in complex ways we do not yet understand. It can also cause a convergence of factors that can drive the emergence of new pathogens.
Conclusion
Climate change poses a significant threat to the global temperature and the impacts of climate change are predicted to worsen if carbon emissions are not drastically reduced. One consequence of climate change is the increased influence of fungi on crops and native plants, as their ecological range expands and they disperse over long distances, leading to new risks. The potential for novel fungal infections in humans and other mammals is less predictable, highlighting the need to explore the relationship between and medical mycology.
Previously, the study of these fields has been isolated from each other. It is now understood that many fungal pathogens relevant to human health, such as Candida albicans, can be found in the environment due to environmental pressures that drive the evolution of new traits, including virulence and resistance to antifungal treatments. Climate change creates a combination of factors that can contribute to the emergence of new pathogens. In the medical field, it may become necessary to consider the possibility of infections caused by unfamiliar or uncommon species, or strains within species that were previously thought to be unlikely to affect mammals or breach the mammalian thermal barrier. Similar to how the HIV-AIDS epidemic and immunosuppression reshaped our understanding of fungal pathogens, climate change has the potential to do the same.