Authors: Sid Sharma, Clare Donaldson, Michael Plant[1] [2]
This is a cross-post from the Happier Lives Institute
Executive summary
We all expect to experience some pain in our lives. For most of us, especially those in high-income countries, these experiences will be mild, bearable, and short. Others are not so fortunate. Millions suffer excruciating pain. Millions more suffer moderate or severe pain. They suffer despite the fact that cheap and effective treatments exist.
This report briefly discusses the measurement of pain then explores three major causes of pain and what might be done to relieve them. The findings are relevant for individuals and organisations considering if and how to put their resources (i.e. their money and/or time) towards this global problem.
Problem 1: Terminal conditions requiring access to opioids
First, we consider pain from terminal conditions such as advanced cancer and HIV.[3] These cause intense suffering if untreated. If carefully treated with opioids[4], 95% of those with severe or moderate-pain report mild or no pain and quality of life greatly improves.
In high-income countries (HICs), some 98% of this need for opioids is met; this figure falls to 5% in low and middle-income countries (LMICs). Cost is not the main barrier, however; a course of 90 days’ opioids is as little as $8.
This ‘access abyss’ stems from an overestimate of the risks from opioids and a focus on life extending treatment (Knaul et al., 2017a). The specific issues include:
- restrictive regulation designed to stop the misuse of opioids;
- a lack of training and awareness among health professionals; and
- fragile supply chains.
We discuss how organisations taking a multipronged approach have succeeded in improving access in India and Uganda.
Problem 2: Headache disorders
Second, we examine two headache disorders: migraines and cluster headaches. The former are common, affecting around one in six people, and sometimes debilitating. They impose a burden of disease roughly comparable with malaria or depression.[5] The latter are agonising and experienced by 0.1-0.2% of the global population.
Inexpensive relief exists for migraines, such as aspirin, triptans, and propranolol. The main barriers appear to be:
- patients do not seek help; and
- if they do, they are often misdiagnosed by doctors who generally receive little training about these conditions.
Education campaigns for physicians, patients, and policymakers seem to be the order of the day.
Temporary relief exists for cluster headaches in oxygen and triptans, while preventative agents can be used to reduce the frequency of headaches. Like migraines, patients are often misdiagnosed and receive the wrong treatment. Yet, even with access to the right treatments, many people with cluster headaches endure a great deal of suffering. In addition to education campaigns, possible solutions include increasing access to proven treatments for people in low-resource settings or expediting the development of, and access to, novel therapies, such as psychedelics.
Problem 3: Low back pain
The third and final issue assessed is low back pain. This is the leading cause of years lived with disability globally.
Little is known about what causes low back pain or how to treat it. Efforts to increase access to existing treatments are therefore unpromising. However, there may be a high value of information in medical research into the nature of and intervention for this problem.
Conclusions and limitations
Our report closes with suggestions for further research and identifies some promising career and donation opportunities. We have not been able to thoroughly evaluate the best ways to make progress within this problem area; so these suggestions should be considered highly preliminary. However, donating to the Walther Centre for Global Palliative Care and Supportive Oncology may be an effective way to improve opioid policy and availability in LMICs. Regarding careers, working as a grantmaker at a foundation that funds medical research and impact evaluations is one stand-out option.
We have not attempted to compare interventions in this area against other options for doing the most good.[6] Nevertheless, we hope this document, which draws together and pushes forward our understanding of the problem, can act as a springboard for further investigation, either by ourselves or others. Our conclusion is optimistic: although pain causes substantial suffering for millions of people, solutions are closer to hand than we might have expected.
1. Introduction
This problem area report investigates one of the most obvious and arresting problems of all: pain. The purpose of this report is to take a broad but fairly shallow look at the issue with a view to working out what can be done about it. Its purpose is not to reach considered conclusions about what the best actions in this area are; that is a matter for further work. This research is part of the broader mission of the Happier Lives Institute to find and share the most cost-effective methods for increasing global well-being.
We expect this report to be most relevant to:
- individuals looking for high-impact careers related to pain or global health;
- philanthropists and grant-makers looking for new, high-impact donation opportunities;
- researchers looking for important problems to work on; and
- campaigners and policymakers who could use their platform to advocate for pain-related issues.
After a brief review of pain-related problems, we chose to focus on three sub-areas in this report for the following reasons:
- Terminal conditions requiring access to opioids in low and middle-income countries (LMICs) – due to the large inequity and unmet need.
- Headache disorders – due to the large burden of disease (migraines) and severe intensity of pain (cluster headaches).
- Low back pain – due to the large burden of disease.
We considered investigating other pain-related problems, but decided not to:
- Kidney stones and trigeminal neuralgia – although very painful conditions, they are not as painful as cluster headaches or as common as advanced cancer.
- Neck pain and pelvic pain – although the burden is large, it is at least three times smaller than the burden of low back pain.
This report expands the nascent work to date on how individuals and organisations might best try to reduce the global burden of pain.[7] It proceeds as follows. Section two discusses the measurement of pain and the challenges of comparing the intensity and overall scale of different sources of pain. Sections three to five take one major cause of pain in turn: terminal conditions requiring access to opioids, headache disorders, and lower back pain, respectively. Section six outlines further research and section seven makes some suggestions for what can be done to reduce worldwide pain. Section seven makes concluding remarks.
We consider this report to be shallow in depth. Approximately 150 hours was spent on the review. The number of conditions we studied that fall within the category of pain is limited and we do not quantitatively analyse the cost-effectiveness of interventions.
2. Characteristics and scale of the three conditions
In the following subsections, we explore some of the metrics that characterise pain for the conditions we investigate in this report. We then make very rough estimates of the global scale of each. Our aims are threefold. Firstly, to provide an overview of the relative intensity of the pain of these conditions. Secondly, to explore the quantitative information available that could enable us to estimate the effect on well-being from pain, both to an individual and globally - although we do not make any firm conclusions. Finally, to indicate the scale of these problems in terms of Disability-Adjusted Life Years (DALYs), which are readily comparable to other global health burdens. We would prefer to use Well-being-Adjusted Life Years (WELLBYs, Frijters et al., 2020)[8], but estimating these was out of the scope of this report.
2.1 Measuring the intensity of pain
First, we consider the intensity of pain - how it feels as it is being experienced. Pain is commonly assessed in a healthcare setting using a numerical rating scale. Patients are often asked to verbally rate their pain between 0 and 10, or given a visual analogue scale to grade the intensity of their pain on a 100mm line with reference points (Karcioglu et al., 2018). More complex scales delineate a detailed description, such as the McGill Pain Questionnaire and the Brief Pain Inventory (Melzack, 1975; Cleeland & Ryan, 1994).
As far as we know, very few studies directly compare intensities of pain amongst various conditions.[9] To indicate to the reader the relative badness of different conditions[10], we approximate the intensity of pain on a 0-10 pain scale. We take studies using a variety of pain scales, assign intervals to categorical scales where necessary, rescale to 0-10, and average the results. We also provide estimates, constructed in the same manner, of the average resulting pain level when recommended treatments are administered. Results are displayed in Figure 1 and the full details given in [Appendix 1]. We intend for these rough results to be interpreted only as an illustration of the relative intensity of pain for these conditions.[11]
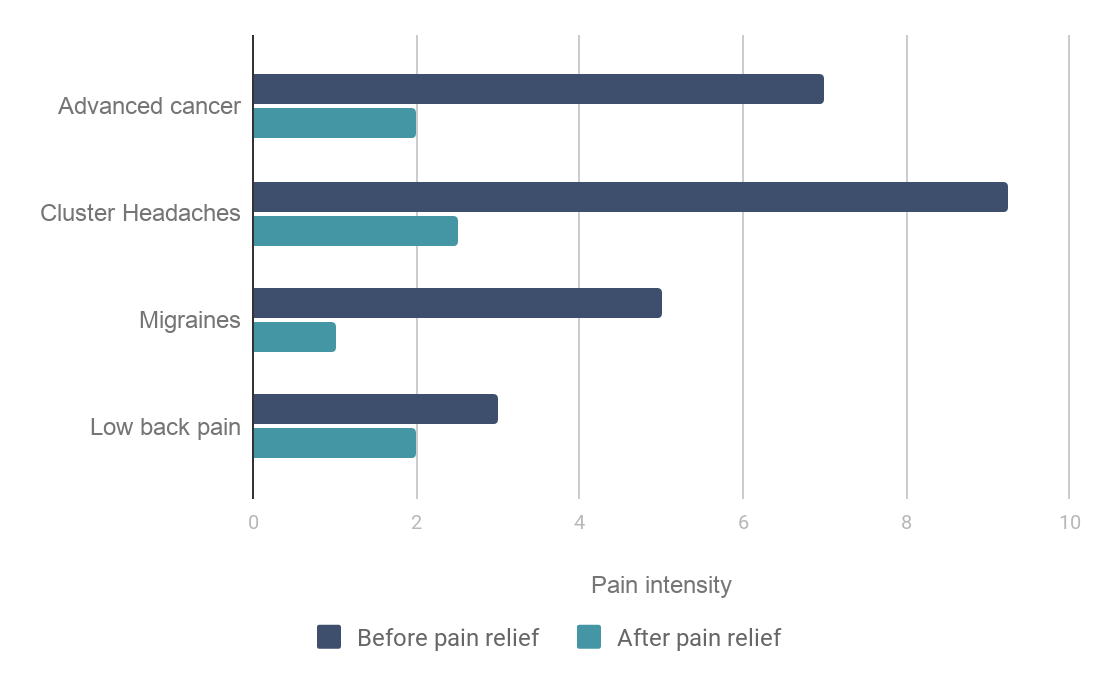
Figure 1: Estimated pain intensities. Estimates were derived from available literature that assessed pain intensities prior to treatment using pain scales. Where necessary, categorical scales were assigned to intervals and all estimates were re-scaled to a 0-10 scale. Treatment effects were estimated based on available systematic reviews and randomized control trials that utilised pain scales. Full details are given in [Appendix 1].
2.2 Pain and subjective well-being
Of course, the effect of pain on well-being does not only depend on its intensity but also on its frequency, duration, regularity, associated symptoms, and impact on functioning and mental health (Ferrell, 1995).
Ideally, we would like to know the impact of pain on measures of subjective well-being - self-reports of happiness and life satisfaction - so that we can compare the impact of pain to other life events and circumstances.[12] Subjective well-being may capture more information about the effect of pain on someone’s overall quality of life than health-focussed metrics.
A report by the Happiness Research Institute (HRI) studied the effect of being in mild, moderate, or severe pain on life satisfaction from a European panel dataset of over-45s (HRI, 2020). HRI estimates that a change from experiencing mild to severe pain (on a 4-point scale: none, mild, moderate, severe pain) is associated with a drop in the average life satisfaction of respondents of 1 point (on a 0-10 scale), whilst controlling for other variables. For comparison, the same report estimates that average life satisfaction is ~0.6 points lower for those in debt or who are unemployed, and ~1.3 points lower for people with depression (HRI, 2020, p50). We expect that this research does not capture the severity of pain from cluster headaches or from terminal illnesses without pain relief, given the rarity of these circumstances in the studied European population.
McNamee and Mendolia (2014), using an Australian panel dataset and a fixed effects model (accounting for individual unobserved characteristics), estimate that being in chronic pain results in a decrease in life satisfaction of 0.1 points (on a 0-10 scale). They do not describe the typical severity of the chronic pain of the sample. Migraines are related to a -0.11 to -0.25 drop in life satisfaction (Groot & van den Brink, 2004). In a Chinese sample, people with cluster headaches report the quality of their life on a 1-5 scale (with 1 as bad and 5 as good) as 2.38 (standard error = 0.81) while people with migraines report 3.22 (0.67) (Table 5, Dong et al., 2013). A more thorough review of the available literature was outside the scope of this report.
2.3 Scale of the problem in Disability-Adjusted Life Years
We next turn to the effect of pain on disability weights, used to construct Disability-Adjusted Life Years (DALYs).[13] Disability weights indicate the quality of someone’s overall health state from 0 (full health) to 1 (death). They provide another means with which to compare the relative severity of the conditions in this report. Our primary motivation, however, is that using DALYs allows us to roughly compare the scale of the conditions to other problems in the global burden of disease.
In Table 1, we provide the disability weights and corresponding descriptions of the health state for a range of relevant conditions.[14] Advanced cancer has a high weight at about 0.5; migraines are slightly lower at 0.44; our generated weight[15] for cluster headaches is the highest at 0.66; and low back pain is the lowest, varying between 0.02 for mild pain to 0.32 when the pain is severe and extends to the legs as well. There is also a seemingly strange result: the disability weights for terminal cancer patients with vs without pain relief are implausibly similar - 0.569 vs 0.540 - implying that cancer without pain relief is barely worse. This is especially odd considering that multiple randomized controlled trials have captured the significant effect opioids have on pain (see Wiffen et al., 2017 and [Appendix 1].
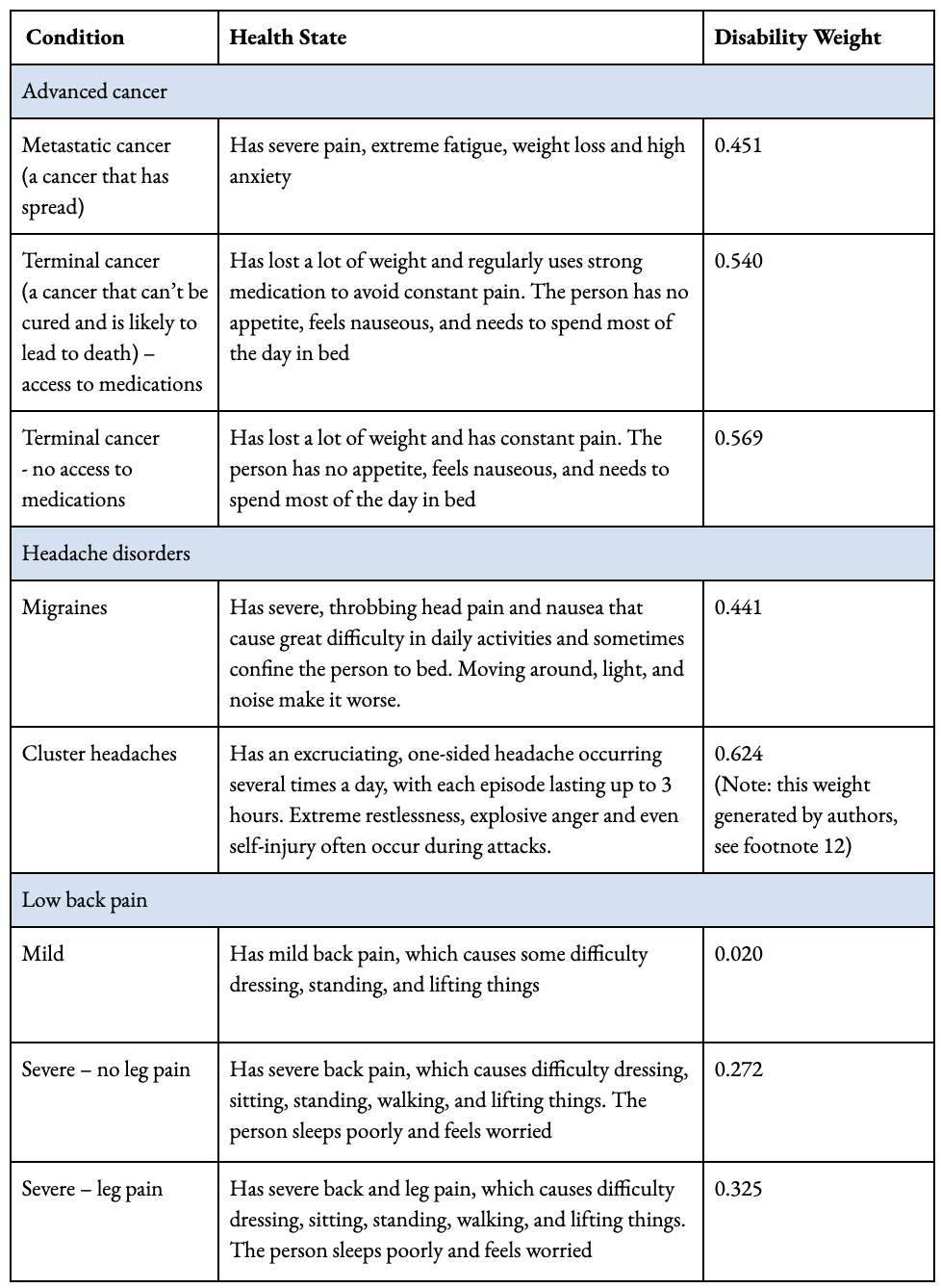
Table 1: Description of health states and corresponding disability weights. Derived using paired comparison questions (Saloman et al., 2015). *Note cluster headaches DW footnote is actually 15, not 12 as indicated in the table.
Next, we estimate the scale of each sub-area of the problem in DALYs. We retrieved data for migraines and low back pain directly from the Global Burden of Disease’s online database. To estimate the burden from cluster headaches we made crude back-of-the-envelope estimates described in full in [Appendix 2]. In brief, we used DALYs for migraines, and adjusted for the relative prevalence with cluster headaches (0.1% to 14%; Ekbom et al., 2006; Leone & May, 2020; Stovner et al., 2018), naively assuming that those with migraines and cluster headaches each suffer for the same duration, on average, over a given time period. The results are displayed in Figure 2 alongside the burden from depression and malaria to facilitate comparison.[16]

Figure 2: Global burden of pain-related disorders and depression in Disability-Adjusted Life Years (DALYs) . Note that we derived an estimate for disability weight for cluster headaches; see Appendix [2] for details.
Figure 2 shows the huge disease burden from low back pain and migraines, discussed further below, which are each roughly comparable in scale to depression and malaria. A few things are worth noting. First, the DALYs from cluster headaches are roughly two orders of magnitude smaller than the other issues referenced. Second, moral views which give additional weight to more extreme forms of suffering will conclude the moral importance of cluster headaches is relatively more important than the DALY total indicates. Third, the size of different problems does not, by itself, tell us which will be the most cost-effective to work on; assessing cost-effectiveness is a further issue and outside the scope of this report.
Estimating the number of DALYs for terminal conditions requiring opioids is trickier. The seminal paper of the Lancet Commission on Global Access to Palliative Care and Pain Relief (LC), (Knaul et al., 2017a), has been the largest effort to quantify the burden of health-related suffering associated with a need for palliative care and pain relief. Using their derived figures for serious health-related suffering,[17] we can make crude estimates of “DALYs”.[18] [19] The LC analysed the 20 health conditions that cause most of the burden of serious health-related suffering; their total estimate is between 6 and 21 billion days, depending on how symptoms are delineated.
As a rough lower bound, we take the proportion of serious health-related suffering caused by moderate-severe pain (6%; other causes include fatigue, weakness, and depression), on the basis that this would be highly treatable with opioids. Applying the disability weight for terminal cancer (0.57) - although noting that cancer is only one of the associated conditions - produces ~0.56 million “DALYs” for moderate or severe pain during life-threatening and life-limiting conditions, for which opioids would be likely indicated. Opioids could also help treat mild pain and shortness of breath. Including these symptoms, and using the same disability weight (noting this is likely an upper bound), the numbers rise to ~2.72 million “DALYs” (see [Appendix 2] for full details). It is also worth noting that opioids are an essential medicine outside of palliative care; the Lancet Commission on Global Access to Palliative Care and Pain Relief (LC) estimated the total need of opioids, including operative and traumatic settings, to be approximately seven times that which is needed for palliative care (Knaul et al., 2017a).
Our discussion now turns to three possibilities for pain relief: terminal conditions requiring access to opioids, headache disorders, and low back pain, respectively.
3. Terminal conditions requiring access to opioids
3.1 The problem
Individuals with life-limiting conditions such as cancer and HIV often experience excruciating pain that is most pronounced towards the end of their lives (Portenoy & Lesage, 1999; Spencer et al., 2019). Cancer causes pain through several mechanisms. A tumour can invade surrounding tissue such as bones, muscles, nerves, and organs, and/or infiltrate and obstruct hollow organs. Treatment can also be a major source of pain, occurring through nerve injury post surgery or as a side effect of chemotherapy. People with HIV typically experience lower limb bone pain, headaches and nerve-related pain for reasons that are not well understood (Perry et al., 2013). If not controlled adequately, pain can affect other facets of wellbeing restricting one’s mobility, functionality, and causing significant psychological distress. The pain can be so severe that suicidal thoughts are expressed when pain relief is not available (see Box 1).
At least seven million people experience moderate to severe cancer-related pain a year, with 75% residing in low and middle-income countries (Knaul et al., 2017a). The incidence will only rise in the future as deaths from communicable diseases fall, with experts forecasting that by 2060 more than double the number of cancer patients will die every year with serious health-related suffering (Sleeman et al., 2019). Another four million people currently living with HIV are estimated to experience moderate to severe pain, also warranting the need for opioids (Knaul et al., 2017a). Cancer and HIV cause over 700 million serious health-related suffering moderate to severe pain days, and are responsible for approximately 90% of total serious health-related suffering moderate to severe pain days, based on Figure 7 in the LC report (Knaul et al., 2017a).

See here to access links
Palliative care refers to treatment that aims to alleviate suffering and maintain quality of life, as opposed to curing the condition that causes the suffering (Clark, 2007). Opioids are an essential (and arguably the most important) instrument[20] in the palliative care toolkit, helping control moderate to severe pain (Portenoy & Lesage, 1999) and other aspects of serious health-related suffering such as shortness of breath (Peiffer, 2011).[21] Despite the high incidence of side effects[22] experienced by users, opioids are considered the most reliable and effective analgesic currently known to the medical profession. A Cochrane review concluded that 19 out of 20 people with moderate to severe cancer-related pain had their pain reduced to mild or no pain with the careful adjustment of the dose to control pain (Wiffen et al., 2017). Further, a review of oral morphine demonstrated that more than 6 out of 10 patients were very satisfied with their morphine treatment, with only 1 out of 20 stopping their morphine, primarily because of side effects (Wiffen, Wee & Moore, 2016). Because of these properties, combined with its affordability, the World Health Organisation lists opioids as an essential medicine (WHO, 2019).
In many parts of the world it is difficult or virtually impossible to access opioids and palliative care. LC estimated the unmet opioid need by comparing the burden of pain and shortness of breath requiring opioids to what was actually available in countries (Knaul et al., 2017a). They determined that less than 5% of opioid need for serious health-related suffering is met in LMICs, which is substantially lower than the 98% that is met in HICs.[23] This is displayed in Figure 3.[24] As previously noted, opioids are also needed outside of serious health-related suffering, such as for pain post-surgery and acute traumatic injuries; when this is included, less than 1% of opioid need is met (Knaul et al., 2017a).[25]
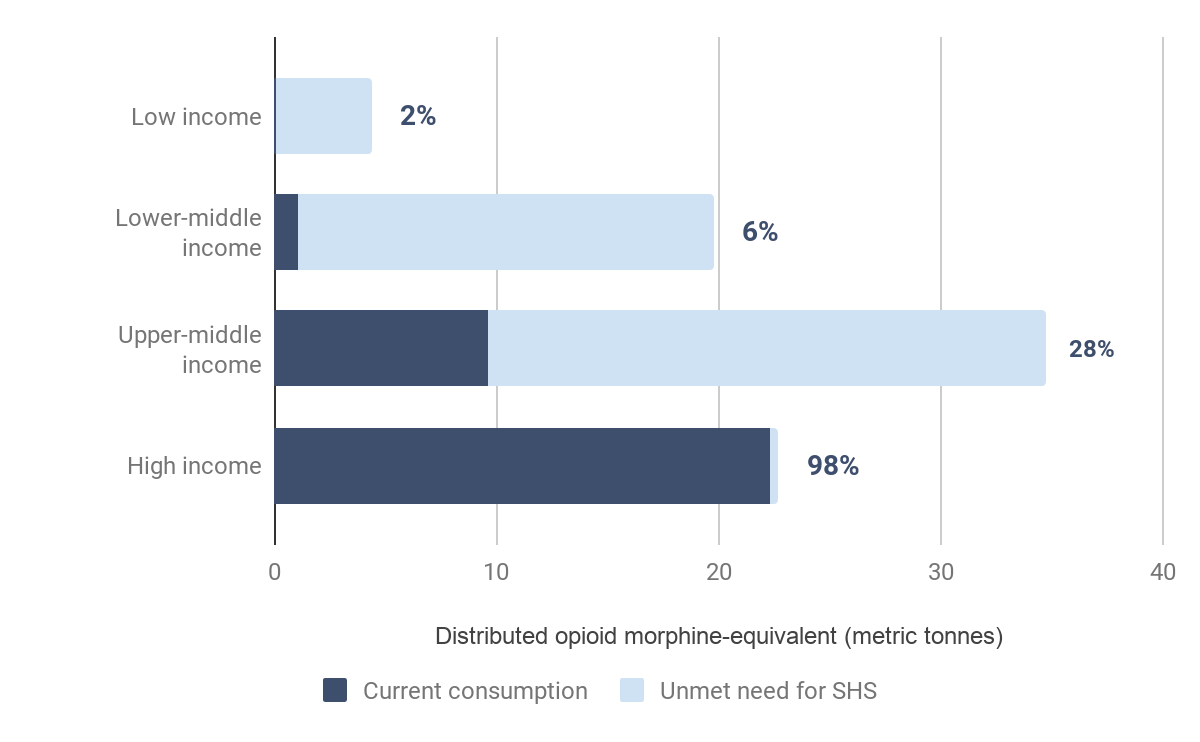
Figure 3: Estimates of met opioid need in palliative care
Grouped by World Bank Income Group. Diagram adapted from Knaul et al., 2017a. See Footnote 23 for further information about how Knaul et al. derived estimates.
3.2 Barriers to progress
The price of morphine itself is not the main barrier. At the lowest international price, morphine costs US $8 per patient for three months (at $0.011 per milligram this provides enough morphine to last 90 days of moderate to severe pain) (MSH, 2015). To meet the global shortfall, LC estimated that it would cost $145 million a year, only 0.009% expenditure of the health budgets of LMICs (Knaul et al., 2017a).
Instead, untreated moderate to severe pain, and other serious health-related suffering, exists because of a complex and interrelated array of barriers, which we summarise below as: (1) laws and regulations, (2) education of health professionals, and (3) supply chains. This is displayed below in Figure 4.
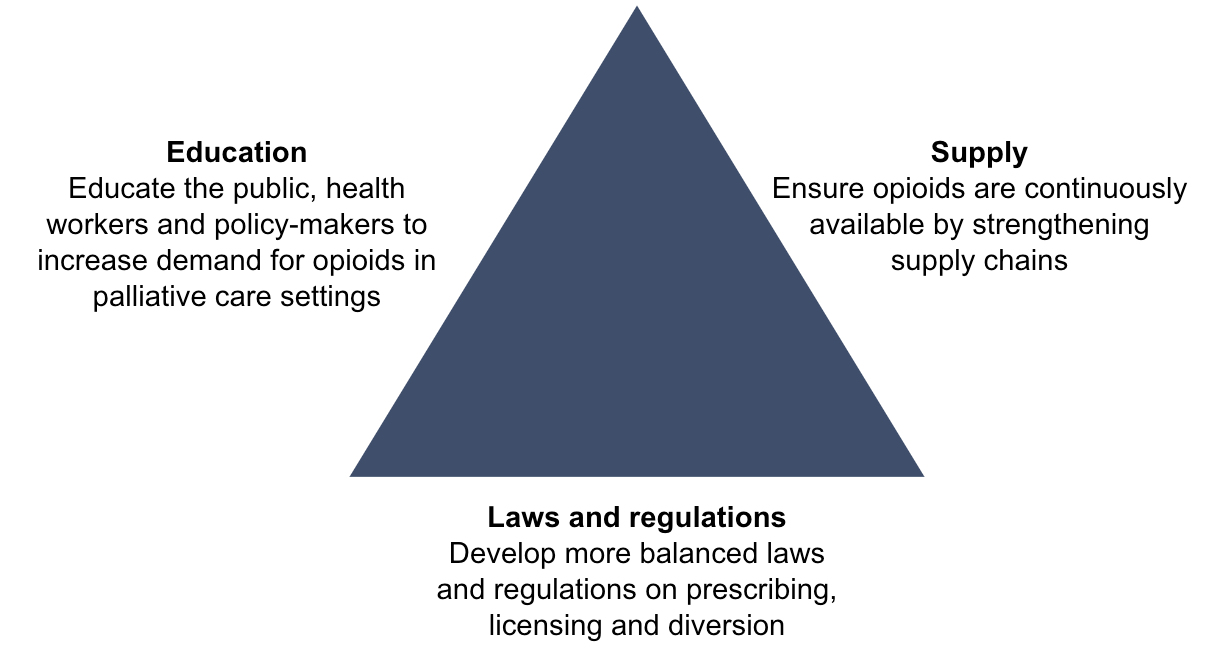
Figure 4: Primary barriers to opioid consumption for serious health-related suffering. Discussed in more detail under the heading ‘Barriers to Progress.’ All three barriers need to be addressed to effectively and sustainably improve opioid consumption for serious health-related suffering.
Laws and regulations
Unbalanced laws and regulations have evolved from the world’s effort to prevent the illicit production and consumption of narcotics (Clearly, Gelband &Wagner, 2015; Knaul et al., 2017a). Since an international treaty, the Single Convention on Narcotic Drugs, was adopted by UN member states in 1961 to prevent opioid misuse and ensure access for medical purposes, countries have closely regulated and monitored opioid availability to minimise the risk of diversion and illicit use (INCB, 2015).
The International Narcotics Control Board (INCB), was founded in 1968 - not long after the signing of the Single Convention was established - to monitor and assist countries to fulfil the goals of the treaty (Messac, 2014). However, their model policies were considered ill-suited for resource-poor settings with weak health care systems and poor administrative capacity, and did not give due consideration to the availability of opioids for serious health-related suffering. For example, early on the INCB used ‘number of doctors per capita’ as a guide to ascertain appropriate levels of opioid consumption, and recommended countries apply trade embargoes on countries that produced or imported more opioids than the level they set (Messac, 2014). Naturally, this method forced LMICs with fewer doctors per capita to under-report their requirements. Combined with the rhetoric that surfaced surrounding the widespread fears about opioids, the increased international emphasis on opioid control led to many jurisdictions implementing laws and regulations that were deemed excessive in retrospect.
The INCB has since admitted they have unfairly emphasised the risk of opioid misuse to the detriment of availability of opioids for medical purposes (Clearly & Maurer, 2018). They now are actively involved in helping delineate and address the global barriers to opioid consumption for medical purposes and strongly advise countries to create favourable environments that facilitate access to pain relief.
Examples of the administrative hurdles and disincentives that health professionals face include: (Cleary et al., 2013; INCB, 2015; Fraser et al., 2017):[26]
- Physicians require special licences to be able to prescribe opioids (most HICs grant all doctors with prescribing rights the ability to prescribe opioids).
- Patients must be formally registered to receive opioids.
- Prescriptions must be written on a particular script paper, which in some countries is difficult to source (in Mexico, authority script pads were unavailable outside large cities until 2015).
- A provider can only issue opioids for a short time period (a limit of two days in some countries) before a patient must come back to attain another script.
The examples above relate to prescribing, but strict regulations also curtail the activity of pharmacies, hospitals, and producers. For example, the 1985 Narcotic Drugs and Psychotropic Substances (NDPS) Act in India resulted in hospitals requiring three or four licences (depending on the state) to supply morphine (Rajagopal & Joranson, 2007). The validity of one license would frequently expire by the time a hospital would acquire another, rendering it nearly impossible to stock and dispense morphine. As evidence of the impact of these restrictions, the consumption of morphine in India fell by 97% from 716 kg in 1985 to 18kg in 1997 after the NDPS Act was adopted (Rajagopal, Joranson & Gibson, 2001).
In aggregate, these regulations can have a significant influence on clinical practice and opioid availability, particularly in low-resource settings.

See here to access links in box
Education of health professionals
After sustained advocacy from local and international organisations, some LMICs have eased their overly restrictive regulations to facilitate opioid consumption to treat serious health-related suffering (INCB, 2015; Rajagopal, 2015; Rhee et al., 2018). However, decades of restrictions have created an environment in which health professionals do not feel comfortable prescribing opioids, partly because of the fear of abuse, but also because of their unfamiliarity with the properties of opioids (Knaul et al., 2017a; Clearly, Gelband & Wagner, 2015).
When the INCB interviewed government officials about the major barriers to opioid access in their respective countries, the most common impediments reported were a lack of training and awareness amongst health professionals, followed by a fear of addiction and limited resources (INCB, 2015). A Nigerian study found that 90% of teaching hospital physicians had received no formal training on opioids for pain management, with only 50% reporting they would consider prescribing opioids for severe cancer pain (Ogboli-Nwasor, Makama & Yusufu, 2013). Palliative care is yet to be integrated into the national undergraduate medical curriculum for doctors and nurses in India (Rajagopal 2015; Rajagopal & Joranson, 2007).
Supply chains
Low prescription rates combined with security costs, small profit margins for the sale of opioids, and fragile supply chains have meant few hospitals and pharmacies stock opioids in LMICs. Resources are required to store morphine safely (such as a locked safebox secured to a wall) given the risk of opioid diversion (Knaul et al., 2017a). The low price of morphine proves a double-edged sword, as dispensaries have little financial incentive to sell morphine (Clearly & Maurer, 2018). Similarly, the high-security costs and lack of profitability dissuades local manufacturers from producing morphine, forcing many countries into importation.
LMICs generally don’t have the resources and governance to maintain efficient supply chains, which is only exacerbated by onerous regulatory and reporting requirements that are required for importation (Rhee et al., 2017; Clearly & Maurer, 2018). Altogether, there is a high chance someone with a prescription for morphine in LMICs would not be able to find somewhere in their vicinity to provide them with morphine. Of 120 health facilities in Kenya and Uganda, only 7% dispensed morphine (Fraser et al., 2017). Of those with stock, it was more common to run out of morphine than to throw away expired morphine.
3.3 How can we improve opioid availability?
Improving the availability of opioids for serious health-related suffering in LMICs therefore requires a multifaceted, context-specific solution that incorporates measures to safeguard against the risk of opioid diversion and dependence. Despite the task appearing difficult, many LMICs have made considerable progress in addressing these barriers (Clearly, Gelband & Wagner, 2015; Knaul et al., 2017a). Some of the most notable success stories are from India and Uganda.
Case study: The Pain and Palliative Care Society, India (now Pallium India)
Only two clinics stocked oral morphine in 1993 in the state of Kerala in India because of the barriers mentioned above. At the time, local leaders established the Pain and Palliative Care Society, an outpatient palliative care clinic based at a government-funded hospital (Rajagopal, 2015). They primarily operated on voluntary community support and donations from local and international actors. Within months, the clinic was seeing more patients then their capacity allowed, spurring them to expand their services. By 1998 they had established twenty satellite centres across Northern Kerala that were managed by local volunteer groups with doctors consulting when required (Vijay & Monin, 2018).
During this period, the Pain and Palliative Care Society recognised the difficulties patients had in accessing morphine. With help from the Pain and Policy Studies Group (now the Walther Centre for Global Palliative Care and Supportive Oncology) based in Madison, Wisconsin, they analysed the NDPS Act (as discussed above) and proposed simpler regulations and laws (Rajagopal, 2015). After sustained advocacy, in 1998 the government of India endorsed their recommendations, and urged state governments to do the same. The states of Kerala, Tamil Nadu, Karnataka, New Delhi, and others made changes to their regulations.
Whilst significant improvements in opioid access were seen in Kerala (further information below), Rajagopal (2015) states that some states showed a "modest" improvement and the change in most other states was "minimal". Experts we spoke to emphasised that all three sides of the barriers triangle (Figure 4) need to be tackled concurrently; it is not enough to just change regulations. Based on this, the success in Kerala is, at least in part, because of the increased demand for opioids resulting from the engagement with local people on the ground. There have since been many further developments including (Rajagopal, 2015; Vijay & Monin, 2018):
- The formation of Pallium India, a not-for-profit that has carried on the work of the Pain and Palliative Care Society.
- The integration of palliative care within existing health care infrastructure in Kerala resulted in the deployment of government-funded palliative care nurses at 900 community clinics.
- Delivery of a palliative care educational course across India to health professionals and community volunteers.
The number of institutions stocking morphine and the availability of morphine per capita have increased significantly in Kerala. As of 2014:
- 170 institutions stock oral morphine (compared to two in 1993) (Rajagopal, 2015)
- 1.426 mg of morphine equivalent (ME) is available per capita, an order of magnitude higher than India’s availability of 0.11mg ME per capita (Rajagopal, Karim & Booth, 2016). However, it still remains well below the estimated opioid need to alleviate serious health-related suffering (Knaul et al., 2017a).[27]
Notably, India’s success has transpired without demonstrable evidence of opioid diversion. In an attempt to identify accounts of opioid diversion, Rajagopal and colleagues (2001) closely monitored the oral morphine procured, the stock held, and dispensed at a clinic in Kerala over a two-year period and found no discrepancies in stock quantities that suggested instances of opioid diversion.
Case study: Hospice Africa Uganda
An increase in Uganda’s opioid availability was catalysed by similar work by Hospice Africa Uganda (HAU) (Merriman & Harding, 2010). Founded in 1993, HAU began by providing in-home palliative care services to those that needed it most. From the beginning, HAU built a strong relationship with Uganda’s Ministry of Health, after recognising the importance of attaining the government's support to address the systemic barriers to palliative care (Fraser et al., 2017).
HAU convinced the Ministry of Health to import morphine powder in September 1993, which they were able to effectively convert into oral morphine. After demonstrating the benefit that palliative care and opioids brought to patients with life-limiting illnesses, the government continued to support HAU, allowing them to expand their services across Uganda (Merriman & Harding, 2010). They also listed morphine as an essential medicine, making it available to patients with cancer-related pain at no cost.
Another major breakthrough came in 2004, when the Ministry of Health approved legislation allowing nurses who had undergone nine months of palliative care training with HAU to prescribe oral morphine (Clark et al., 2007). An apparent rise in opioid availability reflects their success, having increased by 35% between 2001 and 2013[28] which is significant when considering that Africa’s availability reportedly fell by 18%[29] during that same time period (INCB, 2015).
Other examples
Other LMICs have had similar successes, including Vietnam, Serbia, Mexico, Kenya, and Jamaica, providing more evidence of tractability (Knaul et al., 2017a; Cleary, Gelband & Wagner, 2015; Cleary & Maurer, 2018; Fadhil, Lyons & Payne et al., 2017). Still, even these countries remain substantially short of facilitating widespread access.
In summary, factors on both the supply and demand sides need to be addressed: reducing onerous regulations, educating health professionals, expanding palliative care services, raising general awareness, and broadening the types of caregivers who can prescribe opioids. This complex array of solutions adds to the costs of attempts to improve access to opioids.
A range of nonprofits are actively trying to improve access to pain relief and palliative care in LMICs. We provide a brief description of their efforts and accomplishments below. Cost-effectiveness analyses are beyond the scope of this report; we expect any cost-effectiveness analyses would be fairly uncertain, given the limited data currently available, and the difficulties of causal attribution with policy changes. Nevertheless, it is something we wish to see in the future, drawing on the methods proposed by Founders Pledge and GiveWell, if only to make assumptions explicit.
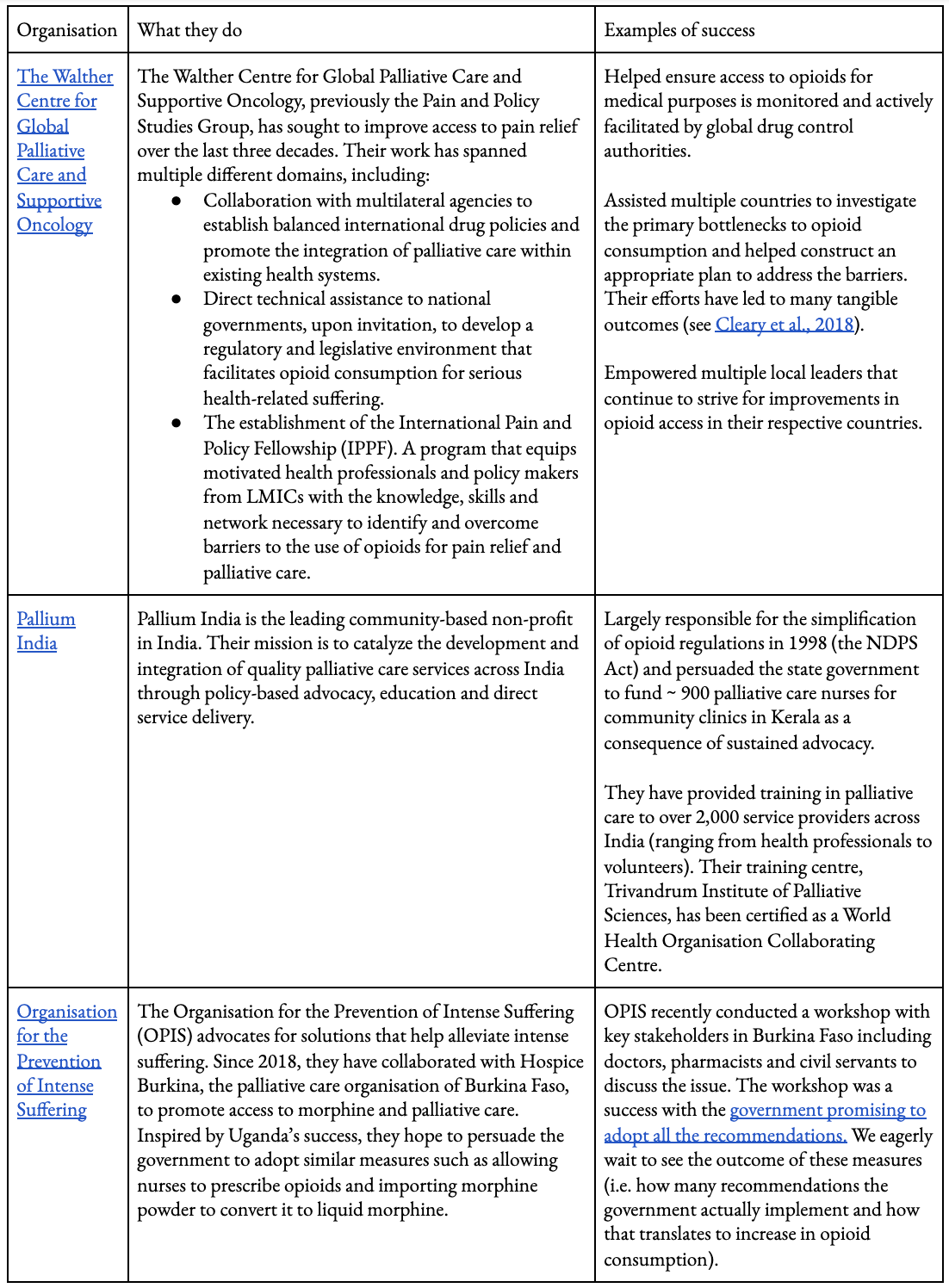
Table 2: A sample of the leading nonprofits helping to improve access to pain relief and palliative care. See here to access links in table.
4. Headache disorders
Headache disorders make up a large portion of the global burden of disease. 47 million Disability-Adjusted Life Years (DALYs) are lost to headache disorders each year, more than any other neurological disorder, and comparable to malaria (46 million DALYs) and depression (47 million DALYs) (Global Burden of Disease’s Online Database). The most common forms of headache disorders are migraines, tension-type headaches, and medication-overuse headache (Steiner & Stovner, 2019).[30] They vary significantly in severity both between and within the various causes. At one end of the spectrum are tension headaches, often considered a minor annoyance that the majority of the population has had to endure. At the most extreme end are cluster headaches, frequently referred to as ‘suicide headaches,’ (Rozen & Fishman, 2012; Buture et al., 2020). In this section, we explore migraines as they constitute the majority of DALYs attributed to headache disorders, and cluster headaches because of the degree of pain and suffering they inflict upon an individual.
4.1 Migraines
Migraines are common, disabling, and recurring headaches affecting up to 14% of the population (Stovner et al, 2018). They are characterised by unilateral, pulsating, moderate to severe headaches that are typically associated with intense nausea and a heightened sensitivity to light and sound, lasting between 4-72 hours (Charles, 2013). Some experience an aura before or during their migraine, which are transient neurological disturbances commonly manifesting as visual disturbances such as flashing lights or zig zag lines, or sensory disturbances such as numbness and tingling in a hand, lips or tongue. It is also common to feel fatigued, low in mood, and find it hard to concentrate before the onset and the completion of a migraine. Together these symptoms can be debilitating, making it impossible to work or lead a normal life, particularly for those without access to effective treatments (Steiner et al., 2018; Guitera et al., 2002). This is made worse by the general population’s lack of awareness surrounding the severity of headache disorders leading to stigma and unrealistic societal expectations, e.g. that individuals with an acute migraine should be able to carry out their normal tasks (Steiner & Stovner, 2019). Migraines are the second leading cause of years lived with disability after low back pain (Global Burden of Disease visualisation) and estimated to cost the European Union over $100 billion each year, driven largely by its negative effects on productivity (Linde et al., 2012).
Scientists are still uncertain about the biological basis of migraines. The leading hypothesis is that neurons activate spontaneously in a self-propagating wave across the brain, triggering the activation of the trigeminal nerve, the nerve responsible for transmitting pain signals to the brain from facial structures (Noseda & Burstein, 2013). Despite not knowing exactly how migraines evolve, effective treatments to abort and prevent migraine are available. COX inhibitors like Aspirin and Ibuprofen effectively terminate 1 in 2 people’s migraines (Kirthi, Derry & Moore, 2013; Rabbie, Derry & Moore, 2013). 60% of people who take the more expensive triptans are pain free within two hours (Derry, Derry & Moore, 2014).[31] Side effects are reasonably common with triptans, particularly when administered under the skin - in an RCT, the intervention arm was twice as likely to report an adverse effect. Effects are of a mild to moderate severity; common side effects include chest tightness, vomiting, and local injection site reactions (Law, Derry & Moore, 2013). Other acute treatments are also available for the migraines that are particularly severe, at times combining the use of two agents (NICE, 2012). Preventative agents are effective as well, with propranolol and topiramate reducing the monthly frequency of migraine by over 50% (Linde & Rossnagel, 2004, Salisbury-Afshar, 2014). Such medications allow patients, particularly those with severe migraine disorders, to control their migraines and substantially reduce the effect it has on their lives.
Misdiagnosis and poor-quality care appear to be the primary barriers from a population health perspective. In a survey conducted in collaboration with the WHO, experts estimated that only 40% of individuals who suffered from migraines received a professional diagnosis worldwide, and this figure is 13% in low-income countries (WHO, 2011). They believed the reasons for this are two-fold:
- People with headaches do not seek help: Only 15-25% of those with probable migraines sought care from a health professional across Europe according to the Eurolight survey (Katsarava et al., 2018). The general public do not recognise the seriousness and treatability of migraines – incorrectly attributing it to psychological factors and stress (Steiner & Stovner, 2019).
- Health professionals are misdiagnosing: Lack of education of health professionals was the most commonly reported barrier by experts (WHO, 2011). On average, fewer than five hours of training are utilised to instruct about headache disorders across medical schools (WHO, 2011).
Even if patients do receive a diagnosis, it appears unlikely that they would receive the correct treatment. The Eurolight survey discovered the majority of those who would benefit from preventive agents (i.e. having more than 5 migraines a month), were not taking a preventative agent (Katsarva et al., 2018). However, it was not clear how many of those chose not to take preventative agents due to contraindications or personal reasons. The survey also found some reluctance by GPs and specialists to prescribe triptans.
It appears then that the best way to reduce the burden of migraines is through education: of the public, health professionals, and key decision-makers.
In an attempt to accomplish this, Lifting the Burden (LTB) – an international not-for-profit - launched the Global Campaign against Headaches in 2004 (Steiner et al., 2010). Over the last two decades they have conducted epidemiological research in an attempt to convince governments to allocate resources to this cause (Steiner & Stovner, 2019; Steiner et al., 2010). In 2015, they modelled multiple interventions to reduce the burden of headaches at the population level in four LMICs and found that aspirin used to abort a headache would cost less than $100 per healthy life-year gained (Linde, Steiner & Chisholm, 2015). LTB argues that this potentially under-estimates the effectiveness as it does not incorporate productivity gains. It hypothesises that, from a societal perspective, aspirin for migraines may be a cost-saving intervention (Steiner & Stovner, 2019).
More recently, LTB assisted neurologists in Georgia and successfully established the country’s first self-sustaining headache service (i.e. it does not require government funding to keep running), which expanded to seven other services in the country (Giorgadze et al., 2018). Although this doesn’t directly address the insufficient understanding of headache disorders among health professionals, it provided preliminary evidence demonstrating that the public were willing to pay to have their headaches assessed and treated. LTB envisages such services could strengthen the whole health system to manage headache disorders, through providing support and guidance to pharmacies, clinics (where most headaches should be managed), and hospitals. In Estonia, LTB helped deliver an educational intervention to nearly 300 GPs that led to a decrease in the number of investigations[32] they requested such as CT scans and blood tests (from 26% to 4%) and an increase in medication initiation (from 58% to just above 80%) one year after program delivery (Braschinsky et al., 2016). However, the number of requested investigations climbed back up to 23% at two years after program delivery (Brachinsky et al.,2017).
We have not evaluated LTB; here are open questions we would want to address with further work, such as:
- What is the best method to improve health professional practice? Should it be done in isolation, or part of more comprehensive educational reform?
- To what degree would specialist headache services be required if LMICs had more robust primary care services? What is the best method to strengthen the quality of care provided by primary health care services for headache disorders?
We understand LTB is conducting a comprehensive analysis of the cost-effectiveness of structured headache services with the London School of Economics (Steiner & Stovner, 2019), which we anticipate will clarify some of our uncertainties.
4.2 Cluster headaches
Cluster headaches (CHs) are a rare form of headache with an estimated prevalence of 0.1 – 0.2%, affecting approximately 7-15 million people around the world (Ekbom et al., 2006; Leone & May, 2020). They are discussed widely across the literature because of the intensity of the pain they inflict. Cluster headaches most commonly manifest as excruciating one-sided headaches that have very particular temporal patterns (May, 2005). The attacks last between fifteen minutes to three hours, and can occur several times a day. Individuals with the episodic form have an attack everyday for six to twelve weeks, after which they experience no attacks for up to twelve months. Individuals with the chronic form have a much shorter, or no, remission period. Since they were first documented, the intensity of pain has always been a salient feature of the condition.[33] It is frequentely referred to as the ‘suicide headache’; reports suggest between 30% to 60% of people contemplate suicide during an episode over a twelve-month period.[34]
For the majority of cluster headache sufferers, some relief can be attained. High-flow oxygen and injectable triptans[35] are proven abortive treatments (see Table 3) (May et al., 2018; Law, Derry & Moore, 2013; Bennet et al., 2015). Preventative agents can reduce headache frequency and disability to some degree for some individuals (May et al., 2018). Guidelines recommend verapamil, a calcium channel blocker, as a first-line preventative agent due to its efficacy and tolerability (NICE, 2012). A small double-blind placebo-controlled trial found that 80% of the patients that had verapamil had over a 50% reduction in headache frequency and abortive treatments (Leone et al., 2000).[36]
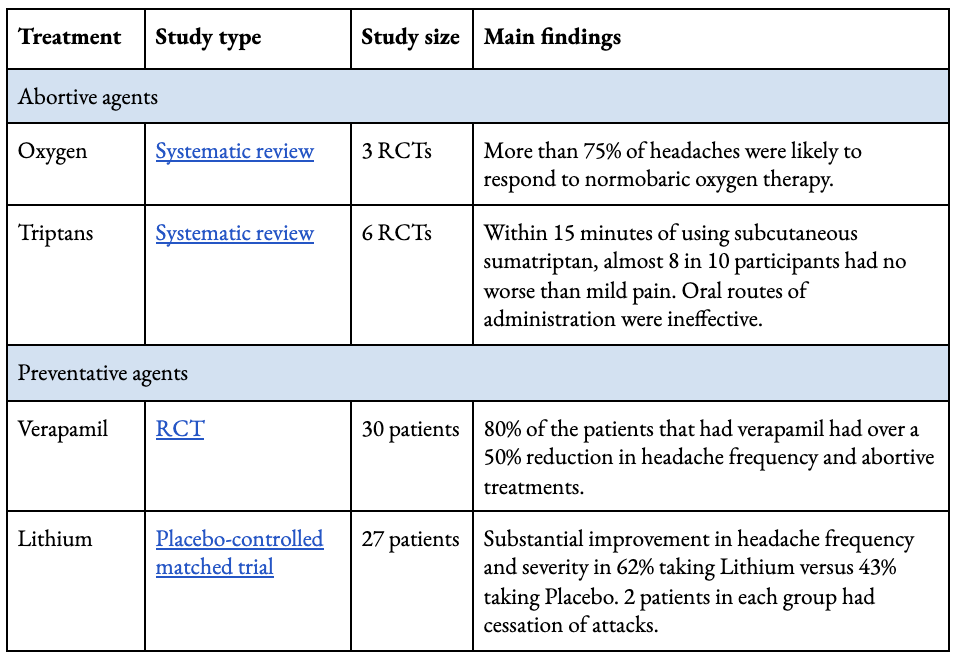
Table 3: Effective treatment options available to abort and reduce frequency of attacks with corresponding trials. See here to access links in table.
Similar to migraines, a significant bottleneck to reduce societal burden appears to be the recognition of cluster headaches by health professionals (Buture et al., 2019). The mean time to diagnosis for patients in the UK was 2.6 years, with a patient on average seeing three family doctors before they received a diagnosis (Bahra & Goadsby, 2004). Similarly, a survey across the US found that 42% of patients received a diagnosis only five years after the onset of disease (Klapper, Klapper & Voss, 2000). It was commonly mistaken for trigeminal neuralgia (a chronic pain condition that affects the trigeminal nerve), sinusitis (inflammation of the sinuses), dental problems, or migraines, with reports of patients undergoing invasive procedures such as tooth extractions and intracranial surgeries in a desperate attempt to find much-needed relief (Buture et al., 2020).
Medication access is another barrier. In a large online survey that mainly received responses from those living in the UK, USA, and Canada, 51% of respondents had some difficulty in accessing oxygen (Pearson et al., 2019). In another survey of headache societies across 22 countries (primarily high-income countries), 50% reported that no reimbursement was available for oxygen therapy which can incur non-negligible out of pocket costs (Evers et al., 2017). Similarly, injectable triptans are relatively expensive, costing approximately $50 for two subcutaneous injections, and are not available in all LMICs (Steiner & Stovner, 2019).
For 10-20% of CH sufferers the available treatments are ineffective or contraindicated (May et al., 2018). Individuals living with CHs consistently report a diminished quality of life despite access to effective abortive agents (Jürgens et al., 2010). Multiple trials are underway to identify effective agents that could be used safely to treat refractory cluster headaches (May et al. ,2018). Patient surveys and many anecdotal reports suggest the use of psilocybin and other psychedelics can abort and prevent an entire cycle of attacks (OPIS, 2020). Schindler and colleagues at Yale University are currently investigating the efficacy of psilocybin in a small randomized trial.
If the hypothesized association holds, this could greatly improve the quality of life of individuals with cluster headaches, particularly if they are found to have a substantial preventative effect. The Organisation for the Prevention of Intense Suffering (OPIS) has launched a campaign in Finland to legalize access to patients with cluster headaches, arguing that a lack of RCT evidence shouldn’t disallow access to agents that could significantly improve an individual's life (OPIS, 2020). They are also looking to guide policymakers internationally on this issue. Further research is required to determine whether drug policy reform is a more cost-effective approach than improving access to currently legal treatments.
Very little of the data we found on barriers originated from LMICs, though we expect equivalent barriers exist there. It would be helpful to gain a better understanding of the barriers in LMICs, in order to determine whether the main issue lies in delay of diagnosis or difficulty in treatment access. Tailored interventions then may look like better access to oxygen or triptans, educational programs for health professionals, construction of headache services, or an acceleration of current trials (see Table 4).
5. Low back pain
Low back pain is the largest single contributor to years lived with disability globally, and is responsible for 60.1 million DALYs in 2015 (Vos et al., 2017). Its prevalence is rising as the population ages; there has been a 54% increase in Years Lived with Disability globally between 1990 and 2015 (Vos et al., 2017). It is increasingly seen as a long-lasting condition of persistent or fluctuating pain of low to medium intensity (Kongstead et al., 2016).
Most adults will experience back pain sometime in their life (Lemeunier, Leboeuf-Yde & Gagey, 2012). The majority will spontaneously recover with a substantially lower level of pain after one year (Kongstead et al., 2016). The pain will persist in a small proportion, become disabling, and lead to substantial reductions in their ability to perform their work-related tasks and health-related quality of life (Da Silva et al., 2017). This group of severe cases (28%) constitute 77% of the overall burden of years lived with disability (Hartvigsen et al., 2018). The potential disabling nature of low back pain is particularly concerning in LMICs where social security is sparse; the consequences of low-back pain in low- and middle-income countries are not well studied (Hartvigsen et al., 2018).
In most cases of low back pain, the exact cause or source of the pain remains unknown and is therefore referred to as non-specific low back pain (Hartvigsen et al., 2018). Structural abnormalities increase someone’s risk of having back pain, but it is common to have the abnormality and not experience pain (Brinjikji et al., 2015). Hartvigsen et al. (2018) identify multiple other risk factors, including:
- Lifestyle factors – increased body mass index (BMI), reduced physical activity and smoking.
- Psychological factors – depression, anxiety, catastrophising (an irrational belief that something is far worse than it really is) and fear avoidance beliefs (fear of pain leads to avoidance of activities and hypervigilance of pain sensations) (Gatchel et al., 2016).
- Social factors – physically demanding occupation, low educational attainment, features of social insurance systems (e.g. schemes providing easy access to workforce compensation) (Bartys et al., 2017).
Similar factors contribute to the development of disabling low back pain. Pain catastrophising and self-efficacy (belief in one’s ability to influence events affecting one’s life) are two important factors that increase someone’s risk of developing disabling pain (Wertli et al., 2014a; Wertli et al., 2014b).
Like headache disorders, there is a disconnect between recommended and actual practice (Foster et al., 2018). Many practitioners order unnecessary investigations, administer analgesics that may do more harm than good, and provide the wrong advice (Rosenberg et al., 2015; Buchbinder et al., 2009). There is also very little published analysis from LMICs (Foster et al., 2018).
Unlike headache disorders, the recommended treatments are not particularly helpful. Anti-inflammatories have a small effect on pain intensity for acute and chronic back pain (Chou et al., 2016). Exercise-graded programmes and psychological therapies are effective in trial settings, but the benefits are limited (NICE, 2016). There is no evidence to suggest self-management education (i.e. supporting patients to manage their acute pain indepently) is effective when compared to sham for acute back pain, and limited when compared to usual care (NICE, 2016).[37]Despite increasing popularity surrounding the use of cannabis for chronic pain, early observational evidence found cannabinoid users experienced no reduction in pain severity or interference after four-years of use (Cambell et al., 2018).
There is also limited evidence to suggest preventative interventions reduce the incidence of back pain and disability. A recent meta-analysis (Steffens et al., 2017) examined multiple different preventative interventions including exercise, education, exercise + education, back belts, shoe insoles and ergonomic equipment. They found two low-quality randomised controlled trials (RCTs) (Bredo et al., 2001; Soukup et al., 2001), showing exercise + education programs may reduce your risk of having an episode of low back pain by approximately 40%. The RCT also found that the intervention group had taken fewer sick days than the control group. The meta-analysis found no benefit for the other interventions, based on the limited and poor-quality evidence that was available (Steffens et al., 2017).
The main way to tackle the problem therefore seems to be through basic research. Given the large disease burden, the value of information from understanding the underlying mechanisms of pain and finding new, effective solutions is high. Borsook (2012) highlights some future directions for chronic pain (relevant for low-back pain as well as migraines, osteoarthritis, and other conditions). He looks to national funders and public-private partnerships to fund research, as well as to provide incentives to biotechnology companies “at a time when large pharmaceutical companies are pulling out of drug development for brain disorders, including pain, due to perceived risk”, and finally to philanthropists to fund high-risk, high-return projects that could go on to leverage larger funders. For example, Open Philanthropy has awarded grants for pain research.
6. What can be done to help?
In this section, we offer some initial ideas for how individuals and organisations might be able use their time and/or donations to help with this problem. Before that, we provide a non-exhaustive list of considerations relevant to choose whether to prioritise this area.
Considerations that would lead someone to relatively prioritise this problem:
- Valuing alleviating suffering more than increasing enjoyment.
- Valuing alleviating extreme suffering more than alleviating less intense forms of suffering.
- Valuing the additional information derived from working on this cause area. For example, if one thought research into pain, or how to alter healthcare systemic, would have useful applications elsewhere.
Considerations that would lead someone to relatively _de_prioritise this problem:
- Valuing philanthropic programmes or career paths with a stronger evidence-base of effectiveness and/or requiring shorter causal chains, e.g funding the distributing of proven physical health treatments such as life-saving antimalarial bednets.[38]
- Valuing future generations and believing there are more effective ways to positively influence the very long-term future than having impact over the short-term (i.e. the next 100 years).
Career paths
Grant-making at a foundation may be a promising way to work on this problem. For example, by providing research grants for: impact evaluations of interventions like those described in this report; work on metrics that could be used for this purpose; or for novel drug research.
Advocacy is another route. Individuals or organisations with a public platform could spend some of their time raising awareness of cluster headaches and migraines, particularly with medical professionals, or highlighting the often-excessive regulations restricting access to opioids. The International Association for Hospice & Palliative Care offers a structured advocacy program to individuals through palliative care organisations with an overarching aim of improving access and quality of palliative care. Journalism, grassroots activism, working at think-tanks, or in politics or policy are examples of careers within advocacy.
Pursuing a career in research - such as in academia, or research organisations - is another way to further our understanding of this area. For example, by addressing the questions listed below, or by working on understanding the underlying mechanisms that cause pain.
Finally, it may be possible to work directly on pain at the organisations mentioned in this report. More generally, careers in global health and epidemiology, such as at international health organisations (e.g. WHO) or international non-profits (e.g. Gates Foundation), would be a way to gain relevant skills and could be an indirect way to work on this problem.
We recommend visiting 80,000 Hours for more detailed advice on having an impactful career and to read profiles of several of the careers mentioned above.
Volunteering or internships
- OPIS and Clusterbusters are looking for volunteers.
- Pallium India offers internships for students.
- Lifting the Burden is looking for volunteer translators.
Donate
We have not extensively searched for or evaluated donation opportunities. From speaking to leaders of the organisations working to improve access to opioids in Table 2 (The Walther Centre for Global Palliative Care and Supportive Oncology, OPIS, and Pallium India) we understand the organisations are funding-constrained and would be grateful for support. We have not tried to identify fundable entities working on the basic research that seems relevant for low back pain. We would be excited to see further work on evaluating the impact of such organisations but it is outside our current capacity to undertake it.
Further research
Many open questions remain, as alluded to throughout the report. We think the following would be particularly valuable to explore:
- Further research on measuring the scale of pain that improves on our initial calculations in terms of DALYs. This could include:
- A more thorough review of the relationship between pain and subjective well-being. How does pain relief compare to other interventions that improve subjective well-being? Do people adapt to some kinds of pain? What is the effect of extreme pain on subjective well-being?
- Given the relatively large number of cases worldwide (prevalence ~0.1%) and the significant suffering it causes an individual, it would be helpful if future epidemiological studies estimated the burden and distribution of cluster headaches in DALYs. This could make decision-makers and researchers more cognisant of its existence.
- Further analysis on tractability and cost-effectiveness of approaches described in this report: A. How cost-effective are these interventions? As with any impact evaluation of organisations looking to influence policy, we expect this task to be difficult and the results uncertain (see this report from Founders Pledge into evaluating attempts to affect policy). B. How do different ethical viewpoints, such as suffering-focused ethics, change someone’s prioritisation of alleviating extreme pain?[39]
- What is the current state of research on novel pain agents? To what extent should this research be prioritised over improving access to existing treatments?
- Exploration of pain-related topics not included in this review, such as other conditions that cause pain (e.g. endometriosis, trigeminal neuralgia, fibromyalgia).
7. Concluding remarks
It is a tragedy that some suffer tremendously when this could be easily prevented. To some extent, the current state of affairs likely reflects the preferences of politicians and the public to cure diseases and avert deaths, rather than reduce pain which only improves the quality of lives. We hope we have shown that much can, in fact, be done to ease the burden. Further, we hope that this report acts as a springboard for further work both into how best to reduce worldwide pain and whether this problem is a top priority for improving global well-being.
An assortment of further reading is provided below.
8. Learn more
Access to opioids
- 2017 paper by the Lancet Commission on Global Access to Palliative Care and Pain Relief
- The International Narcotics Control Board analysis of the unmet need of opioids for medical purposes, incorporating surveys involving member states
- Disease Control Priorities summary of the barriers and priority solutions to increase opioid availability for palliative care
- Walther Centre for Global Palliative care’s online course “Increasing Patient Access to Pain Medicines Around the World: A Framework to Improve National Policies that Govern Drug Distribution”
Headache disorders
- A comprehensive summary of the societal impact of headache and the measures required to reduce the burden
- Cost-effectiveness analysis of interventions for migraine in four low- and middle-income countries
- The WHO’s characterisation of the barriers preventing adequate treatment of headache disorders
- Disease Control Priorities summary of the barriers and priority solutions to reduce the burden of headache disorders
- A Nature narrative literature review illustrating the cause, biology, clinical features and treatment for cluster headaches
- Effectiveness of Oxygen and Other Acute Treatments for Cluster Headache
- Systematic literature review on the delays in the diagnosis and misdiagnosis of cluster headache
- OPIS policy paper on legalising access to psilocybin
Low back pain
- A Lancet review of the preventative and treatment options for low back pain
- NICE guidelines: Low back pain and sciatica in Over 16s
References
Alva Staufert, M. F., Ferreira, G. E., Sharma, S., Gutiérrez Camacho, C., & Maher, C. G. (2020). A look into the challenges and complexities of managing low back pain in Mexico. Global Public Health, 1-11.
Bahra, A., & Goadsby, P. J. (2004). Diagnostic delays and mis‐management in cluster headache. Acta neurologica scandinavica, 109(3), 175-179.
Bartys, S., Frederiksen, P., Bendix, T., & Burton, K. (2017). System influences on work disability due to low back pain: an international evidence synthesis. Health Policy, 121(8), 903-912.
Bennett, M. H., French, C., Schnabel, A., Wasiak, J., Kranke, P., & Weibel, S. (2015). Normobaric and hyperbaric oxygen therapy for the treatment and prevention of migraine and cluster headache. Cochrane Database of Systematic Reviews, (12).
Buchbinder, R., Underwood, M., Hartvigsen, J., & Maher, C. G. (2020). The Lancet Series call to action to reduce low value care for low back pain: an update. Pain, 161(1), S57.
Burish, M., Pearson, S., Shapiro, R., Yan, Y., & Schor, L. (2019). Cluster headache is one of the most painful human conditions: Epidemiology of cluster headache and probable cluster headache from a large international sample. [Poster]. Retrieved from https://documentcloud.adobe.com/link/track?uri=urn:aaid:scds:US:30c54af9-a174-40c0-844f-03837ff611fb#pageNum=1
Braschinsky, M., Haldre, S., Kals, M., Arge, M., Saar, B., Niibek, M., ... & Steiner, T. J. (2018). Structured education to improve primary‐care management of headache: how long do the benefits last? A follow‐up observational study. European journal of neurology, 25(3), 497-502.
Braschinsky, M., Haldre, S., Kals, M., Iofik, A., Kivisild, A., Korjas, J., ... & Steiner, T. J. (2016). Structured education can improve primary-care management of headache: the first empirical evidence, from a controlled interventional study. The journal of headache and pain, 17(1), 24.
Brinjikji, W., Luetmer, P. H., Comstock, B., Bresnahan, B. W., Chen, L. E., Deyo, R. A., ... & Wald, J. T. (2015). Systematic literature review of imaging features of spinal degeneration in asymptomatic populations. American Journal of Neuroradiology, 36(4), 811-816.
Buture, A., Ahmed, F., Dikomitis, L., & Boland, J. W. (2019). Systematic literature review on the delays in the diagnosis and misdiagnosis of cluster headache. Neurological Sciences, 40(1), 25-39.
Buture, A., Ahmed, F., Mehta, Y., Paemeleire, K., Goadsby, P., & Dikomitis, L. (2020). Perceptions, experiences and understandings of cluster headache among GPs and neurologists: a qualitative study. British Journal of General Practice.
Campbell, G., Hall, W. D., Peacock, A., Lintzeris, N., Bruno, R., Larance, B., ... & Blyth, F. (2018). Effect of cannabis use in people with chronic non-cancer pain prescribed opioids: findings from a 4-year prospective cohort study. The Lancet Public Health, 3(7), e341-e350.
Charles, A. (2013). The evolution of a migraine attack–a review of recent evidence. Headache: The Journal of Head and Face Pain, 53(2), 413-419.
Chou, R., Deyo, R., Friedly, J., Skelly, A., Hashimoto, R., Weimer, M., ... & Grusing, S. (2016). Noninvasive treatments for low back pain.
Clark, D. (2007). From margins to centre: a review of the history of palliative care in cancer. The lancet oncology, 8(5), 430-438.
Cleary, J., Gelband, H., & Wagner, J. (2015). Cancer pain relief. Disease Control Priorities 3.
Cleary, J., Powell, R. A., Munene, G., Mwangi-Powell, F. N., Luyirika, E., Kiyange, F., ... & Cherny, N. I. (2013). Formulary availability and regulatory barriers to accessibility of opioids for cancer pain in Africa: a report from the Global Opioid Policy Initiative (GOPI). Annals of Oncology, 24(suppl_11), xi14-xi23.
Cleary, J. F., & Maurer, M. A. (2018). Pain and Policy Studies Group: two decades of working to address regulatory barriers to improve opioid availability and accessibility around the world. Journal of pain and symptom management, 55(2), S121-S134.
Cleeland, C. S., & Ryan, K. M. (1994). Pain assessment: global use of the Brief Pain Inventory. Annals, Academy of Medicine, Singapore.
Cohen, A. S., Burns, B., & Goadsby, P. J. (2009). High-flow oxygen for treatment of cluster headache: a randomized trial. Jama, 302(22), 2451-2457.
Corniola, M. V., Stienen, M. N., Joswig, H., Smoll, N. R., Schaller, K., Hildebrandt, G., & Gautschi, O. P. (2016). Correlation of pain, functional impairment, and health-related quality of life with radiological grading scales of lumbar degenerative disc disease. Acta neurochirurgica, 158(3), 499-505.
D'Amico, D., Raggi, A., Grazzi, L., & Lambru, G. (2020). Disability, Quality of Life, and Socioeconomic Burden of Cluster Headache: A Critical Review of Current Evidence and Future Perspectives. Headache: The Journal of Head and Face Pain, 60(4), 809-818.
Da Silva, T., Mills, K., Brown, B. T., Herbert, R. D., Maher, C. G., & Hancock, M. J. (2017). Risk of recurrence of low back pain: a systematic review. journal of orthopaedic & sports physical therapy, 47(5), 305-313.
Derry, C. J., Derry, S., & Moore, R. A. (2014). Sumatriptan (all routes of administration) for acute migraine attacks in adults‐overview of Cochrane reviews. Cochrane Database of Systematic Reviews, (5).
Ekbom, K., Svensson, D. A., Pedersen, N. L., & Waldenlind, E. (2006). Lifetime prevalence and concordance risk of cluster headache in the Swedish twin population. Neurology, 67(5), 798-803.
Ekbom, K., Monstad, I., Prusinski, A., Cole, J. A., Pilgrim, A. J., Noronha, D., & Sumatriptan Cluster Headache Study Group. (1993). Subcutaneous sumatriptan in the acute treatment of cluster headache: a dose comparison study. Acta Neurologica Scandinavica, 88(1), 63-69.
Emilsson, A. (2019a). Cluster Headache Frequency Follows a Long-Tail Distribution. EA Forum.
Emilsson, A. (2019b). Logarithmic Scales of Pleasure and Pain: Rating, Ranking, and Comparing Peak Experiences Suggest the Existence of Long Tails for Bliss and Suffering. EA Forum.
Evers, S., Rapoport, A., & International Headache Society. (2017). The use of oxygen in cluster headache treatment worldwide–a survey of the International Headache Society (IHS). Cephalalgia, 37(4), 396-398.
Ferrell, B. (1995). The impact of pain on quality of life. A decade of research. The Nursing Clinics of North America, 30(4), 609-624.
Fadhil, I., Lyons, G., & Payne, S. (2017). Barriers to, and opportunities for, palliative care development in the Eastern Mediterranean Region. The Lancet Oncology, 18(3), e176-e184.
Foster, N. E., Anema, J. R., Cherkin, D., Chou, R., Cohen, S. P., Gross, D. P., ... & Turner, J. A. (2018). Prevention and treatment of low back pain: evidence, challenges, and promising directions. The Lancet, 391(10137), 2368-2383.
Fraser, B. A., Powell, R. A., Mwangi-Powell, F. N., Namisango, E., Hannon, B., Zimmermann, C., & Rodin, G. (2017). Palliative care development in Africa: lessons from Uganda and Kenya. Journal of Global Oncology, 4, 1-10.
Furlan, A. D., Harvey, A. M., & Chadha, R. (2020). Warning from Canada: Latin America, South Africa and India may face an opioid epidemic in the coming years. Journal of Global Health, 10(1).
Giorgadze, G., Mania, M., Kukava, M., Dzagnidze, A., Mirvelashvili, E., Steiner, T. J., & Katsarava, Z. (2018). Implementation of effective, self-sustaining headache services in the Republic of Georgia: evaluation of their impact on headache-related disability and quality of life of people with headache. Cephalalgia, 38(4), 639-645.
Gulliford, M. (2020). Opioid use, chronic pain and deprivation. EClinicalMedicine, 21.
Guitera, V., Muñoz, P., Castillo, J., & Pascual, J. (2002). Quality of life in chronic daily headache: a study in a general population. Neurology, 58(7), 1062-1065.
Hartvigsen, J., Hancock, M. J., Kongsted, A., Louw, Q., Ferreira, M. L., Genevay, S., ... & Smeets, R. J. (2018). What low back pain is and why we need to pay attention. The Lancet, 391(10137), 2356-2367.
Horton, B. T. (1941). The use of histamine in the treatment of specific types of headaches. Journal of the American Medical Association, 116(5), 377-383.
Humphreys, K. (2017). Avoiding globalisation of the prescription opioid epidemic. The Lancet, 390(10093), 437-439.
INCB. (2015). Availability of Internationally Controlled Drugs: Ensuring Adequate Access for Medical and Scientific Purposes.
James, S. L., Abate, D., Abate, K. H., Abay, S. M., Abbafati, C., Abbasi, N., ... & Abdollahpour, I. (2018). Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet, 392(10159), 1789-1858.
Jürgens, T. P., Gaul, C., Lindwurm, A., Dresler, T., Paelecke-Habermann, Y., Schmidt-Wilcke, T., ... & Leinisch, E. (2011). Impairment in episodic and chronic cluster headache. Cephalalgia, 31(6), 671-682.
Karcioglu, O., Topacoglu, H., Dikme, O., & Dikme, O. (2018). A systematic review of the pain scales in adults: which to use?. The American journal of emergency medicine, 36(4), 707-714.
Katsarava, Z., Mania, M., Lampl, C., Herberhold, J., & Steiner, T. J. (2018). Poor medical care for people with migraine in Europe–evidence from the Eurolight study. The journal of headache and pain, 19(1), 10.
Kirthi, V., Derry, S., Moore, R. A., & McQuay, H. J. (2010). Aspirin with or without an antiemetic for acute migraine headaches in adults. Cochrane database of systematic reviews, (4).
Klapper, J. A., Klapper, A., & Voss, T. (2000). The misdiagnosis of cluster headache: A nonclinic, population‐based, Internet survey. Headache: The Journal of Head and Face Pain, 40(9), 730-735.
Knaul, F. M., Farmer, P. E., Krakauer, E. L., De Lima, L., Bhadelia, A., Kwete, X. J., ... & Connor, S. R. (2017a). Alleviating the access abyss in palliative care and pain relief—an imperative of universal health coverage: the Lancet Commission report. The Lancet, 391(10128), 1391-1454.
Knaul FM, Farmer PE, Krakauer EL, De Lima L, Bhadelia A, Jiang Kwete X, Arreola-Ornelas H, et. al. (2017b). Technical Note and Data Appendix for “Alleviating the access abyss in palliative care and pain relief—an imperative of universal health coverage: the Lancet Commission report”. Background Document. Miami: University of Miami Institute for Advanced Study of the Americas. Available at: miami.edu/lance
Knaul, F., Bhadelia, A., Atun, R., De Lima, L., & Radbruch, L. (2019). Palliative care: an essential facet of universal health coverage. The Lancet Global Health, 7(11), e1488.
Koehler, P. J. (1993). Prevalence of headache in Tulp's Observationes Medicae (1641) with a description of cluster headache. Cephalalgia, 13(5), 318-320.
Kongsted, A., Kent, P., Axen, I., Downie, A. S., & Dunn, K. M. (2016). What have we learned from ten years of trajectory research in low back pain?. BMC musculoskeletal disorders, 17(1), 1-11.
Krakauer, E. L., Kwete, X., Verguet, S., Arreola-Ornelas, H., Bhadelia, A., Mendez, O., ... & Connor, S. (2017). Palliative care and pain control.
Law, S., Derry, S., & Moore, R. A. (2013). Triptans for acute cluster headache. Cochrane Database of Systematic Reviews, (7).
Lemeunier, N., Leboeuf-Yde, C., & Gagey, O. (2012). The natural course of low back pain: a systematic critical literature review. Chiropractic & manual therapies, 20(1), 33.
Leone, M., D’amico, D., Frediani, F., Moschiano, F., Grazzi, L., Attanasio, A., & Bussone, G. (2000). Verapamil in the prophylaxis of episodic cluster headache: a double-blind study versus placebo. Neurology, 54(6), 1382-1385.
Leone, M., & May, A. (Eds.). (2020). Cluster Headache and other Trigeminal Autonomic Cephalgias. Cham: Springer International Publishing.
Liese, B. H., Gribble, R. S., & Wickremsinhe, M. N. (2019). International funding for mental health: a review of the last decade. International health, 11(5), 361-369.
Linde, M., Steiner, T. J., & Chisholm, D. (2015). Cost-effectiveness analysis of interventions for migraine in four low-and middle-income countries. The journal of headache and pain, 16(1), 15.
Linde, K., & Rossnagel, K. (2004). Propranolol for migraine prophylaxis. Cochrane Database of Systematic Reviews, (2).
Lynch, M. E., Schopflocher, D., Taenzer, P., & Sinclair, C. (2009). Research funding for pain in Canada. Pain Research and Management, 14.
May, A. (2005). Cluster headache: pathogenesis, diagnosis, and management. The Lancet, 366(9488), 843-855.
May, A., Schwedt, T. J., Magis, D., Pozo-Rosich, P., Evers, S., & Wang, S. J. (2018). Cluster headache. Nature Reviews Disease Primers, 4(1), 1-17.
Melzack, R. (1975). The McGill Pain Questionnaire: major properties and scoring methods. Pain, 1(3), 277-299.
Merriman, A., & Harding, R. (2010). Pain control in the African context: the Ugandan introduction of affordable morphine to relieve suffering at the end of life. Philosophy, Ethics, and Humanities in Medicine, 5(1), 1-6.
Messac, L. (2020). The other opioid crisis – people in poor countries can’t get the pain medication they need. The Conversation.
Mol, L., Ottevanger, P. B., Koopman, M., & Punt, C. J. A. (2016). The prognostic value of WHO performance status in relation to quality of life in advanced colorectal cancer patients. European Journal of Cancer, 66, 138-143.
MSH. (2015). International Medical Products Price Guide
Murray, S. A., Grant, E., Grant, A., & Kendall, M. (2003). Dying from cancer in developed and developing countries: lessons from two qualitative interview studies of patients and their carers. Bmj, 326(7385), 368.
NICE. (2012). Headaches: Diagnosis and management of headaches in young people and adults.
NICE. (2016). Low back pain and sciatica in over 16s: assessment and management.
NIH. (2018). Federal Pain Research Strategy Overview.
Noseda, R., & Burstein, R. (2013). Migraine pathophysiology: anatomy of the trigeminovascular pathway and associated neurological symptoms, cortical spreading depression, sensitization, and modulation of pain. PAIN®, 154, S44-S53.
Organisation for the Prevention of Intense Suffering [OPIS]. (2020). Legalising Access to psilocybin to end the agony of cluster headaches [Policy Paper].
Ogboli-Nwasor, E. O., Makama, J. G., & Yusufu, L. M. D. (2013). Evaluation of knowledge of cancer pain management among medical practitioners in a low-resource setting. Journal of pain research, 6, 71.
Pearson, S. M., Burish, M. J., Shapiro, R. E., Yan, Y., & Schor, L. I. (2019). Effectiveness of oxygen and other acute treatments for cluster headache: results from the Cluster Headache Questionnaire, an International Survey. Headache: The Journal of Head and Face Pain, 59(2), 235-249.
Perry, B. A., Westfall, A. O., Molony, E., Tucker, R., Ritchie, C., Saag, M. S., ... & Merlin, J. S. (2013). Characteristics of an ambulatory palliative care clinic for HIV-infected patients. Journal of palliative medicine, 16(8), 934-937.
Pinkerton, R., & Hardy, J. R. (2017). Opioid addiction and misuse in adult and adolescent patients with cancer. Internal Medicine Journal, 47(6), 632-636.
Portenoy, R. K., & Lesage, P. (1999). Management of cancer pain. The Lancet, 353(9165), 1695-1700.
Rabbie, R., Derry, S., & Moore, R. A. (2013). Ibuprofen with or without an antiemetic for acute migraine headaches in adults. Cochrane database of systematic reviews, (4).
Rajagopal, M. R., Joranson, D. E., & Gilson, A. M. (2001). Medical use, misuse, and diversion of opioids in India. The lancet, 358(9276), 139-143.
Rajagopal, M. R., & Joranson, D. E. (2007). India: Opioid availability—an update. Journal of pain and symptom management, 33(5), 615-622.
Rajagopal, M. R. (2015). The current status of palliative care in India. Cancer Control, 22, 57-62.
Rajagopal, M. R., Karim, S., & Booth, C. M. (2017). Oral morphine use in south India: a population-based study. Journal of Global Oncology, 3(6), 720-727.
Rhee, J. Y., Garralda, E., Namisango, E., Luyirika, E., De Lima, L., Powell, R. A., ... & Centeno, C. (2018). An analysis of palliative care development in Africa: A ranking based on region-specific macroindicators. Journal of pain and symptom management, 56(2), 230-238.
Rhee, J. Y., Garralda, E., Torrado, C., Blanco, S., Ayala, I., Namisango, E., ... & Centeno, C. (2017). Palliative care in Africa: a scoping review from 2005–16. The Lancet Oncology, 18(9), e522-e531.
Rozen, T. D., & Fishman, R. S. (2012). Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache: The Journal of Head and Face Pain, 52(1), 99-113.
Rudd, R. A., Aleshire, N., Zibbell, J. E., & Gladden, R. M. (2016). Increases in drug and opioid overdose deaths—United States, 2000–2014. Morbidity and mortality weekly report, 64(50 & 51), 1378-1382.
Peiffer, C. (2011). Morphine-induced relief of dyspnea: what are the mechanisms? American Journal of Respiratory and Critical Care Medicine, 184(8).
Salisbury-Afshar, E. (2014). Topiramate for the prophylaxis of episodic migraine in adults. American Family Physician, 90(1), 24-24.
Salomon, J. A., Haagsma, J. A., Davis, A., de Noordhout, C. M., Polinder, S., Havelaar, A. H., ... & Murray, C. J. (2015). Disability weights for the Global Burden of Disease 2013 study. The Lancet Global Health, 3(11), e712-e723.
Sharkey, L. (2017). Increasing Access to Pain Relief in Developing Countries - An EA Perspective. EA Forum.
Sharkey, L., Loring, B., Cowan, M., Riley, L., & Krakauer, E. L. (2018). National palliative care capacities around the world: results from the World Health Organization Noncommunicable Disease Country Capacity Survey. Palliative Medicine, 32(1), 106-113.
Singer, P. (2018). Prisoners of Pain. Project-syndicate.
Sleeman, K. E., de Brito, M., Etkind, S., Nkhoma, K., Guo, P., Higginson, I. J., ... & Harding, R. (2019). The escalating global burden of serious health-related suffering: projections to 2060 by world regions, age groups, and health conditions. The Lancet Global Health, 7(7), e883-e892.
Spencer, D. C., Krause, R., Rossouw, T., Moosa, M. Y. S., Browde, S., Maramba, E., ... & Mahomed, A. (2019). Palliative care guidelines for the management of HIV-infected people in South Africa. Southern African Journal of HIV Medicine, 20(1), 1-26.
Steiner, T. J., Birbeck, G. L., Jensen, R., Katsarava, Z., Martelletti, P., & Stovner, L. J. (2010). Lifting The Burden: the first 7 years.
Steiner, T. J., & Stovner, L. J. (Eds.). (2019). Societal Impact of Headache: Burden, Costs and Response. Springer Nature.
Steiner, T. J., Stovner, L. J., Vos, T., Jensen, R., & Katsarava, Z. (2018). Migraine is first cause of disability in under 50s: will health politicians now take notice?.
Stovner, L. J., Nichols, E., Steiner, T. J., Abd-Allah, F., Abdelalim, A., Al-Raddadi, R. M., ... & Edessa, D. (2018). Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet Neurology, 17(11), 954-976.
Wertli, M. M., Eugster, R., Held, U., Steurer, J., Kofmehl, R., & Weiser, S. (2014a). Catastrophizing—a prognostic factor for outcome in patients with low back pain: a systematic review. The Spine Journal, 14(11), 2639-2657.
Vijay, D., & Monin, P. (2018). Poisedness for social innovation: The genesis and propagation of community-based palliative care in Kerala (India). Management, 21(4), 1329-1356.
Vos, T., Abajobir, A. A., Abate, K. H., Abbafati, C., Abbas, K. M., Abd-Allah, F., ... & Aboyans, V. (2017). Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet, 390(10100), 1211-1259.
Wertli, M. M., Rasmussen-Barr, E., Weiser, S., Bachmann, L. M., & Brunner, F. (2014b). The role of fear avoidance beliefs as a prognostic factor for outcome in patients with nonspecific low back pain: a systematic review. The spine journal, 14(5), 816-836.
WHO. (2019). WHO Model Lists of Essential Medicines.
WHO. (2011). Atlas of headache disorders and resources in the world 2011.
WHO. (2004). Atlas: country resources for neurological disorders 2004 : results of a collaborative study of the World Health Organization and the World Federation of Neurology.
WHO. (2017). Atlas: Country resources for neurological disorders 2017.
WHO. (2016). Global Atlas of Palliative Care at the End of Life.
Wiffen, P. J., Wee, B., Derry, S., Bell, R. F., & Moore, R. A. (2017). Opioids for cancer pain‐an overview of Cochrane reviews. Cochrane Database of Systematic Reviews, (7).
Wiffen, P. J., Wee, B., & Moore, R. A. (2016). Oral morphine for cancer pain. Cochrane Database of Systematic Reviews, (4).
Appendix 1 - Pain intensities and effect of treatment
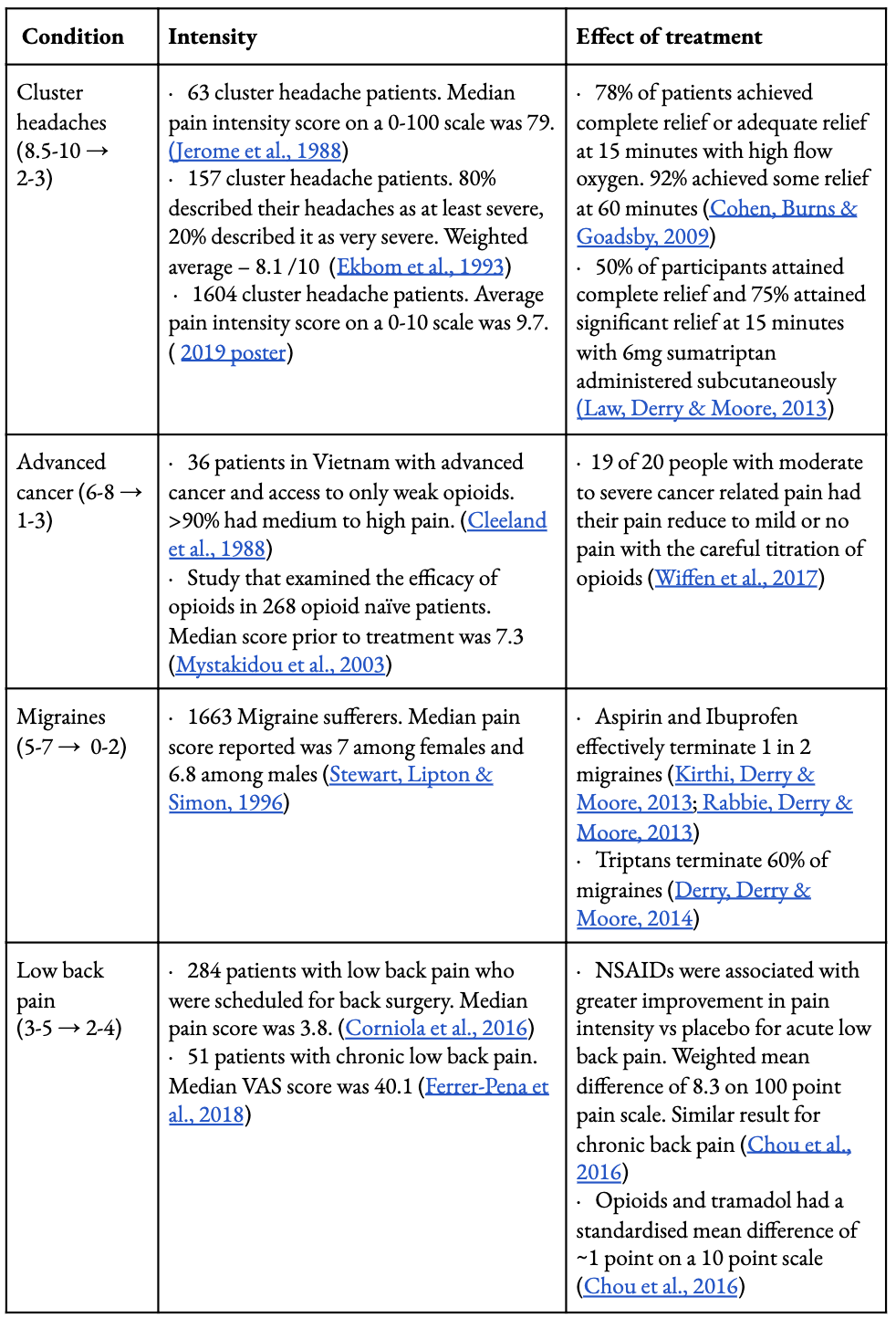
See here to access links in table
Appendix 2 - DALY estimates
Moderate-severe pain that could be treated with opioids during terminal conditions
LC’s seminal paper (Knaul et al., 2017) quantifies the burden of serious health-related suffering associated with a need for palliative care and pain relief. Serious health-related suffering encompasses physical suffering (pain, shortness of breath, nausea and vomiting, and constipation) and psychological (anxiety, depression, confusion and dementia) suffering. The LC analysed the 20 health conditions that cause most of the burden of serious health-related suffering; 49% of the total estimated days of serious health-related suffering are from cancer.[40]
LC estimated the total annual burden of serious health-related suffering days to be between 6 billion and 21 billion days, where the upper bound does not assume overlap (e.g. the types of suffering are experienced on separate days) and the lower bound assumes overlap, calculated by only accounting for the symptom experienced for the longest duration (Knaul et al., 2017a, see Panel 6).
We used LC’s lower bound serious health-related suffering days to approximate DALYs, given the Global Burden of Disease generally calculates disease burden and health loss assuming symptom overlap. Our upper bound incorporates serious health-related suffering caused by moderate-severe pain, mild pain and shortness of breath (1.74 billion days or 4.77 million years) and the disability weight for terminal cancer (0.57).
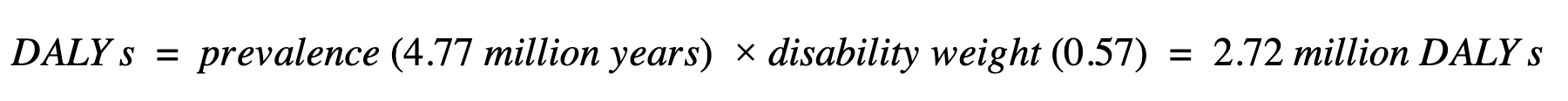
However we recognise not all mild pain and shortness of breath would be best treated with opioids. Hence we only consider moderate- severe pain (0.36 billion days or 0.99 years) for our lower bound.
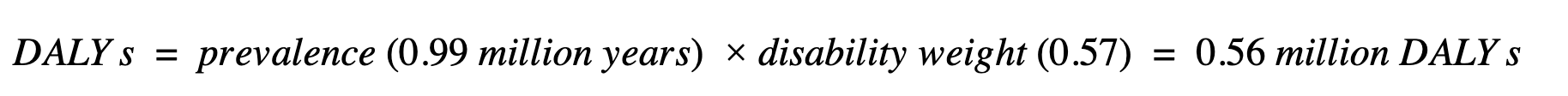
Our choice of the disability weight could be contested, as not all serious health-related suffering moderate to severe pain days would be experienced by patients at the terminal stage, or have cancer. However we felt it would be inappropriate to use a disability weight less than the migraine disability weight (0.441), as we consider diseases causing moderate to severe pain and requiring opioids to be more severe. Further, our DALY estimate would not be particularly sensitive to a disability weight between 0.441 and 0.56.
Cluster headaches
To estimate the global DALYs for cluster headaches, we use the global DALYs for migraines as a reference point. To adjust for prevalence, we multiplied migraine DALYs (47 million) by the ratio of the prevalence of cluster headaches (0.1%; Ekbom et al., 2006; Leone & May, 2020) to migraines (14%; Stovner et al., 2018), naively assuming that the active headache time period the demographic distribution and age distribution is equivalent for migraine and cluster headache sufferers.
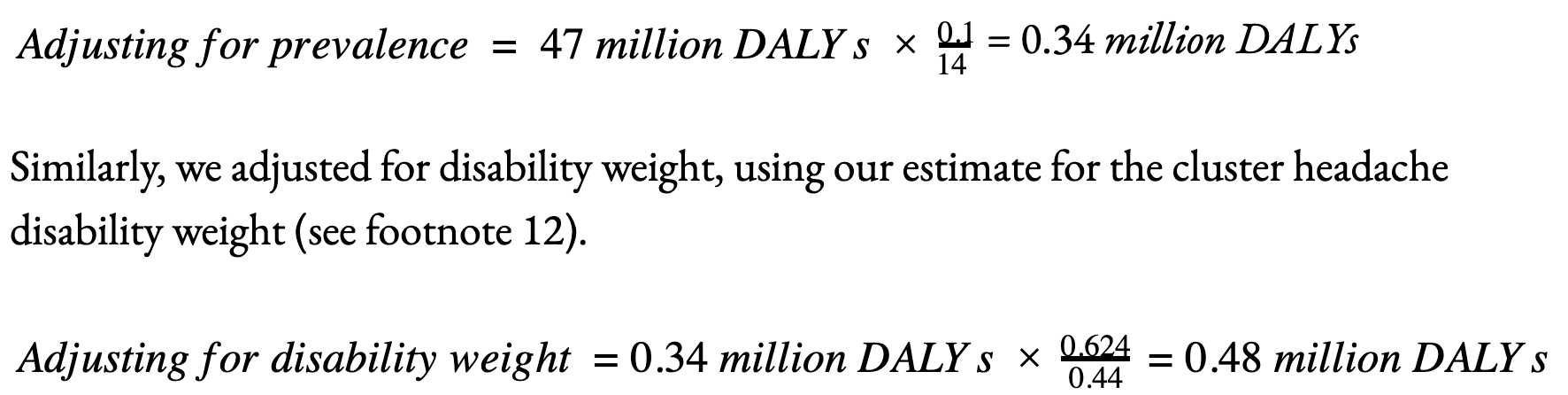
Sid Sharma conducted the research and literature review, prepared expert interviews, and wrote the report. Clare Donaldson managed the project, participated in expert interviews, and contributed to the report draft. Michael Plant provided additional guidance and contributed to the report draft. ↩︎
Acknowledgements: Thank you to the experts we spoke to: Jonathan Leighton, Jim Cleary, Afsan Bhadelia, and M.R Rajagopal. Thanks also go to John Halstead, Lee Sharkey, and Barry Grimes for providing feedback and to HLI’s funders for supporting this work. ↩︎
We use the phrase ‘terminal conditions’ to refer to both ‘life-limiting conditions’ (where the person will die with the condition and their lifespan will be shortened) and to ‘life-threatening conditions’ (where the person will not necessarily die with the condition but it has the potential to cause premature death, particularly with limited access to treatment), that requires palliative care. Opioids may be needed for both life-threatening and life-limiting conditions. ↩︎
We use the term opioids in the report to refer to a broad group of pain-relieving drugs that act on opioid receptors in the brain. Common types of opioid used in medical settings include morphine, oxycodone, fentanyl, and buprenorphine. ↩︎
Disability-Adjusted Life Years lost in 2019, according to the Global Burden of Disease, were 42 million for migraines, 46 million for malaria, and 47 million for depression (source). ↩︎
For example, GiveWell recommends charitable donations such as cash transfers and anti-malarial bed-nets, which could be described as ‘micro’ interventions. The kind of interventions described in this report, such as advocating for access to opioids, are ‘macro’ interventions. It is challenging to make a comparison of cost-effectiveness and out of the scope of this report. ↩︎
For example, this Effective Altruism Forum post by Lee Sharkey and this op-ed by Peter Singer raise the issue of access to opioids, and this post by Qualia Computing discusses cluster headaches. ↩︎
Also see the Happier Lives Institute’s post in which we use WELLBYs to estimate the relative value of averting deaths and reducing poverty. ↩︎
Jonathan Leighton drew our attention to this research poster by Burish et al. that surveyed patients with cluster headaches, and asked them to rate how painful (a limited number of) different experiences were. ↩︎
We use advanced cancer to represent terminal conditions that require opioids for pain relief because it is the largest contributor (38%) of the moderate to severe pain that could be treated with opioids during palliative care (Knaul, 2017a). ↩︎
If a quantitative exercise like this were to be used in order to decide where to spend resources, there are some potentially serious limitations to bear in mind. Firstly, there are concerns that verbal numerical rating scores for pain are not linear (Loadsman and Craigie, 2019), although there is some evidence that they are linear (Miles 1999; Miles and Urquhart, 2005). It has also been suggested that the relationship between reported and experienced pain is in fact logarithmic i.e. that going from 8/10 to 9/10 represents a bigger increase than going from 1/10 to 2/10.The size of the relative difference would depend on the exact logarithmic relationship (Emilsson, 2019). Plant (2020) provides a theoretical rationale for why subjective scales are, in general, likely to be linear (and, more broadly, interpersonally and intertemporally cardinal) and argues that subjective well-being data support this hypothesis. However, it is possible that individuals report on pain scales differently from subjective well-being or other subjective scales. Finally, reported pain intensities may change considerably depending on the methods the interviewer uses, the population investigated, or the way the scale is anchored and framed (Karcioglu et al., 2018). ↩︎
For a discussion of and guidelines for measuring subjective well-being, see e.g. OECD (2013) ↩︎
A societal measure of disease burden that is derived by combining years of life lost (YLL) and years lived in disability (YLD) as a consequence of a disease. ↩︎
Descriptions of health states are used to estimate the disability weight for a disease via paired comparison questions. Members of the public are asked to consider lay descriptions of two hypothetical people, each with a particular health condition and to specify which person they regard as being healthier than the other. See Salomon et al., 2015 for more details ↩︎
The Global Burden of Disease (GBD) did not produce a disability weight for cluster headaches (James et al., 2017). We crudely estimated the disability weight attributable to cluster headaches, by using results from a study (Jurgens et al., 2010) that examined the impairment that cluster headaches and migraines caused an individual using the Headache Disability Inventory (a headache-specific impairment questionnaire). Patients with active cluster headaches had a mean Headache Disability Inventory score of 59.4, compared to 42.0 for patients with migraines (Table 2, Jurgens et al., 2010). We then multiplied the proportionate difference in impairment by the disability weight GBD derived for a migraine to arrive at a disability weight for cluster headaches. Other quality of life studies confirm the significant impairment cluster headaches give rise to, with subjects consistently reporting scores much worse than the general population and others with primary headache disorders (D’Amico et al., 2020). Due to the different assessments used to evaluate the quality of life of people living with cluster headaches, we chose not to integrate the results of these studies into our quantitative estimate given the added complexities it gives rise to. ↩︎
Note that for all conditions except malaria, the DALY contribution is entirely made up of Years Lived with Disability ↩︎
Serious health-related suffering is a construct developed by LC to capture the burden of suffering related to life-limiting or life threatening illness that requires palliative care (Knaul et al., 2017a). It encompasses physical (e.g. pain, shortness of breath, constipation, nausea, and vomiting) and psychological (e.g. anxiety, depression, confusion, and dementia) suffering. LC derived estimates for the number of patients experiencing serious health-related suffering and the number of days of serious health-related suffering in a stepwise manner that involved considering the top twenty conditions that most often generate serious health-related suffering, the proportion of those who experience serious health-related suffering, the type and incidence of serious health-related suffering associated with each condition and the duration of each type of suffering. All steps were informed by literature reviews and assessed by an expert panel (see data appendix, Knaul et al., 2017b). They used 2015 WHO global health estimates mortality data as a foundation for their estimates, and considered both those who died in 2015 (decedents) and those who did not but experienced serious health-related suffering (non-decedents) to better capture the burden of serious health-related suffering. This constitutes the first approximation of the burden of serious health-related suffering and as such was not intended to provide a robust set of point estimates, but rather accuracy within an order of magnitude ↩︎
Thanks to Founders Pledge for the suggestion of how to estimate the DALYs lost due to pain requiring opioids ↩︎
We do not show these in Figure 2 because it is not an exact like-for-like comparison; DALYs aim to measure the burden of all symptoms from a certain cause, we are only considering pain that could be treated with opioids in palliative care. ↩︎
For a full list of the resources required to provide a basic level of palliative care care see Krakauer et al., 2018. ↩︎
The exact mechanism for how opioids provide relief from shortness of breath is unknown. It is postulated that it may work through reducing the perception of air hunger or the workload of breathing (Peiffer, 2011). ↩︎
Common side effects include nausea, vomiting, constipation, fatigue and drowsiness (Wiffen et al., 2017). ↩︎
To estimate opioid requirements for serious health-related suffering, LC considered the amount of opioids required for each of the 20 conditions based on clinical judgement and literature reviews, and combined this with their previous estimate for the burden of serious health-related suffering (Knaul et al., 2017a). Again, this was intended to provide accuracy in an order of magnitude. They estimated opioid needs met based on INCB data, which measures distributed opioid morphine-equivalent (DOME) by determining opioids that were available in a country in a given year and delivered to a health facility. DOME serves as a useful proxy, but it is likely to be an overestimate of the moderate to severe pain met by opioids as it does not decipher opioids that were actually dispensed and consumed by patients. ↩︎
John Halstead, in correspondence, notes one response to this discrepancy would be just to focus on economic growth, rather than pain relief, and expect pain relief to follow. See this Effective Altruism Forum post for a suggestion along these lines. It is not obvious that this less direct route would be more effective, or that it would be easier to make this comparison, but we note the possibility. ↩︎
LC considered the median DOME of Western Europe as an appropriate benchmark for opioid need, that is inclusive of opioid need outside of serious health-related suffering. Canada, USA and Australia all have substantially higher levels of DOME than the Western Europe benchmark. ↩︎
It is hard to define what constitutes overly restrictive regulations, as it's dependent on the number of regulations, the type of regulations, the other barriers to opioids, and the dynamic relationship between these factors in specific contexts. However, Cleary et al. (2013) outline eight 'restrictive domains' which provide some indication of which countries, and how many countries, have onerous regulations. Of 25 African countries: 7 have 6-7 of these restrictions, 12 have 4-5, and 6 have 2-3. ↩︎
This is assuming the opioid need is the same per capita across states in India. If India only meets 4% of its opioid need for serious health-related suffering with 0.11mg ME per capita as per LCs estimate, then one could extrapolate Kerala meets 52% of its need with 1.426mg ME per capita (Knaul et al., 2017b; Rajagopal, Karim & Booth, 2016). ↩︎
From 23 to 31 S-DDD (defined daily dose for statistical purposes) per million per day between 2001 and 2013 ↩︎
S-DDD must be interpreted carefully given the inherent imprecision with population measures, and the limited capacity national authorities have to provide accurate estimates ↩︎
Tension-type headaches are the most common form of headache disorders, producing a dull or heavy band of pain usually on both sides of the head They’re less severe and disabling than migraines. Medication-overuse headaches, as the name implies, are a type of headache disorder that develops in patients with a pre-existing headache disorder after an overuse of headache abortive agents. ↩︎
A group of abortive medications used to treat migraines and cluster headaches. Common triptans include Zolmitriptan, Rizatriptan and Sumatriptan. ↩︎
Unnecessary investigations are costly, and the number of investigations serves as a proxy for the delivery of high-quality care (i.e. it would be expected that doctors confident in their knowledge of headache disorders would order fewer investigations). ↩︎
The earliest description by the 17th century Dutch physician, Dr Nicolas Tulp, illustrated a patient feeling as if they would succumb if the headache continued any longer (Koehler, 1993). In 1941, Dr Horton discussed a case series of cluster headaches, in which nearly all his 72 cases contemplated suicide (Horton, 1941). ↩︎
One large US survey reported 55% of sufferers had thoughts of ending their life, with 2% actually attempting to do so (Rozen & Fishman, 2012). Another study that recruited 193 patients across 15 hospitals in Korea found that 36% of patients had active suicidal thoughts during an attack and 2% reported suicidality between a bout (Lee et al., 2019). ↩︎
The precise mechanism in which triptans and oxygen abort a headache is unknown. Triptans are thought to trigger the activation of serotonin receptors leading to the constriction of blood vessels supplying parts of the brain that are implicated in the pathophysiology of cluster headaches (Law, Derry & Moore, 2013). It is proposed that oxygen has a similar effect on blood vessels, in addition to modulating inflammatory pathways around involved structures (Bennet et al., 2015). ↩︎
The remaining 3/15 patients did not experience a reduction in headache frequency over a 2-week period compared to a placebo. ↩︎
An inactive treatment or procedure that is intended to mimic as closely as possible a therapy in a clinical trial. ↩︎
For more, see e.g. GiveWell ↩︎
See this Wikipedia entry for a primer. ↩︎
See Table 3 in Knaul et al. (2017); cancer is referred to as malignant neoplasms. ↩︎

I'm pleased to see this work.
Obviously childbirth accounts for relatively few hours of one's life spent in pain, but I wonder if you've looked into it. Nitrous oxide is safe and relatively cheap and does not need an anesthetist because the patient can administer it themselves. It's commonly used in some Western countries but only getting started in the US, and I can't find anything about its use in middle or low income countries.
Hi Julia, thanks for drawing our attention to this! No, we didn’t look into it. From a quick review of the evidence, the effectiveness appears variable and uncertain. But as you rightly point out, it doesn't require an anaesthetist, and it could therefore play an important role in a low resource setting.
To me "Inhaled analgesia appears to be effective in reducing pain intensity and in giving pain relief in labour" sounds like a ringing endorsement from Cochrane given that their usual bottom line seems to be "not enough evidence." Just about anything for childbirth pain seems to be pretty hit-or-miss, so the fact that it's not that effective for some people seems like not that big of a downside compared to other methods or no method.
Yes, good point! Based on the Cochrane review, we could confidently say nitrous oxide has an effect on pain. I should have said the evidence for the degree of pain relief appears variable and uncertain, although on a closer look, Cochrane's estimate suggests 3.5 on a 0-10 scale vs placebo (which is pretty great).
Hi Julia, I've spoken to several women who said that they received absolutely no pain relief from the epidurals they were given during birth- none had ever returned to the hospital to give feedback as they had more pressing concerns. Has anyone ever systematically asked women whether their epidural worked? Any data?
Yes, there's been a lot of research on this. Here's an analysis of 40 trials with 11,000 women.
My guess is that epidurals are unlikely to be the most cost-effective intervention to increase access to, since it requires an anesthesiologist. I was guessing that nitrous oxide might be more promising because it's so easy to administer.
I wrote a short blog post a little while ago on preventing low back pain with exercise. I think your problem area report might have missed several important meta-analyses on low back pain. In particular, Huang et al., 2018 and Shiri, Coggon and Hassani, 2017 seem to supersede Steffens et al., 2017, and Lin et al., 2018 seems broader and more recent than NICE, 2016. I think your assessment of the quality of evidence in favour of exercise interventions for low back pain might reasonably update with respect to these references.
Lower back pain cause is simple—we are not using our great ape bodies in the way that evolution designed them to be used. Our ancestors hunted and gathered. They moved their bodies every day and for much of the day walking, running, crouching squatting, grabbing etc. Any seats certainly didn't have backrests. All of our ancestors cores were in great shape because they were working them out nearly every waking moment. Our desire to obtain comfort has led us to "convenience ourselves to death".