12 articles including 4 podcasts
EA/LW Intro: I believe clinical trial abundance could be an EA cause area - there's still a lot of disability/disease burden in the world, even in developed countries, and increasing the pace of progress is very tractable. And it's not just a matter of speed/quantity of innovation: the current system selects against ambitious risky bets. It deserves an EA-specific post, but for now here's a curated reading list.
Since the 1950s, the cost of developing a new drug has increased by ~80x. It now costs on the order of a billion dollars to get one drug approved (including the cost of failures). Consequently, fewer drugs get invented, ambitious but risky areas are avoided, and patients pay the price.
Why have clinical trials gotten so expensive, and what can we do about it? Why isn't Big Pharma interested in diseases like ME/CFS and Long COVID? Why won't advanced AI automatically lead to biomedical breakthroughs?
There's a growing movement of researchers, policy wonks, and patient advocates trying to answer these questions and fix what's broken. It's loosely organized under the banner "Clinical Trial Abundance." Here's what to read to understand it.
1. The pharma industry from Paul Janssen to today: why drugs got harder to develop and what we can do about it - by Alex Telford, 2023
This was my original point of entry to Clinical Trial Abundance. It's a pretty long article but covers a lot of history as well as many of the important concepts and dynamics.
He describes how the field moved from small, quick (and sometimes very unethical!) trial-and-error to large preclinical research projects trying to predict drug efficacy before even moving to clinical trials that altogether take many years.
I'm a big fan of his blog, but unfortunately he's mostly not writing anymore.
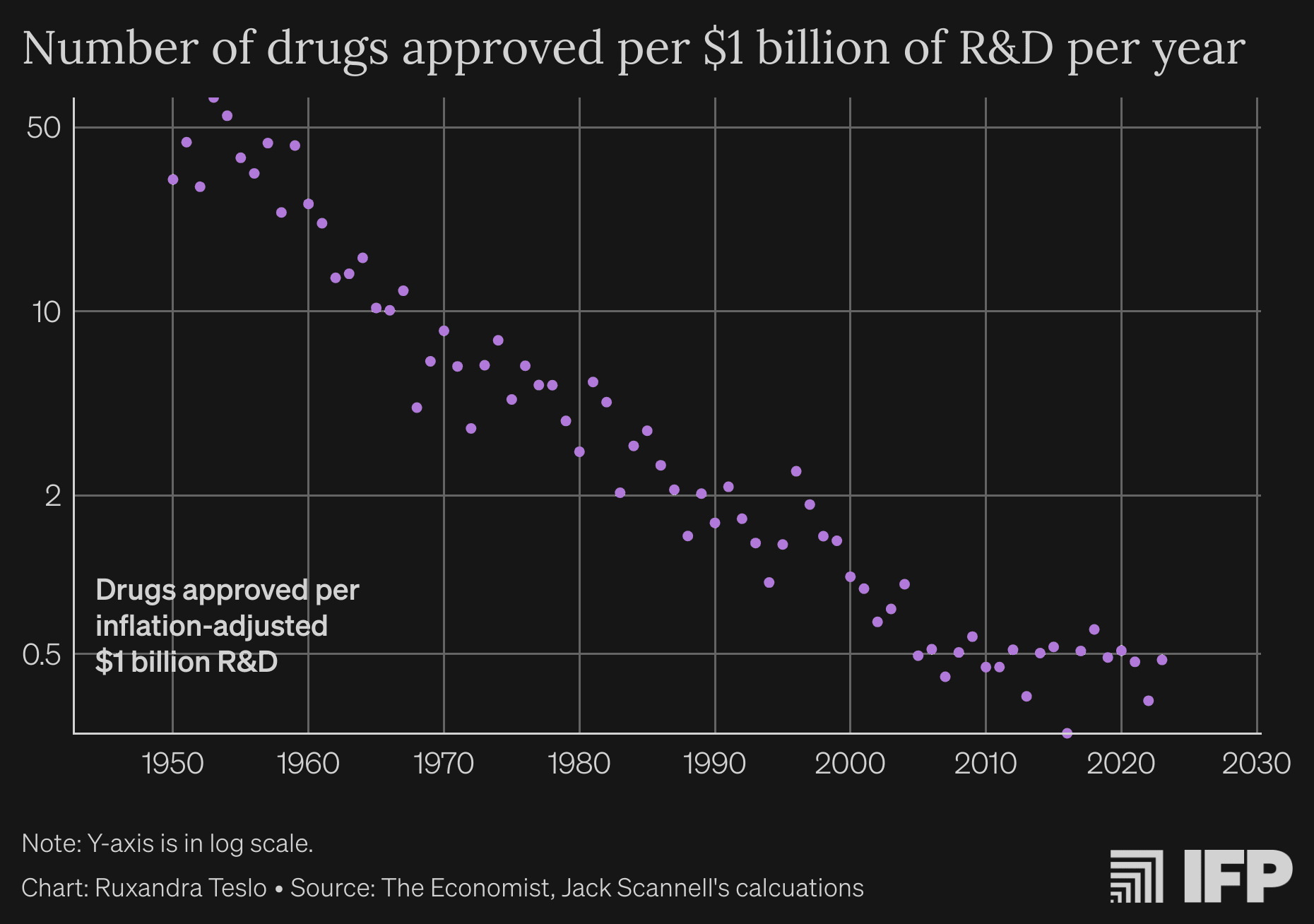
2. Diagnosing the decline in pharmaceutical R&D efficiency - by Jack Scannell et al., 2012
Probably the OG of this field, Scannell et al. identified the trend that drug development has become exponentially more expensive over time and coined the term Eroom’s Law for this - the opposite of Moore’s Law (which refers to chips/computing power becoming exponentially less expensive over time).
An updated version of the original graph, sourced from the next article on the list!
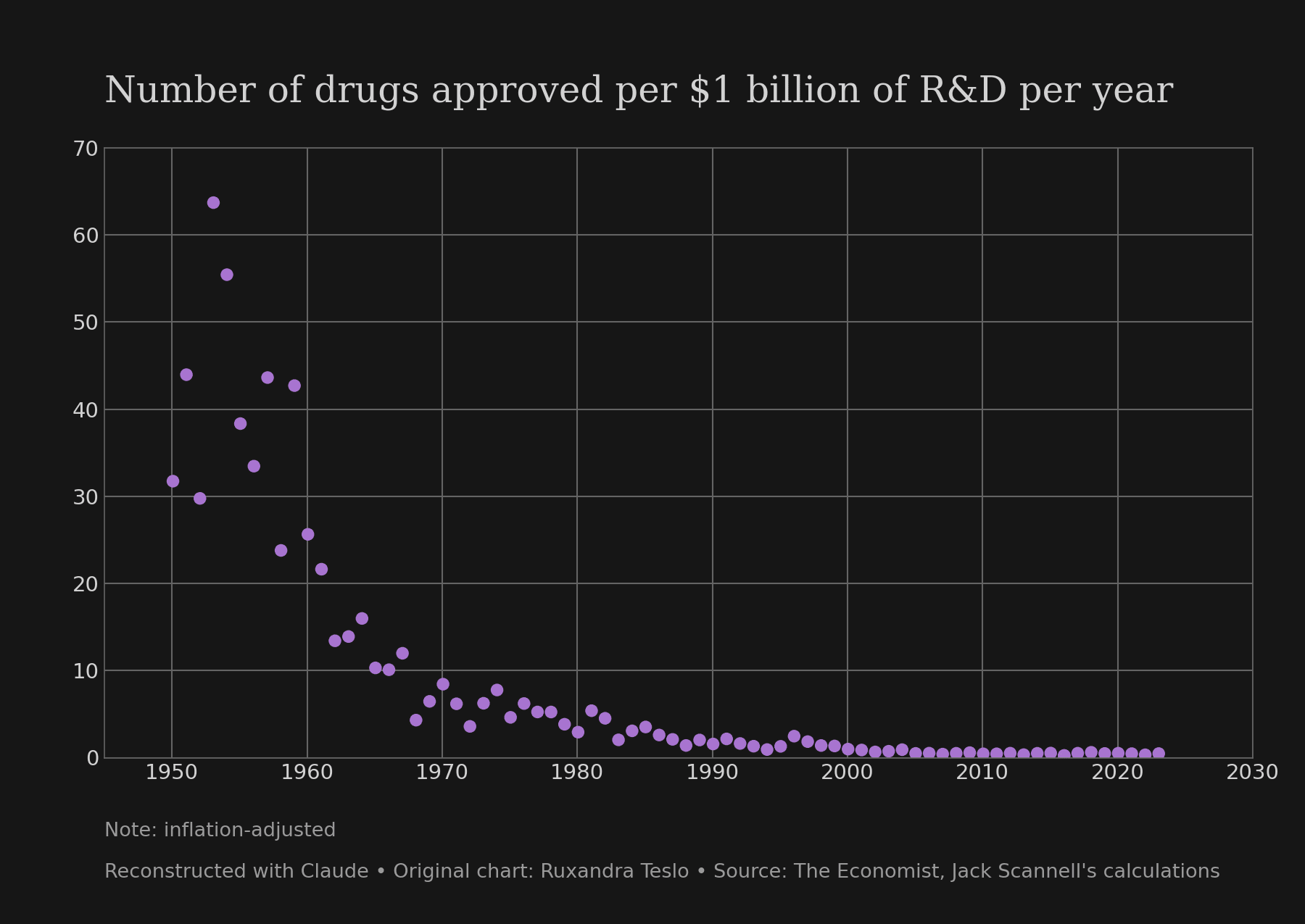
Now, that's a pretty dramatic and continuous trend.[1] But to drive the point home, I used Claude to transpose that graph onto a linear y-axis:
Note that this is about R&D efficiency, not total output. Companies have been able to invest much more into R&D than before, offsetting efficiency losses.
They hypothesized 4 factors that would cause the decline in R&D efficiency:
- better than the Beatles' problem: as standard of care becomes more effective, it's harder and harder to develop even better treatments
- the 'cautious regulator' problem: regulators get punished for negatives and little reward for positives, making them risk averse.
- the 'throw money at it' tendency: no explanation needed (though I'm skeptical about this factor)
the 'basic research-brute force' bias: companies have put evermore effort into prediction and yet “the probability that a smallmolecule drug successfully completes clinical trials has remained more or less constant for 50 years” (a strong claim that I'm not sure about[2])
3. To Get More Effective Drugs, We Need More Human Trials - by Ruxandra Teslo and Jack Scannell, 2025
Ruxandra is arguably the driving force and leader of the Clinical Trial Abundance project. I highly recommend subscribing to her Substack.
This is a good, brief introduction to Clinical Trial Abundance and why it's so important. In it, she also dispels two myths: the myth that we just need a libertarian approach to drug approval, as well as the myth that AI will magically solve everything. Both have the same myth-busting reason: we still need to rigorously test medicine in humans to find out whether it's effective.
4. Why clinical trials are inefficient. And why it matters - by Adam Kroetsch, 2025
I really enjoyed this recent post. Adam worked at the FDA and has a lot of insight into the dynamics at big pharma companies.
His main point is that trials are expensive because each one is seen as a unique, one-off project, rather than an engineering task that needs to be standardized and ruthlessly optimized for efficiency. Think Space Shuttle vs. SpaceX rockets.
He also argues that companies’ risk aversion is not just the result of regulation, and there are opportunities for entrepreneurs to run cheaper, leaner trials if they cultivate the right consumer niche.
5. Clinic-in-the-Loop - by Ruxandra Teslo in Asimov Press, 2025
In this short article, Ruxandra Teslo argues that the goal of Clinical Trial Abundance is not just about moving more drugs through the funnel, but about creating a tighter feedback loop with clinical trials helping to build our understanding of human diseases. I wrote a comment with more examples of (unexpected) lessons we got from trials.
Audio version here.
5. Interview with the CEO of Eli Lilly, Dave Ricks - by Patrick and John Collison
This is a podcast (Spotify) with a transcript. It's 2 hours long and very interesting. Obviously Ricks has his biases that the listener needs to be aware of.
I learned many things and still need to dig into some things he said. For example, when they purchase a compound they often do a whole other innovation loop bringing an optimized compound to market, but why? They also discuss trial enrollment being a major obstacle, Institutional Review Board fragmentation, and how to incentivize one-off treatments.
(An important fact to understand why the discussion is so US-centric is that 60% of revenues come from there!)
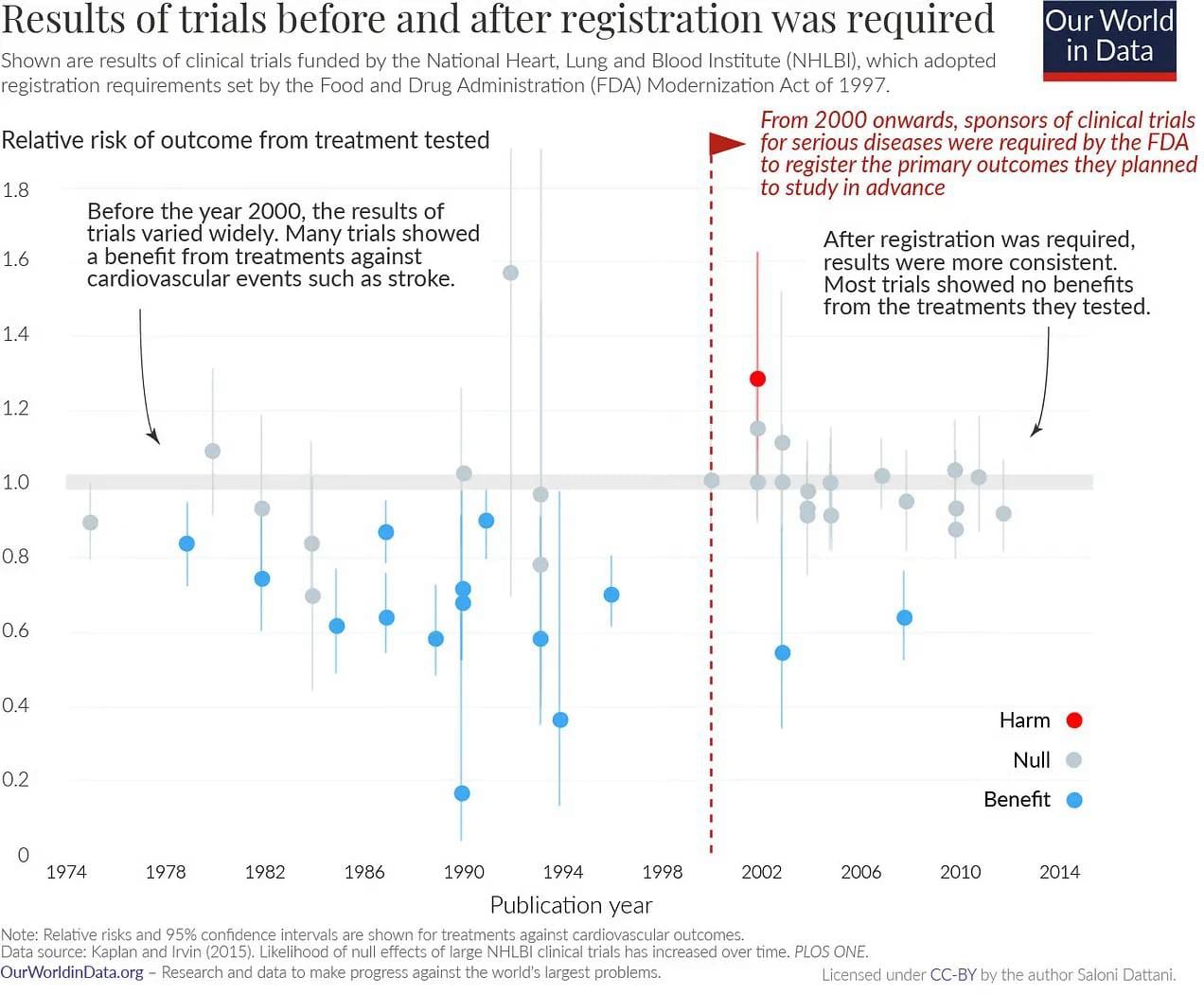
6. Clinical trial reforms that once seemed radical - by Saloni Dattani
Saloni Dattani often writes about the history of medicine on Our World in Data or discusses it on the podcast Hard Drugs. Now she writes for the Clinical Trial Abundance blog, a recently launched Substack by a number of the authors featured in this list.
This post discusses
- the rise of RCTs
- the rise of protocol preregistration
- the rise of trial results actually getting posted
.. and argues we shouldn't treat the current system as the end of history: changes that seem radical at first can quickly become seen as obviously good once implemented.
When you pre-register the primary outcomes of a trial, it becomes much harder to spin the results positively!
This post also suggests that a substantial part of rising development costs has been the rising bar for evidence, not a bad thing!
7. Biotech's Lost Archive - by Ruxandra Teslo, 2025
Not all inefficiency is driven by overregulation. Perhaps a bigger factor is regulatory uncertainty. The decision-making of regulators is opaque. For companies, it's unclear which data will be necessary and sufficient for approval, which experiments to do, which outcomes to track. As a consequence, they try to cover all their bases and become very risk averse.
Teslo’s solution: buy the Common Technical Documents of failed companies when they dissolve, then publish them. This includes all experiments done, why they've done it, and all their interactions with and guidance from the FDA/EMA.
She also talks about it on this great Patrick MacKenzie podcast (transcript here).
8. Why More Biotechs Are Starting Trials In Australia - by Dan Schell, 2025
In Australia, Phase 1 trials are much faster and cheaper, and have been so for 3 decades without any meaningful costs to safety. This brief, industry-oriented article describes how:
- The regulator doesn't need to approve the running of a trial. The system relies only on Institutional Review Boards
- Good Manufacturing Practices (GMP), the quality control of the medications, is less stringent for early trials
- Financial & tax incentives
- A weaker currency
Contains links to 9 essays with concrete ideas for improvement. Proposals include
- streamlining consent forms to reduce bureaucratic burden
- human challenge trials: intentionally exposing people to pathogens
- requiring the FDA to publish redacted Complete Response Letters (currently, the letters are fully private)
10. Some questions about biotech that I find interesting - by Alex Telford, 2024
As Alex was winding down his writing, he wrote up a long list of 27 questions he still has with some short thoughts on each of them. Great food for thought! I especially liked the 2 papers studying how much public funding it costs to get to 1 approved drug (median estimates of $400M - $700M in 2010-dollars, with large uncertainty intervals).
Here's a 47min podcast interview about it if you prefer listening, but it doesn't cover everything.
11. Clinical Trial Abundance (policy framework) - by 1DaySooner
This isn't really an essay. It's a framework with a lot of policy proposals by the organization 1DaySooner. From what I can tell, they originally came from the effective altruism network trying to speed up covid vaccine approvals by advocating for human challenge trials: letting people volunteer to be infected after being vaccinated, because this is much faster than needing to vaccinate and monitor tens of thousands of people and wait for natural infections. Now they have broadened their remit to pandemic preparedness and clinical trial abundance.
I have a Substack where I write about solving diseases — especially poorly understood ones like ME/CFS and POTS/OI, which have disabled me since 2020. Questions I focus on are:
- How can we make clinical trials cheaper and more effective?
- What makes for good science and how can we increase the quality of a field?
- What makes for effective patient advocacy?
- What does the evidence support regarding how ME/CFS works?
Find me on Twitter as @PatientPersists. Everything written with brain fog and limited spoons.
Subscribe- ^
The trend may have plateaued since ~2005. Maybe we can now start reversing it?
- ^
They support this claim of unchanged approval rates with a link to this research: DiMasi et al. (2010) Trends in risks associated with new drug development: success rates for investigational drugs. However, that only compares two six-year periods (1993-1998 & 1999-2004), not 50 years.

I feel like clinical trial abundance has at least the whiff of a new cause area within global health. I'm curating this post as a way to kickstart the issue a little bit.
I agree with the author that "It deserves an EA-specific post", i.e. I'd love to see someone make the case for clinical trial abundance as a cause area. It could be compared to economic growth/ progress work (as a more targeted niche) or to the distribution of existing medicines (vaccines for malaria etc...).
Thanks for writing this @Siebe!
Executive summary: The author argues that rising clinical trial costs and inefficiencies are a major, tractable bottleneck to biomedical progress, and curates a reading list supporting the “Clinical Trial Abundance” view that expanding and improving trials could accelerate innovation.
Key points:
This comment was auto-generated by the EA Forum Team. Feel free to point out issues with this summary by replying to the comment, and contact us if you have feedback.