Summary
- Life history classification will hide some significant differences in the lives of wild animals. Not all species within a given classification possess all of the traits associated with that group even across all years or all locations. Therefore, when making moral decisions, one also has to consider how average quality of life should be determined in the face of large variance
- Among insect herbivores, some lifespans are relatively long, some modes of death are very quick, and some small-bodied herbivores may lead lives characterized by ample food resources
- Although determining the affective states of wild animals from this data is impossible, it seems quite likely that the majority individuals in some subgroups, such as those sheltered from both the elements and predation by feeding from within plant tissues, lead very high quality lives
- Knowing a group of organisms produce many offspring, have high mortality rates, small body size and are short-lived is not sufficient to determine that their lives are a net negative (or positive)
The argument from life history suggests that since many species produce many more offspring than survive to adulthood, are of small size and so subject to many abiotic and biotic threats, and are short-lived relative to humans, that there is more suffering than happiness in nature and therefore we have a moral obligation to end this suffering (e.g., Tomasik 2015). Here, we do not attempt to examine such moral quandaries (for a thought experiment on these issues see Brennan 2017). Instead, we aim to improve the quality of discussions by examining available data on one group of wild animals. Because of their broad scope, some previous analyses of wild animal welfare have lumped many species together at one pole of a continuum of life history strategies. Given these large groupings, and some issues with the life history classifications themselves, it is unclear to what extent this approach actually informs us about animal suffering in general. On the other hand, examinations of the lives of particular species are also unsatisfactory, since they refer to the specific rather than the majority. By narrowing our focus to one group, we may be able to bring more data to bear on our intuitions regarding wild animal welfare. In this post, we explore the literature regarding one group of organisms that are classically grouped in the “r-strategist” life history category: terrestrial insect herbivores.
Terrestrial insects are described by some as having net negative welfare. They are one of the largest groups of terrestrial animals in terms of number of species, biomass and estimated number of individuals. As May (1988) noted, “To a rough approximation, and setting aside vertebrate chauvinism, it can be said that essentially all organisms are insects.” Bar-On et al. (2018) estimate the biomass of terrestrial arthropods at 0.2 gigatons of carbon, and insects are the largest classification in the arthropod phylum, accounting for over 80% of all described species in this group (Zhang 2013). For example, Hölldobler & Wilson (1990) suggest that 15-20% of the biomass of terrestrial animals is composed of ants. Insects are ~1/3 of all described species on the earth (Grimaldi & Engel 2005), while estimates for the total number of insect species range between five and seven million (e.g., Stork 2018).
Various approximations of the total number of insect individuals range between 10^17-10^19 (Williams 1960 , Hölldobler & Wilson (1990), Bar-On et al. (2018)). As small-bodied primary consumers, herbivores will be at the bottom of the trophic energy transfer in any community. Since only ~10% of primary production passes between trophic levels, this group and detritivores will make up ~90% of all terrestrial insects. Assuming equal division between these two groups and 1018 insect individuals, terrestrial insect herbivores will then comprise approximately 10^17 individuals. In other words, this is one of the largest groups of terrestrial animals on the earth.
Life history classification of terrestrial insects
Terrestrial insects are often grouped near the “r” or “fast” end of life history classifications (see our prior post on life history). The r- and K-selected classification scheme is based on the different evolutionary pressures in uncrowded, resource rich environments and crowded resource poor environments. (MacArthur and Wilson 1967). K-selected species will frequently experience resource scarcity and starvation, while r-selected species will experience an abundant environment. Extreme r-strategies are small-bodied and short-lived, reach reproductive maturity early, reproduce once (semelparous), have a large number of offspring at that time, do not provide parental care, and have high juvenile mortality (Pianka 1970). The fast-slow categorization of life history that has superseded r- and K- classification has similar traits for “fast” species. However, there will always be exceptions: eggplant lace bugs provide extensive parental care (Tallamy 1999), and cicadas can live for nearly 2 decades (Simon 1988). Moreover, some species have life histories that are not captured by these dichotomous classifications. In particular, those in harsh abiotic conditions may have longer lifespans, slower growth rates, and lower reproductive output, because of a larger investment in survival adaptations (e.g., arctic wooly bears Kukal & Dawson 1989).
Analyses of terrestrial insect herbivore life history
Here we report on available literature data regarding fecundity, mortality and lifespan in an effort to provide some quantitative insight regarding the lives of terrestrial insect herbivores. We conclude that variability in life history is the primary characteristic of this group, but certainly these species are, on average, shorter-lived, more fecund, and less likely to provide parental care than mammals.
Life span
Both r-selected and fast life history classifications describe insects as short-lived, and most do live less than two years. Life spans of insects, however, are quite variable. Carey (2001) notes that the between-group variation is enormous: for herbivores, this range includes aphids with a lifespan of weeks, to xylem-feeding beetles that take several years to reach maturity, to termite queens that can live for decades. This 5000-fold difference in the life spans of insects can be contrasted with the smaller 40-fold difference in the life spans of mammals: from small rodents that live less than 2 years to humans that live an average ~80 years.
It is incorrect to believe that life span for any insect species is a single fixed age. The lifespan of a monarch butterfly (Danaus plexippus) is approximately 2-3 months when it is in the reproductive mode during the summer months but 6-10 months when it is in the migratory mode during the winter months (Grace 1997). Some commentators have confused the length of a single life stage with that of the entire lifespan of an individual. For example, while some mayfly adults live only a few hours, mayfly nymphs take longer, up to two years, to mature. For some species (e.g., Lepidoptera: butterflies and moths), these juvenile stages tend to be the largest proportion of the total lifespan in temperate regions (e.g., see data in Danks 2006 for adult life spans). For others, (e.g., Coleoptera: beetles), the adult life stage tends to be the longest. For species without distinct larval and pupal stages, the prereproductive stage also tends to be shorter than the adult stage (e.g., Hemiptera: true bugs). We note that these are all very coarse generalizations over very large groups. There are about 177,500 described species of Lepidoptera, 400,000 of Coleoptera, and 79,000 of Hemiptera.
Across insect groups, parental care, monogamy, and eusociality (at least for queens) are all associated with extended life spans. As well, species that must seek out host plants that are scarce or widely dispersed tend to be long-lived (e.g., Heliconius butterflies have widely-dispersed host plants and only lay only a few eggs at a time. These species are long-lived for lepidopterans in temperate locations, sometimes exceeding 4–6 months (Ehrlich 1987, Gilbert 1972).
As ectotherms, total lifespan and development time are also related to temperature for insects. For example, at 10 °C Aphis gossypii take 75 days to reach reproductive maturity, and live a maximum of 103 days, while at 30 °C they take 5 days to reach reproductive maturity and live a maximum of 37 days (Kocourek et al 1994). In addition, increased longevity can be associated with periods of resource limitation. Extreme life extensions, such as diapause for more than 10 years, usually affect only a very small fraction of the population, and have only been recorded in about 64 species (Gill et al. 2017). However, more modest extensions, such as development over 2 years when 1 year is more normal, are relatively common (Danks 1992).
In addition, species that are subject to uncertain or harsh environments frequently exhibit extended longevity associated with extended or repeated diapause. Overwintering diapause normally lasts for 9-10 months in the temperate zones (Gill et al. 2017). However, Convey (1997) reports extended lifespan in Antarctic arthropods as compared to their close phylogenetic relatives in more temperate regions (e.g., 2-5 years vs ~1 year) associated with repeated diapause over several winters. However, lifespan can be shortened or lengthened to deal with a brief summer season in polar regions (Danks 2004). Given this, we generally expect increased variability in lifespan with increasing latitude and also altitude (e.g., Laiolo & Obeso 2017).
Overall, very short lifespans (less than 20 days) seem fairly rare (Danks 2006), and long lives (>3 years) are rarer still. Species from cool temperate regions tend to have longer life cycles with about one generation per year (e.g., Danks and Foottit 1989), as do species living in areas that have a dry season. But we note that for many of these species, variable environmental conditions determine how many generations there are per year, and in addition, the overwintering generation will have a longer lifespan than growing season generations.
Fecundity
The number of offspring produced by terrestrial herbivores varies with body size, parental care, and the environment. While infant care is not common among insect species, it certainly occurs, and is negatively correlated with reproductive output as expected from a fast-slow life history classification. Gilbert and Manica (2010a) examined fecundity data for 220 non-caring, 23 offspring guarding, and 32 offspring provisioning terrestrial insects (Note: this analysis was not restricted to herbivores, but did exclude eusocial species). Fecundity is defined as the number of offspring produced over the lifetime of an individual. Dung beetles species had the lowest lifetime fecundity (~2 offspring), while mayflies had the largest (~4000 offspring). For terrestrial herbivores in this dataset, the range extends from 12-15 offspring for some leafminers and wood-boring beetles to ~3000 for cutworms. The median lifetime fecundity (measured as number of eggs per individual) was 138 but varied with the type of parental care (166 eggs for no care, 66 for guarding and 40 for provisioning insects; all values calculated from Gilbert and Manica 2010b; but note the authors did not identify the number of eggs that were actually viable).
Contrary to the fast-slow life history classification, fecundity often, although not always, positively correlates with body size in insects (see Leather 1988 for discussion). In species where parents provide no care or simply guard eggs (e.g., the hibiscus harlequin bug Tectocoris diophthalmus aggressively defends newly laid eggs, Giffney & Kemp 2016), larger-bodied species produced more and larger eggs. In species that provision offspring, such as burrower bugs that provide mint nutlets to their nymphs (Sehirus cinctus, see Kight 1997), those with larger bodies also produced larger eggs but laid fewer (Gilbert and Manica 2010a). In this respect, the life histories of provisioning insects resembled those of birds or mammals (traditionally viewed as slow or K-selected species) rather than those of related species that invested less in parental care.
In addition to the form of parental care and body size, environment also has an impact on insect fecundity. Herbivores that feed on the plant exterior have higher fecundities (Cornell & Hawkins 1995), but this finding is partially confounded by the fact the external feeders tend to have larger body sizes than species that feed from within plant tissues (e.g., endophytics such as gall formers, stem borers and leaf miners). More generally, plant quality can alter the fecundity of insect herbivores (Awmack & Leather 2002).
Temperature also determines fecundity, for example mean total fecundity of Aphis gossypii ranges from 36 larvae per female at 10 °C to 76 larvae at 30 °C. (Kocourek et al. 1994). Broader environmental conditions also impact fecundity through both effects on time to reproductive maturity and egg viability. For example, the developing larvae of the arctic woolly bear moth, Gynaephora groenlandica, feed on a northern willow species only in June when ambient temperatures are relatively high and the host plant has the highest nutrient content. The larvae then leave the plant until the following summer. Because of the short growing season, the moth larvae take between 7-14 years to reach maturity (Kukal & Dawson 1989, Morewood and Ring 1998), and females have low lifetime fecundity, producing ~ 5 viable eggs (Kukal & Kevan 1987).
Juvenile Mortality
Survival trajectories for juvenile insect herbivores are quite variable, but do show a trend of higher mortality for younger stages. The most frequent cause of mortality is predators and parasitoids. Cornell & Hawkins (1995) examined 530 datasets for 124 species of herbivorous insects that had 4 distinct stages: egg, larvae, pupae and adult regarding the survival of the juvenile stages to determine the most frequent causes of death. Sprayed, caged, or laboratory populations were excluded from analyses, as were insects that had no distinct pupal stage (i.e., these are species such as beetles and butterflies, but not aphids). The most common cause of death across both groups is predators and parasitoids followed by weather, plant defences and competition (Fig 1).
However, the authors find wide variation in survival trajectories regardless of whether the same species and population was sampled within the same year in different locations or between years. Between species, this variance is even larger. For example, in most cases 50% of eggs of Dacus oleae, the olive fruit fly, survived to adulthood, while less than 5% of cabbage root fly (Erioschia brassicae) eggs did. If an average across this wide variance is taken, there are similar survival trajectories of juveniles across species groups, with slightly higher rates of mortality occurring in the early life stages and slightly lower rates in the later ones. Sources of mortality shift as herbivores grow. Physiological factors, weather and plant factors more frequently kill early stages, whereas parasitoids and predators more frequently kill later stages.
External feeders were more likely to be killed by parasitoids and predators, while plant factors were more likely to kill species that feed from inside the plant tissue (endophytic species). This group of herbivores had higher survival in the older juvenile stages than species that fed from the outside of the plant (Cornell & Hawkins 1995). On average about 70% of eggs became larvae, 20-30% of eggs became late stage larvae (depending on whether the species was endophytic or not), 5-20% became pupae and 2-10% of eggs reached adult stage. These findings broadly support Price's (1974) earlier claim of lower mortality in larvae concealed in plant tissue, but not the claim that this group exhibited convex survival curves, with most mortality occurring past the mid- to late larval stage.
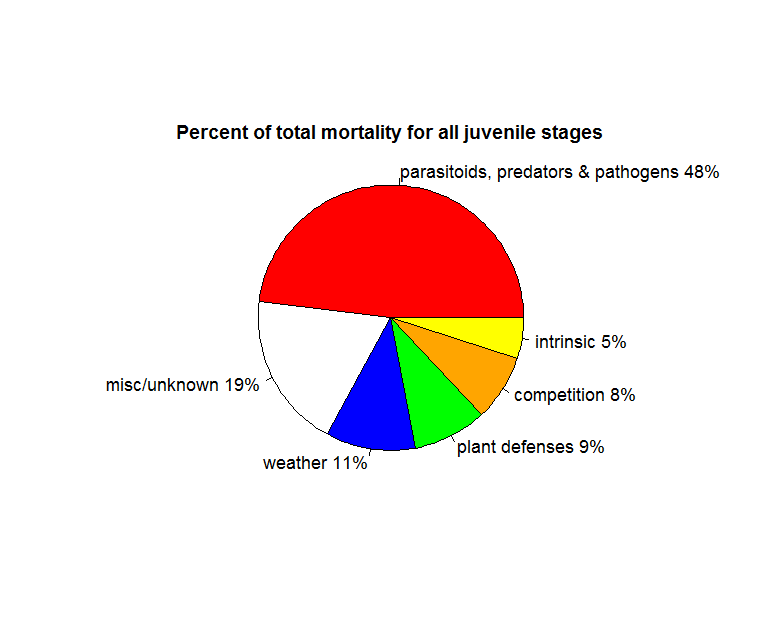
Figure 1: Summary of the frequency of mortality factors for all juvenile insect herbivores in Cornell & Hawkins (1995).
Competition
The initial idea behind r-selection, that species on this end of the spectrum are not limited by resources may be supported by Cornell and Hawkins’s (1995) analysis of mortality. Competition is a very small factor in total juvenile mortality, and the vast majority of mortality events are caused by predators and parasitoids. The weather, intrinsic factors related to reproduction (egg viability) and plant defences also explained more mortality than competition. This lower impact of competition is possibly because there are very few species that reach very large population sizes (e.g, Ayres and Lombardero 2000, Faeth 1987). In addition, even among these species, such population outbreaks are rather rare, usually local and of short duration (Kozlov & Zvera 2017).
On the other hand, meta-analyses consistently find evidence of negative effects of one insect herbivore species on another (Kaplan & Denno 2007, Bird et al. 2019); however, the suggested mechanisms for this impact are predominantly indirect effects (e.g., inducing stronger plant defences and increasing predator populations). Direct lethal impacts caused by food shortages are considered less common. Moreover, in a meta-analysis of 75 published studies Vidal & Murphy (2018) found that predators and parasitoids have a larger impact on herbivore abundance, survival, growth and fecundity than plant quality. There are also significant beneficial effects of insect herbivores species on each other (e.g., by overwhelming host plant defences, distracting natural enemies, or by creating shelters such as leaf mines and rolls; Cornelissen et al. 2016, Soler et al. 2012), so that there is no necessary direct relationship between increasing numbers of herbivores and negative impacts.
Predation, pathogens and parasitoids
Natural enemies (parasitoids, predators and pathogens) emerged as the most frequent mortality factor (48% of all deaths) in Cornell and Hawkins (1995), surpassing every other source of mortality over all life stages and, in some cases, exceeding all others combined. These factors were a larger source of mortality in later juvenile stages. In a study of 78 insect herbivore species with a range of 100-1000 individuals for each, Hawkins et al. (1997) report close to 0% median percent mortality for eggs and early larval stages from natural enemies, although this statistic is partially due to the extremely low mortality among endophytic species. In this study, natural enemies killed a median of 1% of mid larval stage individuals, 3% of late stage and 5% of pupal stage.
Hawkins et al. (1997) find that overall ~4% of all individuals are killed by parasitoids, ~1% by predators and less than 0.5% by pathogens. These authors report that many herbivores, particularly species feeding within plant tissues, suffer little or no mortality from pathogens. So while the authors note that disease can be important in some groups of insects (e.g., forest Lepidoptera Myers 1993), on average it does not seem represent an important mortality source in phytophagous insect populations. Hawkins et al. (1997) also find that parasitoid deaths are more common in temperate regions than in tropical regions, where predators have a somewhat larger impact. In addition, both Cornell and Hawkins (1995) and Hawkins et al. (1997) report that endophytic herbivores suffer lower mortality by predators. Leaf miners suffer the greatest parasitoid-induced mortality, while gall-formers, stemborers and root feeders suffer the least. Hawkins et al. (1997) note that for koinobiont parasitoids (see below), attack will occur in earlier larval stages, while death will occur later. So herbivore mortality rates increase through time, where this mortality includes both both delayed mortality from koinobionts as well as immediate mortality by later attacks from predators and idiobiont parasitoids.
Predators
True predators are less specialized than parasites and parasitoids, and they usually catch prey smaller than themselves (Griffiths 1980). It has been suggested that the impact of death by predation on the net value of a life may be partially determined on the length of the predation event with respect to length of life. Since insect life spans are extremely variable, so are the length of juvenile stages. However, as noted by Plant (2016), death by predation is often a relatively small proportion of even a very short life span. For example, video capture of a generalist predator Harmonia axyridis (Asian multi-coloured ladybug) feeding on aphids suggests consumption rates of 20-60 aphids in a one hour period, implying that each prey individual was consumed in a couple of minutes (Feng et al. 2019). In contrast Michálek et al. (2017) used high speed photography to capture hunting tactics in both a specialist and generalist spider species. Specialist predators tend to consume relatively large prey. The authors recorded body part consumption rates, and if we assume that prey death occurred after maximum consumption time by the specialist predator, then the prey would experience about 300 minutes of predation. Using these times, and noting that the majority of juvenile mortality by predation occurs in the late larval to pupal stage, which for the shorter lived species would be on the order of a couple of weeks, the experience of death by a true predator would range from 0.007-1.0% of total lifespan (assuming death at 3 weeks). For species that tended to take longer to mature (e.g., ~1 yr), and assuming that the late larval or pupal stage occurs occurs in the fall, the average estimate would be lower (~0.002-0.3% of total lifespan, assuming death at 3 months).
Parasitoids
Across all stages, parasitoids kill more herbivores than either predators or pathogen (Hawkins et al. 1997). Death by parasitoid is most common late larval and pupal stages (Hawkins et al. 1997). Koinobiont parasitoids (cf Harvey & Malcicka 2016), such as Cotesia vestalis, allow the host to continue feeding and develop so that death takes quite a long time. In contrast idiobionts cause host development to cease once parasitized, either by causing death or paralysis during oviposition. Therefore, juvenile herbivores inoculated by parasitoids can experience a much longer time frame to death (e.g., 12 days in one study). However, this species group is enormous, and it seems difficult to make any generalizations regarding time to death by different classes of parasitoids. Koinobiont parasitoids often represent the most speciose portion of parasitoid complexes (Mills 1994), so it is possible that this is the most common form of death for insect herbivores.
The percentage of time the insect herbivore experiences the effects of the parasitoid will be highly variable, but related to the age at which is parasitized. If we assume inoculation at the most common time of early-mid larval stage, and a koinobiont parasitoid that keeps the larvae alive for another 10 days of development, then ~40% of total lifespan may serve as an estimate for species that mature in about 60 days (death at just over 3 weeks), and ~20% for those that take about 1 year, where late larval or pupation stage occurs at about 4 months (i.e., September - October in northern temperate regions, and therefore death at ~6 weeks). However, whether larvae being eaten by parasitoids feel pain during the experience is unclear, given that both parasitoids and parasites can cause extensive behaviour modification in their hosts (e.g., Chen et al 2017).
Weather
Following unknown or miscellaneous factors, the weather was the second largest known cause of mortality (11%) in Cornell and Hawkins (1995), and this factor had a larger impact on early juvenile states. Weather-generated mortality was due mainly to rainfall and overwintering deaths. The higher frequency of weather-generated mortality in early larval stages is probably due to higher risk of dislodgement by rainfall. The authors suggest that the importance of weather in later stages probably depends on whether these stages overwinter. Of course, species that live within the plant tissues (e.g., gall-formers, stem borers and and leaf-tiers) were less affected by weather than species that live on plant surfaces. Gregarious species are also less likely to be affected by weather conditions. For example, tent caterpillars raise their temperatures by basking in groups and also construct elaborate group shelters (Stamp and Bowers 1990). We also point out that even extremely harsh condition are not necessarily a problem for species adapted to those locations. Arctic woolly bear winter mortality is quite low (~13%) because the larvae seek out well protected locations and have special physiological adaptations for cold hardiness (Kukal et al. 1987, 1989).
Therefore, mortality of juvenile herbivores can be quite high, but then again, the juvenile stage may be the longest life stage for some of these species (e.g., Lepidopterans). Mortality of juvenile herbivorous insects is dominated by parasitoids and predators, in that order, in temperate regions. Endophytic species are less likely to die from these causes (with the exception of leaf miners). Juvenile insect herbivores seem to be seldom short of food (although food quality may vary), and are only occasionally significantly impacted by weather.
Conclusion
This examination of data on insect herbivores suggests that life history classification will hide some significant differences in the lives of wild animals. In particular, claims about large fecundity, lack of parental care and short lives are not correct for all species that are represented as belonging to a single group within a life history classification, nor are they true for all years or all locations of the same species. Therefore, when making moral decisions about the quality of wild animal lives, it seems likely that one also has to consider one’s position regarding how average quality of life should be determined in the face of such large variance.
Setting aside problems with generalization, we note that the mere observation a large group of organisms produce many offspring, have high mortality rates, small body size and are short-lived is still not sufficient to determine that their lives are net-negative. Even if death is very painful, it is unclear that experiences previous to death are not sufficiently pleasurable to compensate. While this possibility has often been dismissed on the grounds that these animals have such a short lifespan, some of the data we explore suggest that some juvenile lifespans are relatively long, some modes of death are very quick, and that small-bodied herbivores may often lead lives characterized by ample food resources. In fact, it seems quite possible that the majority individuals in some subgroups, such as those sheltered from both the elements and predation by feeding from within plant tissues, lead very high quality lives. We are careful to note, however, that attempting to determine the affective states of wild animals from this data is currently impossible.
References
Awmack, C. S., & Leather, S. R. (2002). Host plant quality and fecundity in herbivorous insects. Annual review of entomology, 47(1), 817-844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Ayres, M. P., & Lombardero, M. J. (2000). Assessing the consequences of global change for forest disturbance from herbivores and pathogens. Science of the Total Environment, 262(3), 263-286. https://doi.org/10.1016/S0048-9697(00)00528-3
Bar-On, Y. M., Phillips, R., & Milo, R. (2018). The biomass distribution on Earth. Proceedings of the National Academy of Sciences, 115(25), 6506-6511. https://doi.org/10.1073/pnas.1711842115
Bird, G., Kaczvinsky, C., Wilson, A. E., & Hardy, N. B. (2019). When do herbivorous insects compete? A phylogenetic meta‐analysis. Ecology Letters. https://doi.org/10.1111/ele.13245
Brennan, O. 2017. Infant mortality and the argument from life history. Retrieved 02.05.19 https://was-research.org/blog/infant-mortality-argument-life-history/
Carey, J. R. (2001). Insect biodemography. Annual Review of Entomology, 46(1), 79-110. https://doi.org/10.1146/annurev.ento.46.1.79
Chen, W. B., Vasseur, L., You, M. S., Li, J. Y., Wang, C. X., Meng, R. X., & Gurr, G. M. (2017). Parasitised caterpillars suffer reduced predation: potential implications for intra-guild predation. Scientific reports, 7, 42636. https://doi.org/10.1038/srep42636
Convey, P. (1997). How are the life history strategies of Antarctic terrestrial invertebrates influenced by extreme environmental conditions?. Journal of Thermal Biology, 22(6), 429-440. https://doi.org/10.1016/S0306-4565(97)00062-4
Cornell, H. V., & Hawkins, B. A. (1995). Survival patterns and mortality sources of herbivorous insects: some demographic trends. _The American Naturalis_t, 145(4), 563-593. https://www.journals.uchicago.edu/doi/abs/10.1086/285756
Cornelissen, T., Cintra, F., & Santos, J. C. (2016). Shelter-building insects and their role as ecosystem engineers. Neotropical Entomology, 45(1), 1-12.https://doi.org/10.1007/s13744-015-0348-8
Danks, H. V. (1992). Long life cycles in insects. The Canadian Entomologist, 124(1), 167-187. https://doi.org/10.1007/s13744-015-0348-8
Danks, H. V. (2004). Seasonal adaptations in arctic insects. Integrative and Comparative Biology, 44(2), 85-94. https://doi.org/10.1093/icb/44.2.85
Danks, H. V. (2006). Short life cycles in insects and mites. The Canadian Entomologist, 138(4), 407-463. https://doi.org/10.4039/n06-803
Danks, H. V., & Foottit, R. G. (1989). Insects of the boreal zone of Canada. The Canadian Entomologist, 121(8), 625-690. https://doi.org/10.4039/Ent121625-8
Ehrlich, P .R. (1984). The structure and dynamics of butterfly populations. In R.I. Vane-Wright & P.R. Ackery (Eds.), The Biology of Butterflies (pp. 25-40). London: Academic Press https://books.google.ca/books/about/The_Biology_of_Butterflies.html?id=fP8hAQAAMAAJ&redir_esc=y
Faeth, S. H. (1987). Community structure and folivorous insect outbreaks: the roles of vertical and horizontal interactions. Pgs 135-171. In P. Barbosa & J. Schultz (Eds.), Insect Outbreaks (pp. 135-171). San Diego: Academic Press. https://doi.org/10.1016/C2009-0-02860-8
Feng, Y., Li, Y. D., Liu, Z. G., Yu, X. L., Zhu, G. X., Keller, M., & Liu, T. X. (2019). Behavioural patterns and functional responses of a generalist predator revealed using automated video tracking. Pest Management Science, 75(6), 1517-1526 https://doi.org/10.1002/ps.5314
Giffney, R. A., & Kemp, D. J. (2016). Maternal care behaviour and kin discrimination in the subsocial bug Tectocoris diophthalmus (Hemiptera: Scutelleridae). Austral Entomology, 55(2), 170-176. https://doi.org/10.1111/aen.12164
Gilbert LE. 1972. Pollen feeding and reproductive biology of Heliconius butterflies. Proceedings of the National Academy of Sciences USA 69(6), 1403-1407. https://doi.org/10.1073/pnas.69.6.1403
Gilbert, J. D., & Manica, A. (2010a). Parental care trade-offs and life-history relationships in insects. The American Naturalist, 176(2), 212-226. https://www.journals.uchicago.edu/doi/abs/10.1086/653661
Gilbert J, Manica A (2010b) Data from: Parental care trade-offs and life history relationships in insects. Dryad Digital Repository. https://doi.org/10.5061/dryad.1451
Gill, H. K., Goyal, G., & Chahil, G. (2017). Insect diapause: a review. Journal of Agricultural Science and Technology, 7, 454-473. http://www.davidpublisher.org/Public/uploads/Contribute/5a5c6c5a389c3.pdf
Grace, E.S. (1997). The Nature of Monarch Butterflies. Vancouver, Canada: Greystone Books https://www.amazon.ca/nature-monarch-butterflies-Beauty-flight/dp/1550545701
Greenslade, P. J. (1983). Adversity selection and the habitat templet. The American Naturalist, 122(3), 352-365. https://www.journals.uchicago.edu/doi/abs/10.1086/284140
Griffiths, D. (1980). Foraging costs and relative prey size. The American Naturalist, 116(5), 743-752. https://www.journals.uchicago.edu/doi/abs/10.1086/283666
Grimaldi, D. & Engel, M.S. (2005). Evolution of the Insects. New York: Cambridge University Press https://books.google.ca/books/about/Evolution_of_the_Insects.html?id=Ql6Jl6wKb88C
Grime, J. P. (1977). Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. The American Naturalist, 111(982), 1169-1194. https://www.journals.uchicago.edu/doi/abs/10.1086/283244
Harvey, J. A., & Malcicka, M. (2016). Nutritional integration between insect hosts and koinobiont parasitoids in an evolutionary framework. Entomologia Experimentalis et Applicata, 159(2), 181-188. https://doi.org/10.1111/eea.12426
Hawkins, B. A., Cornell, H. V., & Hochberg, M. E. (1997). Predators, parasitoids, and pathogens as mortality agents in phytophagous insect populations. Ecology, 78(7), 2145-2152. https://esajournals.onlinelibrary.wiley.com/doi/abs/10.1890/0012-9658(1997)078%5B2145:PPAPAM%5D2.0.CO;2
Hölldobler, B., & Wilson, E. O. (1990). The ants. Cambridge, Mass: Harvard University Press. https://books.google.ca/books/about/The_Ants.html?id=o87CQgAACAAJ&redir_esc=y
Kaplan, I., & Denno, R. F. (2007). Interspecific interactions in phytophagous insects revisited: a quantitative assessment of competition theory. Ecology Letters, 10(10), 977-994. https://doi.org/10.1111/j.1461-0248.2007.01093.x
Kight, S. L. (1997). Factors influencing maternal behaviour in a burrower bug, Sehirus cinctus (Heteroptera: Cydnidae). Animal Behaviour, 53(1), 105-112. https://doi.org/10.1006/anbe.1996.0282
Kocourek, F., Havelka, J., Berankova, J., & Jaroŝik, V. (1994). Effect of temperature on development rate and intrinsic rate of increase of Aphis gossypii reared on greenhouse cucumbers. Entomologia Experimentalis et Applicata, 71(1), 59-64. https://doi.org/10.1111/j.1570-7458.1994.tb01769.x
Kozlov, M. V., & Zvereva, E. L. (2017). Background insect herbivory: impacts, patterns and methodology. In: Cánovas F., Lüttge U., & Matyssek R. (Eds) Progress in Botany Vol. 79 (pp. 313-355). Cham: Springer. https://doi.org/10.1007/124_2017_4
Kukal, O., & Kevan, P. G. (1987). The influence of parasitism on the life history of a high arctic insect, Gynaephora groenlandica (Wöcke)(Lepidoptera: Lymantriidae). Canadian Journal of Zoology, 65(1), 156-163. https://doi.org/10.1139/z87-022
Kukal, O., & Dawson, T.E. 1989. Temperature and food quality influence feeding behavior, assimilation efficiency and growth rate of arctic woolly-bear caterpillars. Oecologia 79:526–32 https://doi.org/10.1007/BF00378671
Laiolo, P., & Obeso, J. R. (2017). Life-history responses to the altitudinal gradient. In Catalan, J., Ninot, J. M., & Aniz, M. M. (Eds.). High mountain conservation in a changing world (pp. 253-283). Cham: Springer. https://link.springer.com/chapter/10.1007/978-3-319-55982-7_11
Leather, S. R. (1988). Size, reproductive potential and fecundity in insects: things aren't as simple as they seem. Oikos, 386-389. https://www.jstor.org/stable/3565323
May, R. M. (1988). How many species are there on earth?. Science, 241(4872), 1441-1449. https://science.sciencemag.org/content/241/4872/1441
MacArthur, R. H., & Wilson, E. O. (1967). The theory of island biogeography. Princeton: Princeton University Press. Retrived 23/05/2019 https://books.google.ca/books/about/The_Theory_of_Island_Biogeography.html?id=a10cdkywhVgC
Michálek, O., Petráková, L., & Pekár, S. (2017). Capture efficiency and trophic adaptations of a specialist and generalist predator: a comparison. Ecology and Evolution, 7(8), 2756-2766. https://doi.org/10.1002/ece3.2812
Mills, N. J. (1994). Parasitoid guilds: defining the structure of the parasitoid communities of endopterygote insect hosts. Environmental Entomology, 23(5), 1066-1083. https://doi.org/10.1093/ee/23.5.1066
Morewood, W. D., & Ring, R. A. (1998). Revision of the life history of the High Arctic moth Gynaephora groenlandica (Wocke)(Lepidoptera: Lymantriidae). Canadian Journal of Zoology, 76(7), 1371-1381. https://doi.org/10.1139/z98-085
Myers, J. H. (1993). Population outbreaks in forest Lepidoptera. American Scientist, 81(3), 240-251. https://www.jstor.org/stable/29774919
Pianka, E. R. (1970). On r-and K-selection. The American Naturalist, 104(940), 592-597. https://www.jstor.org/stable/2459020
Plant, M. 2016. The Unproven (And Unprovable) Case For Net Wild Animal Suffering. A Reply To Tomasik Retrieved 02.05.19 https://forum.effectivealtruism.org/posts/guvsD78ZXhfCaT7SH/the-unproven-and-unprovable-case-for-net-wild-animal.
Price, P. W. (1974). Insect ecology New York: Wiley. https://books.google.ca/books/about/Insect_Ecology.html?id=rrzc-IkgNx0C&redir_esc=y
Sæther, B. E. (1987). The influence of body weight on the covariation between reproductive traits in European birds. Oikos, 79-88. https://www.jstor.org/stable/3565691
Simon, C. (1988). Evolution of 13- and 17-year periodical cicadas. Bulletin of the Entomological Society of America, 34, 163–176. https://doi.org/10.1093/besa/34.4.163
Soler, R., Badenes‐Pérez, F. R., Broekgaarden, C., Zheng, S. J., David, A., Boland, W., & Dicke, M. (2012). Plant‐mediated facilitation between a leaf‐feeding and a phloem‐feeding insect in a brassicaceous plant: from insect performance to gene transcription. Functional Ecology, 26(1), 156-166. https://doi.org/10.1111/j.1365-2435.2011.01902.x
Stamp, N. E. and Bowers, M. D. 1990. Variation in food quality and temperature constrain foraging of gregarious caterpillars. Ecology 71, 1031–1039. https://doi.org/10.2307/1937371
Stork, N. E. (2018). How many species of insects and other terrestrial arthropods are there on Earth?. Annual Review of Entomology, 63, 31-45. https://doi.org/10.1146/annurev-ento-020117-043348
Tallamy, D. W. (1999). Child care among the insects. Scientific American, 280(1), 72-77. https://www.jstor.org/stable/26058020
Tomasik, B. (2015). The importance of wild-animal suffering. Relations: Beyond Anthropocentrism, 3, 133. https://doi.org/10.7358/rela-2015-002-toma
Vidal, M. C., & Murphy, S. M. (2018). Bottom‐up vs. top‐down effects on terrestrial insect herbivores: a meta‐analysis. Ecology Letters, 21(1), 138-150. https://doi.org/10.1111/ele.12874
Williams, C. B. (1960). The range and pattern of insect abundance. The American Naturalist, 94(875), 137-151. https://www.journals.uchicago.edu/doi/abs/10.1086/282115
Zhang, Z. Q. (2013). Phylum arthropoda. Zootaxa, 3703(1), 17-26. http://dx.doi.org/10.11646/zootaxa.3703.1.6
Credits

This essay is a project of Rethink Priorities. It was written by Kim Cuddington. Thanks to Jason Schukraft, Daniela Waldhorn, David Moss, Marcus Davis and Peter Hurford for comments. If you like our work, please consider subscribing to our newsletter. You can see all our work to date here.

Very interesting about warm-weather diapause and metabolic rate for mosquitoes. I'll agree that during deep cold-weather diapause insects are reducing metabolic rate (goodness, but maybe not when REALLY cold??). A quick lit search turned up seasonally variable brain size and cognitive abilities in shrews (Lázaro et al. 2018)!
No idea how this relates to lived experience tho. Extending this argument, would you also claim that species with slower metabolism have less lived experience than those with faster metabolism (e.g., "less sentience and less hedonic experience per day"), because then comparing between species with different metabolic rates is going to be quite difficult. In fact I think it quite likely that those species with faster metabolic rates have different lived experience rates than species such as humans, e.g., Healy et al. 2013.
Healy, K., McNally, L., Ruxton, G. D., Cooper, N., & Jackson, A. L. (2013). Metabolic rate and body size are linked with perception of temporal information. Animal Behaviour, 86(4), 685-696. https://doi.org/10.1016/j.anbehav.2013.06.018
Lázaro, J., Hertel, M., LaPoint, S., Wikelski, M., Stiehler, M., & Dechmann, D. K. (2018). Cognitive skills of common shrews (Sorex araneus) vary with seasonal changes in skull size and brain mass. Journal of Experimental Biology, 221(2), jeb166595. https://jeb.biologists.org/content/jexbio/221/2/jeb166595.full.pdf